Abstract
Aberrant induction of human leukocyte antigen-G (HLA-G) expression has been observed in various malignancies and is strongly associated with tumor immune escape, metastasis and poor prognosis. To date, great achievements have been made in understanding the underlying mechanisms of HLA-G involved in tumor progression. HLA-G could lead to tumor evasion by inhibition of immune cell cytolysis, differentiation and proliferation and inhibition of cytokine production, induction of immune cell apoptosis, generation of regulatory cells and expansion of myeloid-derived suppressive cells and by impairment of chemotaxis. Moreover, HLA-G could arm tumor cells with a higher invasive and metastatic potential with the upregulation of tumor-promoting factor expression such as matrix metalloproteinases (MMPs), indicating that ectopic HLA-G expression could render multiple effects during the progression of malignancies. In this review, we summarized the mechanisms of HLA-G involved in promoting tumor cell immune escaping, metastasis and disease progression. Special attention will be paid to its significance as an attractive therapeutic target in cancers.
Similar content being viewed by others
Introduction
The pace of tumor growth is dictated by the continuous interaction between cancer cells and the host immune system. With the selection pressure exerted by immune system, various strategies have been developed and applied by tumor cells to avoid recognition and destruction by different immune effectors (1,2). One of the common strategies used by tumor cells to escape innate and adaptive immune response is associated with an induced aberrant expression of the nonclassical class I molecule human leukocyte antigen-G (HLA-G) (3).
HLA-G is the best characterized nonclassical HLA-class Ib molecules, which includes HLA-E, -F and -H (4). Due to its primary mRNA alternative splicing, seven HLA-G isoforms can be generated, where four of them are membrane-bound (HLA-G1 to -G4) and three soluble (HLA-G5 to -G7) (5). Moreover, another soluble isoform termed “shed HLA-G1” can be generated by proteolytic cleavage of the membrane-bound HLA-G1 by metalloproteinases (6).
HLA-G expression was first observed in cytotrophoblasts (7). In physiological conditions, HLA-G expression was also found in erythroid precursors, cornea, thymic medulla and pancreatic islets (8,9–11). However, HLA-G can be neo-expressed in pathological conditions including cancers, transplantation, and inflammatory and autoimmune diseases and viral infections (3,5,12,13).
In malignancies, ectopic induction of HLA-G expression has been observed in various types of tumors where it was more frequently observed in tumor lesions with advanced stage and its clinical relevance has been addressed (5,14). HLA-G can directly inhibit immune cell function through receptor binding and/or through trogocytosis and impairment of chemotaxis (15,16); moreover, HLA-G can render tumor cells with a higher invasive, metastatic potential and an unfavorable prognosis in tumor patients, indicating that HLA-G expression has multiple effects including promoting tumor cells to escape immune surveillance and enhancing their metastasis during the progression of malignancies (17–19). Therefore, the role of HLA-G in malignancies has gained considerable clinical interest for the possibility of exploiting it as a molecular biomarker and a therapeutic target.
Mechanisms of HLA-G Involved in Tumor Immunology
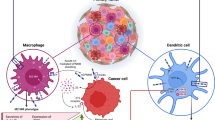
HLA-G expression could render multiple effects during the progression of malignancies, such as inhibiting immune cell cytolysis, inducing immune cell apoptosis and the generation of regulatory cells through receptor binding and/or trogocytosis, and impairing chemotaxis of different immune effector cells (14,16). Several types of HLA-G receptors including immunoglobulin-like transcript 2 (ILT2)/cluster of differentiation 85j (CD85j), ILT4/CD85d, killer cell immunoglobulin-like receptor 2DL4 (KIR2DL4)/CD158d, CD8 and CD160 have been reported. ILT2 is expressed on B cells, some T cells and NK cells and all monocytes and dendritic cells. ILT4 is expressed only by monocytes, dendritic cells and neutrophils. KIR2DL4 is expressed on natural killer (NK) cells, and CD160 expressed by cytotoxic CD8+ T cells and NK cells, a small proportion of CD4+ T cells, as well as endothelial cells. The CD8 is predominantly expressed on the surface of cytotoxic T cells, but can also be found on NK cells (5,20). Moreover, HLA-G could enhance the expression of tumor metastasis-related factors such as matrix metalloproteinases (MMPs), providing profound effects on tumor progression (18,21,22).
HLA-G-mediated inhibition of the functions of both innate and adaptive immune cells depends on cell types and the profiles of HLA-G receptors they expressed. By binding receptors expressed on various cells, HLA-G could directly inhibit the functions of NK cells, cytotoxic T-lymphocyte (CTLs), B cells, neutrophils and dendritic cells (DCs) (5). HLA-G also has long-lasting indirect immunoregulatory activities by inducing tolerogenic cells including HLA-G-expressing T regulatory (Treg) cells, CD4low and CD8low suppressor T cells, T regulatory type 1 (Tr1) cells, myeloid-derived suppressor cells (MDSC) and DC-10 (23–25). Moreover, other immunoinhibitory consequences, including impairment of chemotaxis induced by HLA-G, were also emphasized (26,27) (Figure 1).
The direct immunosuppression induced by HLA-G included inhibition of CTL and NK cell lysis, allogeneic CD4+ T-cell proliferation and the induction of the activated CD8+ T cells and CD8+ NK cell apoptosis (28–31). HLA-G also affects DC maturation, migration, trafficking, antigen presentation and their cross-talk with T and NK cells, and inhibits Vγ9Vδ2 T-cell proliferation and IFN-γ production and cytotoxicity by interacting with the ILT2 inhibitory receptor (32–35). We recently reported that NK cell cytolysis inhibited by HLA-G1 in an expression proportion-dependent manner, and HLA-G1 and HLA-G5 isoforms have an additive inhibitory effect on NK cytolysis (36,37). Indeed, in vitro studies with cell lines of ovarian carcinoma, hepatocellular carcinoma, glioma, melanoma and renal cell carcinoma demonstrated that, upon expression of endogenous HLA-G antigens, tumor cells were protected from lysis by allore-active NK cells and lymphokine-activated killer cells and/or antigen-specific CD8+ T cells, and that this protective effect could be reversed by masking HLA-G antigens using HLA-G or its receptor-specific antibody (18,38–40). It has been demonstrated that soluble HLA-G (sHLA-G) could inhibit B-cell proliferation, differentiation and immunoglobulin (Ig) secretion in both T-cell-dependent and -independent models of B-cell activation through the receptor ILT2 (41). To be noted, HLA-G5 was shown to inhibit phagocytosis and reactive oxygen species production of neutrophils through ILT4 (42).
Indirect inhibitory immune effects of HLA-G could induce the generation of tolerant cells including Treg, DC and MDSC, or by the process of trogocytosis where activated NK cells, T cells and monocytes could also temporarily inhibit immune responses by acquiring HLA-G-containing membranes from cells in their vicinity (43,44). HLA-G-induced Treg cells were observed after allogeneic stimulation by HLA-G1-expressing antigen-presenting cells (APC), which could induce CD4+ T cell anergy and differentiate into suppressive cells (45). Tolerogenic DCs could be generated through HLA-G1 tetramers or HLA-G5 dimers via ILT4-mediated IL-6 signaling pathway and STAT3 activation. Functionally, HLA-G-induced tolerogenic DCs could induce the generation of CD4+CD25+CTLA-4+ (with CTLA being “cytotoxic T-lymphocyte-associated protein”) and CD8+CD28+ regulatory T cells (20,46).
A novel subset of tolerogenic DCs (DC-10), which secrete high levels of IL-10, and express high levels of HLA-G and its receptors ILT2, ILT4 and ILT3, has a potent specific function to induce IL-10-producing adaptive Tr1 through the ILT4/HLA-G signaling pathway. Data showed that the high expression levels of ILT4, HLA-G and IL-10 are necessary for the tolerogenic activity of DC-10 and their ability to prime T cells to become Tr1 cells (47). Recent data showed that HLA-G-expressing DC-10 and CD4+ T cells are highly represented in acute myeloid leukemia (AML) patients with HLA-G positive blasts (48).
MDSC are present in cancer patients and tumor-bearing mice and are, in part, responsible for the inhibition of the cell-mediated immune response against the tumor (49). It has been shown that infiltration of MDSCs in solid tumors is associated with poor prognosis as circulating MDSCs have a negative impact on survival and inversely correlate with the presence of functional antigen-specific T cells in patients with advanced melanoma (50,51). In ILT2-transgenic mice, HLA-G has been found to induce the emergence of CD11b+ Gr1+ MDSCs with an enhanced suppressive activity and is directly involved in the prolongation of allogeneic skin graft survival (52). In an immunocompetent HLA-G1+ M8 (a human melanoma cell line) tumor-bearing mouse model, interaction between HLA-G and the murine receptor PIR-B could expand the population of CD11b+Gr1+PIR-B+ MDSC, which could decrease NK cytotoxic activity (19). In another study, in a mouse model with murine mammary 4T1 cell line, HLA-G5 was observed to favor the CD11b+ Ly6G+ mice G-MDSC expansion in vivo (17).
Trogocytosis is a process of transferring cell-surface membrane proteins and membrane patches from one cell to another during contact. Relevance of trogocytosis is evidenced in tumor immunology such as HLA-G (53). HLA-G expressed by tumor cells can be acquired by activated cytotoxic NK cells and generates the HLA-G-positive NK cells, which behave as suppressor cells capable of protecting tumor cells from NK-mediated cytolysis (54). Similarly, resting or activated CD4+, CD8+ T and monocytes cells could acquire immunosuppressive HLA-G1 from tumor cells or APCs, which could immediately reverse their function from effectors to regulatory cells. Consequently, HLA-G-mediated immune evasion can be extended to HLA-G-negative tumor cells and further amplify HLA-G tolerogenic effect by trogocytosis (55). In multiple myeloma (MM) patients, HLA-G on malignant plasma cells was associated with a poor prognosis and HLA-G could be acquired by T cells from malignant plasma cells via trogocytosis (56).
Moreover, sHLA-G could impair expression and function of different chemokine receptors in T cells, NK cells and B cells through the receptor ILT2 (16). sHLA-G was observed to downregulate the expression of C-C motif chemokine receptor 2 (CCR2), C-X-C motif chemokine receptor 3 (CXCR3) and CXCR5 in CD4+ and follicular helper T cells, CXCR3 in CD8+ T cells, CXCR3 in Th1 clones, CXCR3 in T-cell receptor (TCR) Vδ2γ9 T cells, which was mediated by the interaction with ILT2. In addition, sHLA-G could inhibit chemotaxis of CD4+ T cells toward CCL2, CCL8, C-X-C motif chemokine ligand 10 (CXCL10) and CXCL11, CD8+ T cells toward CXCL10 and CXCL11, Th1 clones toward CXCL10, TCR Vδ2γ9 T cells toward CXCL10 and CXCL11 and follicular helper T cells toward CXCL13 (27). For NK cells, sHLA-G binding to ILT2 could also impair chemotaxis, cytokine and chemokine secretion in CD56bright and CD56dim NK cells in a dose-dependent manner. Morandi et al. (26) found that sHLA-G downmodulates expression of CXCR3, CX3CR1 and CCR2 and migration toward their specific ligands such as peripheral blood NK cells toward CXCL10, CXCL11 and CX3CL1 and CD56bright NK cells toward CCL2 and CXCL10. Also, HLA-G could down-regulate CXCR4 and CXCR5 expression on germinal center B cells (41).
Relevance of HLA-G Expression in Cancers
The expression of HLA-G in cancer was first demonstrated in the context of melanoma (57). Since then, HLA-G expression in more than 2,000 malignant samples among thirty types of tumors including both solid and hematological malignancies has been studied, where HLA-G expression was observed in different sources such as on the cell surface, secreted or in tumor-derived exosomes (14,58). Among these studies, there is a high frequency of tumor cell-surface HLA-G expression with an absence in healthy tissue, and increased sHLA-G levels has been detected in various body fluids in a variety of cancers (14). Expression of HLA-G was found to be correlated with clinical parameters such as more advanced disease stage, tumor metastasis and/or with a worse prognosis in tumor patients, indicating that HLA-G could facilitate tumor immune escape, invasiveness and metastasis; thereby HLA-G expression was found to be associated with advanced clinical stage and disease progression and HLA-G expression was also documented as an unfavorable prognostic factor for many kinds of solid malignancies, including breast cancer (59–64), colorectal cancer (65,66), cervical cancer (67,68), endometrial carcinoma (69,70), esophageal squamous cell carcinoma (71–74), gastric cancer (75–77), glioblastoma (78), hepatocellular carcinoma (79–81), lung cancer (82–84), nasopharyngeal carcinoma (85), oral cavity squamous cell carcinoma (86), ovarian cancer (87–89), pancreatic adenocarcinoma (90), thyroid carcinoma (91,92). These data were summarized in Table 1. Moreover, HLA-G expression was found to distinguish metastatic from nonmetastatic endometrial carcinoma (69). In our recent study, we found lesion HLA-G5/-G6 isoform expression could discriminate adenocarcinoma from squamous cell carcinoma in lung cancer patients, where tumor cell sHLA-G expression was predominately observed in adenocarcinoma lesions (84). These data indicated that lesion HLA-G or specific HLA-G isoform expression may serve as a clinical marker for tumor metastasis or types of histopathological discrimination.
Although HLA-G expression in solid tumors as mentioned above was associated with an unfavorable outcome or prognosis, no clear correlation was established between HLA-G expression and unfavorable clinical outcome in hematological malignancies, thus the clinical significance of HLA-G expression in liquid tumors remains controversial (93–99), as presented in Table 2.
The discrepancy in the clinical significance of HLA-G between solid and liquid tumors may be due to the nature of the tumor cells which, in malignant hematological diseases, are immune cells capable of expressing the HLA-G inhibitory receptor. In this context, studies by Naji et al. (41,100) revealed that, through interaction with ILT2, HLA-G could inhibit the proliferation, differentiation and antibody secretion of activated B cells originating from peripheral blood and secondary lymphoid organs, such as tonsils, and HLA-G could also inhibit the proliferation of ILT2-bearing neoplastic B cell lines such as human B cell lymphomas (Raji, Daudi and Ramos), multiple myelomas (OPM-2, LP1 and RPMI 8226), and B cell leukemia (BV173). These data imply that HLA-G could be correlated with a good prognosis by inhibiting the proliferation of ILT2-expressing tumor cells. Other factors may also account for these controversial results, including differences in expression and interaction of ILT2 and HLA-G by tumor cells, and effects of different chemotherapy status on HLA-G expression between the studies.
To be noted, regulation of HLA-G expression is controlled at the epigenetic, transcriptional and post-transcriptional level. The polymorphic variations in the 5’ upstream regulatory region (5’ URR) and 3’ untranslated regions (3’ UTR) in the HLA-G gene were reported to affect the HLA-G mRNA translation and HLA-G expression by microRNA binding and/or by modifying mRNA stability (101). In this context, a possible association of HLA-G gene polymorphism with susceptibility and/or clinical outcome of malignancies was documented. A 14-base pair insertion/deletion (ins/del) polymorphism (rs66554220) in the 3’ UTR was revealed; both chronic lymphocytic leukemia (CLL) and diffuse large B-cell lymphoma (DLBCL) patients with the del/del genotype had shorter survival than patients with the ins/del or ins/ins genotype (102,103). Additionally, DLBCL patients who carry the HLA-G-725CC genotype in the 5’ URR had a significantly higher probability of overall survival than individuals with other genotype combinations of HLA-G-725C/G/T (103). Furthermore, in the coding segment of the HLA-G gene, the codon 130C deletion occurrence was observed to be significantly associated with a decreased free disease and overall survival in nasopharyngeal carcinoma patients (104). However, more studies on the complete HLA-G gene and its linkage disequilibrium with other genes and on HLA-G expression at both the tumor site and plasma in a specific microenvironment are required to consolidate these findings.
Advances of Tumor-Bearing Animal Models for HLA-G Study
Recently, the development of animal models consolidated the concept that HLA-G+ tumor cells can develop and tolerize the host antitumor immune response in vivo. Although there is no murine homologue of HLA-G, in vivo studies were made possible by the fact that human HLA-G can bind and mediate a signal via the murine receptor-paired immunoglobulin-like receptor (PIR)-B, the homologue of human ILTs (105). Previous data showed that human or murine tumor cells expressing HLA-G can grow in immunocompetent host mice and that blocking HLA-G function by a specific antibody inhibits the development of the tumor, where HLA-G could impair both innate and adaptive immunity resulting in an expansion of CD11b+ Gr1+ myeloid-derived suppressor cells, loss of peripheral T cells and a cytokine balance in favor of a Th2 profile versus Th1/Th17 (17,19).
Other than the tolerant functions of HLA-G in tumor immune escaping, HLA-G was also found in association with the status of tumor invasion and metastasis in patients and preclinical models with the mechanism of upregulating the expression of MMPs (18,106) (Figure 2). MMPs are one of the central mediators of the biology of tumor invasion and metastasis with their ability to degrade basement membrane and extracellular matrix components (107). Indeed, the relationship between HLA-G and MMP expression was addressed in previous studies. Hiden et al. (108) addressed that MMP-15 expression could be induced by TNF-α only in the HLA-G positive first trimester human trophoblast cell line ACH-3P, and only the HLA-G positive extravillous trophoblasts (EVT) have the invasive potential. Much higher MMP-14 expression was also found in HLA-G positive first-trimester invasive trophoblasts (109). Both tumor cells and EVT share similar characteristics in many regards, such as the capability of proliferation, invasion, vasculogenic mimicry, angiogenesis and systemic immune modulation (110). Therefore, similar tactics may be involved in invasion and metastasis of tumor cells.
Using a Balb/c nu/nu model, we reported that with exogenous HLA-G expression the ovarian cancer cell HO-8910 has a higher invasion and metastasis potential and the host is much less likely to survive, suggesting for the first time that HLA-G expression is associated with tumor metastasis and with poor survival in an animal model in vivo (18). Characterization of the mechanisms by which HLA-G is involved in tumor cell invasion and metastasis shows that upregulated MMP-15 expression was in accordance with HLA-G expression and a strong correlation was also observed between HLA-G and MMP-15 expression in a cohort of ovarian cancer lesions. Furthermore, knockdown of the HLA-G-induced MMP-15 expression significantly decreased cell migration and tumor metastasis (18). In accordance with our findings, HLA-G5 was documented to stimulate primary trophoblasts and trophoblastic cell line (JAR and JEG-3) invasion and to increase MMP-2 transcripts and their activity by binding to KIR2DL4 and ILT2 (22). In another study, suppression of HLA-G expression in JEG-3 cells was observed to decrease the MMP-2 and MMP-9 mRNA and protein expression dramatically (21).
Implications for HLA-G-Based Strategies in Cancer Immunotherapy
Since HLA-G is frequently and specifically expressed on tumor cells, and its expression is associated with unfavorable prognosis in various malignancies, HLA-G is an attractive target for cancer therapies (15,111).
Taking the journey of HLA-G-expressing malignant cells and targeting HLA-G-expressing cancer cells with a delivery system based on nanoparticles carrying anticancer drugs to the tumor site would be also important in maximizing the efficacy of anticancer therapies. In this context, Zhang et al. (112) successfully developed mAb HLA-G and methotrexate (MTX)-loaded nanobubbles (mAbHLA-G/MTX/PLGA NBs [with PLGA being “poly(lactic-co-glycolic acid)”]) as an HLA-G-targeted drug carrier. Data showed that the nanobubbles could efficiently target HLA-G-positive tumor tissues both in vitro and in vivo, and that the released MTX from mAbHLA-G/MTX/ PLGA NBs could remove the residual cancer cells and inhibit the reoccurrence of tumors. Moreover, given the immune inhibitory properties of the HLA-G molecule, it would be important to suppress its expression in tumor cells. Previous studies showed that suppression or downregulation of HLA-G by interfering RNA was found to increase NK cell cytolysis against targets, that antibodies blocking HLA-G or its receptors ILT2 and/or ILT4 could restore the functions of T cells and NK cells, and that the HLA-G-derived peptide HLA-G146-154 could effectively induce peptide-specific CTLs cytotoxic activity against HLA-G-expressing HLA-A24+ renal cell carcinoma cells (15,113,114).
Conclusion
On the basis of the above observations, an HLA-G-based immunotherapy can be achieved by employing HLA-G or HLA-G receptor antibodies to block HLA-G expression or interaction between its receptors, by RNA interfering to down-regulate the HLA-G expression levels, or by using HLA-G-derived immunogenic peptide and anti-idiotype antibodies to stimulate immune cell function. Thus, HLA-G-targeted strategies in malignancies could be attractive in the future for the development of nanomedicine and noninvasive medicine for simultaneously targeted imaging, drug delivery and therapy in cancers (Figure 3). Nevertheless, in hematological tumors, HLA-G could inhibit the proliferation of B-cell malignancies, and, therefore, exogenously produced HLA-G or HLA-G-derived polypeptides can be considered as therapeutic agents for leukemia therapy.
Disclosure
The authors declare that they have no competing interests as defined by Molecular Medicine, or other interests that might be perceived to influence the results and discussion reported in this paper.
References
Bruttel VS, Wischhusen J. (2014) Cancer stem cell immunology: key to understanding tumorigenesis and tumor immune escape? Front Immunol. 5:360.
Gajewski TF, Schreiber H, Fu YX. (2013) Innate and adaptive immune cells in the tumor microenvironment. Nat. Immunol. 14:1014–22.
Curigliano G, Criscitiello C, Gelao L, Goldhirsch A. (2013) Molecular pathways: human leukocyte antigen G (HLA-G). Clin. Cancer Res. 19:5564–71.
Kochan G, Escors D, Breckpot K, Guerrero-Setas D. (2013) Role of non-classical MHC class I molecules in cancer immunosuppression. Oncoimmunology. 2:e26491.
González A, Rebmann V, LeMaoult J, Horn PA, Carosella ED, Alegre E. (2012) The immunosuppressive molecule HLA-G and its clinical implications. Crit. Rev. Clin. Lab. Sci. 49:63–84.
Rizzo R, et al. (2013) Matrix metalloproteinase-2 (MMP-2) generates soluble HLA-G1 by cell surface proteolytic shedding. Mol. Cell. Biochem. 381:243–55.
Kovats S, Main EK, Librach C, Stubblebine M, Fisher SJ, DeMars R. (1990) A class I antigen, HLA-G, expressed in human trophoblasts. Science. 248:220–3.
Menier C, Rabreau M, Challier JC, Le Discorde M, Carosella ED, Rouas-Freiss N. (2004) Erythroblasts secrete the nonclassical HLA-G molecule from primitive to definitive hematopoiesis. Blood. 104:3153–60.
Le Discorde M, Moreau P, Sabatier P, Legeais JM, Carosella ED. (2003) Expression of HLA-G in human cornea, an immune-privileged tissue. Hum. Immunol. 64:1039–44.
Crisa L, McMaster MT, Ishii JK, Fisher SJ, Salomon DR. (1997) Identification of a thymic epithelial cell subset sharing expression of the class Ib HLA-G molecule with fetal trophoblasts. J. Exp. Med. 186:289–98.
Cirulli V, et al. (2006) The class I HLA repertoire of pancreatic islets comprises the nonclassical class Ib antigen HLA-G. Diabetes. 55:1214–22.
Deschaseaux F, Delgado D, Pistoia V, Giuliani M, Morandi F, Durrbach A. (2011) HLA-G in organ transplantation: towards clinical applications. Cell. Mol. Life Sci. 68:397–404.
Rizzo R, Bortolotti D, Baricordi OR, Fainardi E. (2012) New insights into HLA-G and inflammatory diseases. Inflamm. Allergy Drug Targets. 11:448–63.
Yan WH. (2011) Human leukocyte antigen-G in cancer: are they clinically relevant Cancer Lett. 311:123–30.
Yan WH. (2011) HLA-G expression in cancers: potential role in diagnosis, prognosis and therapy. Endocr. Metab. Immune Disord. Drug Targets. 11:76–89.
Morandi F, Rouas-Freiss N, Pistoia V. (2014) The emerging role of soluble HLA-G in the control of chemotaxis. Cytokine Growth Factor Rev. 25:327–35.
Loumagne L, Baudhuin J, Favier B, Montespan F, Carosella ED, Rouas-Freiss N. (2014) In vivo evidence that secretion of HLA-G by immunogenic tumor cells allows their evasion from immunosurveillance. Int. J. Cancer. 135:2107–17.
Lin A, Xu HH, Xu DP, Zhang X, Wang Q, Yan WH. (2013) Multiple steps of HLA-G in ovarian carcinoma metastasis: alter NK cytotoxicity and induce matrix metalloproteinase-15 (MMP-15) expression. Hum. Immunol. 74:439–46.
Agaugué S, Carosella ED, Rouas-Freiss N. (2011) Role of HLA-G in tumor escape through expansion of myeloid-derived suppressor cells and cytokinic balance in favor of Th2 versus Th1/Th17. Blood. 117:7021–31.
Fainardi E, et al. (2011) Emerging topics and new perspectives on HLA-G. Cell. Mol. Life Sci. 68:433–51.
Liu H, Liu X, Jin H, Yang F, Gu W, Li X. (2013) Proteomic analysis of knock-down HLA-G in invasion of human trophoblast cell line JEG-3. Int. J. Clin. Exp. Pathol. 6:2451–9.
Guo Y, et al. (2013) Soluble human leukocyte antigen-g5 activates extracellular signal-regulated protein kinase signaling and stimulates trophoblast invasion. PLoS One. 8:e76023.
Amodio G, Gregori S. (2012) Human tolerogenic DC-10: perspectives for clinical applications. Transplant Res. 1:14.
Carosella ED, Gregori S, LeMaoult J. (2011) The tolerogenic interplay(s) among HLA-G, myeloid APCs, and regulatory cells. Blood. 118:6499–505.
Amodio G, et al. (2015) HLA-G expression levels influence the tolerogenic activity of human DC-10. Haematologica. 100:548–57.
Morandi F, et al. (2011) Soluble HLA-G dampens CD94/NKG2A expression and function and differentially modulates chemotaxis and cytokine and chemokine secretion in CD56bright and CD56dim NK cells. Blood. 118:5840–50.
Morandi F, Ferretti E, Bocca P, Prigione I, Raffaghello L, Pistoia V. (2010) A novel mechanism of soluble HLA-G mediated immune modulation: downregulation of T cell chemokine receptor expression and impairment of chemotaxis. PLoS One. 5:e11763.
Le Gal FA, et al. (1999) HLA-G-mediated inhibition of antigen-specific cytotoxic T lymphocytes. Int. Immunol. 11:1351–6.
Lin A, et al. (2007) HLA-G expression in human ovarian carcinoma counteracts NK cell function. Ann. Oncol. 18:1804–9.
Bainbridge DR, Ellis SA, Sargent IL. (2000) HLA-G suppresses proliferation of CD4(+) T-lymphocytes. J. Reprod. Immunol. 48:17–26.
Contini P, et al. (2003) Soluble HLA-A,-B,-C and -G molecules induce apoptosis in T and NK CD8+ cells and inhibit cytotoxic T cell activity through CD8 ligation. Eur. J. Immunol. 33:125–34.
Horuzsko A, Lenfant F, Munn DH, Mellor AL. (2001) Maturation of antigen-presenting cells is compromised in HLA-G transgenic mice. Int. Immunol. 13:385–94.
Liang S, Ristich V, Arase H, Dausset J, Carosella ED, Horuzsko A. (2008) Modulation of dendritic cell differentiation by HLA-G and ILT4 requires the IL-6—STAT3 signaling pathway. Proc. Natl. Acad. Sci. U.S.A. 105:8357–8362.
Gros F, Cabillic F, Toutirais O, Maux AL, Sebti Y, Amiot L. (2008) Soluble HLA-G molecules impair natural killer/dendritic cell crosstalk via inhibition of dendritic cells. Eur. J. Immunol. 38:742–9.
Lesport E, et al. (2011) Inhibition of human Vγ9Vδ2 T-cell antitumoral activity through HLA-G: implications for immunotherapy of cancer. Cell. Mol. Life Sci. 68:3385–99.
Chen BG, Xu DP, Lin A, Yan WH. (2013) NK cytolysis is dependent on the proportion of HLA-G expression. Hum. Immunol. 74:286–9.
Zhang WQ, et al. (2014) HLA-G1 and HLA-G5 isoforms have an additive effect on NK cytolysis. Hum. Immunol. 75:182–9.
Wiendl H, et al. (2002) A functional role of HLA-G expression in human gliomas: an alternative strategy of immune escape. J. Immunol. 168:4772–80.
Riteau B, et al. (2001) HLA-G1 co-expression boosts the HLA class I-mediated NK lysis inhibition. Int. Immunol. 13:193–201.
Bukur J, et al. (2003) Functional role of human leukocyte antigen-G up-regulation in renal cell carcinoma. Cancer Res. 63:4107–11.
Naji A, et al. (2014) Binding of HLA-G to ITIM-bearing Ig-like transcript 2 receptor suppresses B cell responses. J. Immunol. 192:1536–46.
Baudhuin J, et al. (2013) Exocytosis acts as a modulator of the ILT4-mediated inhibition of neutrophil functions. Proc. Natl. Acad. Sci. U.S.A. 110:17957–62.
Carosella ED, Favier B, Rouas-Freiss N, Moreau P, Lemaoult J. (2008) Beyond the increasing complexity of the immunomodulatory HLA-G molecule. Blood. 111:4862–70.
Carosella ED, HoWangYin KY, Favier B, LeMaoult J. (2008) HLA-G-dependent suppressor cells: Diverse by nature, function, and significance. Hum. Immunol. 69:700–7.
LeMaoult J, Krawice-Radanne I, Dausset J, Carosella ED. (2004) HLA-G1-expressing antigen-presenting cells induce immunosuppressive CD4+ T cells. Proc. Natl. Acad. Sci. U.S.A. 101:7064–9.
Ristich V, Liang S, Zhang W, Wu J, Horuzsko A. (2005) Tolerization of dendritic cells by HLA-G. Eur. J. Immunol. 35:1133–42.
Gregori S, et al. (2010) Differentiation of type 1 T regulatory cells (Tr1) by tolerogenic DC-10 requires the IL-10-dependent ILT4/HLA-G pathway. Blood. 116:935–44.
Locafaro G, Amodio G, Tomasoni D, Tresoldi C, Ciceri F, Gregori S. (2014) HLA-G expression on blasts and tolerogenic cells in patients affected by acute myeloid leukemia. J. Immunol. Res. 2014:636292.
Serafini P. (2013) Myeloid derived suppressor cells in physiological and pathological conditions: the good, the bad, and the ugly. Immunol. Res. 57:172–84.
Khaled YS, Ammori BJ, Elkord E. (2013) Myeloid-derived suppressor cells in cancer: recent progress and prospects. Immunol. Cell Biol. 91:493–502.
Weide B, et al. (2014) Myeloid-derived suppressor cells predict survival of patients with advanced melanoma: comparison with regulatory T cells and NY-ESO-1- or melan-A-specific T cells. Clin. Cancer Res. 20:1601–9.
Zhang W, Liang S, Wu J, Horuzsko A. (2008) Human inhibitory receptor immunoglobulin-like transcript 2 amplifies CD11b+Gr1+ myeloid-derived suppressor cells that promote long-term survival of allografts. Transplantation. 86:1125–34.
Carosella ED, Gregori S, Rouas-Freiss N, LeMaoult J, Menier C, Favier B. (2011) The role of HLA-G in immunity and hematopoiesis. Cell. Mol. Life Sci. 68:353–68.
Caumartin J, et al. (2007) Trogocytosis-based generation of suppressive NK cells. EMBO J. 26:1423–33.
HoWangYin KY, Alegre E, Daouya M, Favier B, Carosella ED, LeMaoult J. (2010) Different functional outcomes of intercellular membrane transfers to monocytes and T cells. Cell. Mol. Life Sci. 67:1133–45.
Brown R, et al. (2012) CD86+ or HLA-G+ can be transferred via trogocytosis from myeloma cells to T cells and are associated with poor prognosis. Blood. 120:2055–63.
Paul P, et al. (1998) HLA-G expression in melanoma: a way for tumor cells to escape from immunosurveillance. Proc. Natl. Acad. Sci. U.S.A. 95:4510–5.
Riteau B, et al. (2003) Exosomes bearing HLA-G are released by melanoma cells. Hum. Immunol. 64:1064–72.
Kleinberg L, et al. (2006) Expression of HLA-G in malignant mesothelioma and clinically aggressive breast carcinoma. Virchows Arch. 449:31–9.
Chen HX, et al. (2010) Upregulation of human leukocyte antigen-G expression and its clinical significance in ductal breast cancer. Hum. Immunol. 71:892–8.
He X, et al. (2010) HLA-G expression in human breast cancer: implications for diagnosis and prognosis, and effect on allocytotoxic lymphocyte response after hormone treatment in vitro. Ann. Surg. Oncol. 17:1459–69.
de Kruijf EM, et al. (2010) HLA-E and HLA-G expression in classical HLA class I-negative tumors is of prognostic value for clinical outcome of early breast cancer patients. J. Immunol. 185:7452–9.
da Silva GB, et al. (2013) Expression of the classical and nonclassical HLA molecules in breast cancer. Int. J. Breast Cancer. 2013:250435.
Ramos CS, et al. (2014) Analysis of HLA-G gene polymorphism and protein expression in invasive breast ductal carcinoma. Hum. Immunol. 75:667–672.
Ye SR, Yang H, Li K, Dong DD, Lin XM, Yie SM. (2007) Human leukocyte antigen G expression: as a significant prognostic indicator for patients with colorectal cancer. Mod. Pathol. 20:375–83.
Guo ZY, et al. (2015) Predictive value of HLA-G and HLA-E in the prognosis of colorectal cancer patients. Cell. Immunol. 293:10–6.
Miranda LN, et al. (2015) Greater expression of the human leukocyte antigen-G (HLA-G) and interleukin-17 (IL-17) in cervical intraepithelial neoplasia: analytical cross-sectional study. Sao Paulo Med. J. 133:336–42.
Li XJ, Zhang X, Lin A, Ruan YY, Yan WH. (2012) Human leukocyte antigen-G (HLA-G) expression in cervical cancer lesions is associated with disease progression. Hum. Immunol. 73:946–9.
Barrier BF, Kendall BS, Sharpe-Timms KL, Kost ER. (2006) Characterization of human leukocyte antigen-G (HLA-G) expression in endometrial adenocarcinoma. Gynecol. Oncol. 103:25–30.
Bijen CB, et al. (2010) The prognostic role of classical and nonclassical MHC class I expression in endometrial cancer. Int. J. Cancer. 126:1417–27.
Yie SM, Yang H, Ye SR, Li K, Dong DD, Lin XM. (2007) Expression of HLA-G is associated with prognosis in esophageal squamous cell carcinoma. Am. J. Clin. Pathol. 128:1002–9.
Lin A, et al. (2011) Human leukocyte antigen-G expression is associated with a poor prognosis in patients with esophageal squamous cell carcinoma. Int. J. Cancer. 129:1382–90.
Zheng J, et al. (2014) Human leukocyte antigen G is associated with esophageal squamous cell carcinoma progression and poor prognosis. Immunol. Lett. 161:13–9.
Hu J, et al. (2013) Overexpression of HLA-G Is positively associated with Kazakh esophageal squamous cell carcinoma in Xinjiang, China. Viral Immunol. 26:180–4.
Yie SM, Yang H, Ye SR, Li K, Dong DD, Lin XM. (2007) Expression of human leukocyte antigen G (HLA-G) correlates with poor prognosis in gastric carcinoma. Ann. Surg. Oncol. 14:2721–9.
Tuncel T, et al. (2013) Immunoregulatory function of HLA-G in gastric cancer. Asian Pac. J. Cancer Prev. 14:7681–4.
Du L, et al. (2011) Human leukocyte antigen-G is closely associated with tumor immune escape in gastric cancer by increasing local regulatory T cells. Cancer Sci. 102:1272–80.
Wastowski IJ, et al. (2013) Human leukocyte antigen-G is frequently expressed in glioblastoma and may be induced in vitro by combined 5-aza-2’-deoxycytidine and interferon-γ treatments: results from a multicentric study. Am. J. Pathol. 182:540–52.
Cai MY, et al. (2009) Human leukocyte antigen-G protein expression is an unfavorable prognostic predictor of hepatocellular carcinoma following curative resection. Clin. Cancer Res. 15:4686–93.
Wang Y, Ye Z, Meng XQ, Zheng SS. (2011) Expression of HLA-G in patients with hepatocellular carcinoma. Hepatobiliary Pancreat. Dis. Int. 10:158–63.
Lin A, et al. (2010) Aberrant human leucocyte antigen-G expression and its clinical relevance in hepatocellular carcinoma. J. Cell Mol. Med. 14:2162–71.
Yie SM, Yang H, Ye SR, Li K, Dong DD, Lin XM. (2007) Expression of human leucocyte antigen G (HLA-G) is associated with prognosis in non-small cell lung cancer. Lung Cancer. 58:267–74.
Lin A, et al. (2010) Clinical relevance and functional implications for human leucocyte antigen-g expression in non-small-cell lung cancer. J. Cell Mol. Med. 14:2318–29.
Yan WH, Liu D, Lu HY, Li YY, Zhang X, Lin A. (2015) Significance of tumour cell HLA-G5/-G6 isoform expression in discrimination for adenocarcinoma from squamous cell carcinoma in lung cancer patients. J. Cell Mol. Med. 19:778–85.
Cai MB, et al. (2012) Expression of human leukocyte antigen G is associated with prognosis in nasopharyngeal carcinoma. Int. J. Biol. Sci. 8:891–900.
Gonçalves AS, et al. (2014) The clinicopathologic significance of the expression of HLA-G in oral squamous cell carcinoma. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 117:361–8.
Jung YW, et al. (2009) Correlation of human leukocyte antigen-G (HLA-G) expression and disease progression in epithelial ovarian cancer. Reprod. Sci. 16:1103–11.
Rutten MJ, et al. (2014) HLA-G expression is an independent predictor for improved survival in high grade ovarian carcinomas. J. Immunol. Res. 2014:274584.
Menier C, Prevot S, Carosella ED, Rouas-Freiss N. (2009) Human leukocyte antigen-G is expressed in advanced-stage ovarian carcinoma of high-grade histology. Hum. Immunol. 70:1006–9.
Xu YF, et al. (2015) High expression of human leukocyte antigen-G is associated with a poor prognosis in patients with PDAC. Curr. Mol. Med. 15:360–7.
de Figueiredo Feitosa NL, et al. (2014) HLA-G is differentially expressed in thyroid tissues. Thyroid. 24:585–92.
Nunes LM, et al. (2013) Association between the HLA-G molecule and lymph node metastasis in papillary thyroid cancer. Hum. Immunol. 74:447–51.
Alkhouly N, Shehata I, Ahmed MB, Shehata H, Hassan S, Ibrahim T. (2013) HLA-G expression in acute lymphoblastic leukemia: a significant prognostic tumor biomarker. Med. Oncol. 30:460.
Guo QY, Chen BG, Ruan YY, Lin A, Yan WH. (2011) HLA-G expression is irrelevant to prognosis in patients with acute myeloid leukemia. Leuk. Res. 35:1350–4.
Attia MA, Nosair NA, Gawally A, Elnagar G, Elshafey EM. (2014) HLA-G expression as a prognostic indicator in B-cell chronic lymphocytic leukemia. Acta Haematol. 132:53–8.
Nückel H, Rebmann V, Dürig J, Dührsen U, Grosse-Wilde H. (2005) HLA-G expression is associated with an unfavorable outcome and immunodeficiency in chronic lymphocytic leukemia. Blood. 105:1694–8.
Erikci AA, Karagoz B, Ozyurt M, Ozturk A, Kilic S, Bilgi O. (2009) HLA-G expression in B chronic lymphocytic leukemia: a new prognostic marker? Hematology. 14:101–5.
Perez-Chacon G, et al. (2009) Prognostic irrelevance of HLA-G in B-cell chronic lymphocytic leukemia. Int. J. Lab. Hematol. 31:327–37.
Giannopoulos K, Dmoszyńska A, Bojarska-Junak A, Schmitt M, Rolifński J. (2008) Expression of HLA-G in patients with B-cell chronic lymphocytic leukemia (B-CLL). Folia Histochem. Cytobiol. 46:457–60.
Naji A, Menier C, Maki G, Carosella ED, Rouas-Freiss N. (2012) Neoplastic B-cell growth is impaired by HLA-G/ILT2 interaction. Leukemia. 26:1889–92.
Donadi EA, Castelli EC, Arnaiz-Villena A, Roger M, Rey D, Moreau P. (2011) Implications of the polymorphism of HLA-G on its function, regulation, evolution and disease association. Cell. Mol. Life Sci. 68:369–95.
Rizzo R, et al. (2014) HLA-G is a component of the chronic lymphocytic leukemia escape repertoire to generate immune suppression: impact of the HLA-G 14 base pair (rs66554220) polymorphism. Haematologica. 99:888–96.
Bielska M, et al. (2015) Human leukocyte antigen-G polymorphisms influence the clinical outcome in diffuse large B-cell lymphoma. Genes Chromosomes Cancer. 54:185–93.
Ghandri N, et al. (2011) Association of HLA-G polymorphisms with nasopharyngeal carcinoma risk and clinical outcome. Hum. Immunol. 72:150–8.
Liang S, Baibakov B, Horuzsko A. (2002) HLA-G inhibits the functions of murine dendritic cells via the PIR-B immune inhibitory receptor. Eur. J. Immunol. 32:2418–26.
Ueshima C, et al. (2015) The killer cell Ig-like receptor 2DL4 expression in human mast cells and its potential role in breast cancer invasion. Cancer Immunol. Res. 3:871–80.
Hadler-Olsen E, Winberg JO, Uhlin-Hansen L. (2013) Matrix metalloproteinases in cancer: their value as diagnostic and prognostic markers and therapeutic targets. Tumour Biol. 34:2041–51.
Hiden U, et al. (2007) The first trimester human trophoblast cell line ACH-3P: a novel tool to study autocrine/paracrine regulatory loops of human trophoblast subpopulations—TNF-alpha stimulates MMP15 expression. BMC Dev. Biol. 7:137.
Hiden U, et al. (2008) MT1-MMP expression in first-trimester placental tissue is upregulated in type 1 diabetes as a result of elevated insulin and tumor necrosis factor-alpha levels. Diabetes. 57:150–7.
Holtan SG, Creedon DJ, Haluska P, Markovic SN. (2009) Cancer and pregnancy: parallels in growth, invasion, and immune modulation and implications for cancer therapeutic agents. Mayo Clin. Proc. 84:985–1000.
Amiot L, Ferrone S, Grosse-Wilde H, Seliger B. (2011) Biology of HLA-G in cancer: a candidate molecule for therapeutic intervention? Cell. Mol. Life Sci. 68:417–31.
Zhang X, et al. (2014) Methotrexate-loaded PLGA nanobubbles for ultrasound imaging and synergistic targeted therapy of residual tumor during HIFU ablation. Biomaterials. 35:5148–61.
Komohara Y, et al. (2007) HLA-G as a target molecule in specific immunotherapy against renal cell carcinoma. Oncol. Rep. 18:1463–8.
Jasinski-Bergner S, et al. (2015) Clinical relevance of miR-mediated HLA-G regulation and the associated immune cell infiltration in renal cell carcinoma. Oncoimmunology. 4:e1008805.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (31170879, 31370920, 81372247), the Natural Science Foundation of Zhejiang Province (LR13H160001) and the Zhejiang Provincial program for the cultivation of high-level innovative health talents.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, and provide a link to the Creative Commons license. You do not have permission under this license to share adapted material derived from this article or parts of it.
The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this license, visit (https://doi.org/creativecommons.org/licenses/by-nc-nd/4.0/)
About this article
Cite this article
Lin, A., Yan, WH. Human Leukocyte Antigen-G (HLA-G) Expression in Cancers: Roles in Immune Evasion, Metastasis and Target for Therapy. Mol Med 21, 782–791 (2015). https://doi.org/10.2119/molmed.2015.00083
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2119/molmed.2015.00083