Abstract
There are 22 species of mesocarnivores (carnivores weighing < 15 kg) belonging to five families that live in rangelands of the western United States. Mesocarnivores are understudied relative to large carnivores but can have significant impacts on ecosystems and human dimensions. In this chapter, we review the current state of knowledge about the biology, ecology, and human interactions of the mesocarnivores that occupy the rangelands of the central and western United States. In these two regions, mesocarnivores may serve as the apex predator in areas where large carnivores no longer occur, and can have profound impacts on endemic prey, disease ecology, and livestock production. Some mesocarnivore species are valued because they are harvested for food and fur, while others are considered nuisance species because they can have negative impacts on ranching. Many mesocarnivores have flexible life history strategies that make them well-suited for future population growth or range expansion as western landscapes change due to rapid human population growth, landscape development, and alterations to ecosystems from climate change; however other mesocarnivores continue to decline. More research on this important guild is needed to understand their role in western working landscapes.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Canids
- Felids
- Fur harvest
- Intraguild interactions
- Mesopredator
- Mustelids
- Procyonids
- Rangeland management
- Skunks
1 General Natural History of Mesocarnivores
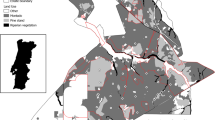
Western rangelands host 22 mesocarnivore species, belonging to five families (Fig. 16.1). Mesocarnivores are defined as mid-sized carnivores, weighing < 15 kg; they are typically more abundant than large carnivores and carry important ecological roles that may regulate trophic levels similar to their larger counterparts (Roemer et al. 2009). The high number of mesocarnivores is reflected by their diverse adaptations to their environment, which results in a diversity of dietary portfolios, and substantial variation in spatiotemporal patterns of habitat use. The diversity among mesocarnivores enables them to effectively partition resources (e.g., via spatial, temporal, or dietary partitioning) and coexist within similar habitats throughout their range (Roemer et al. 2009; Lesmeister et al. 2015).
Range of mesocarnivore species inhabiting rangelands of western North America and adjoining regions. Canidae: Coyote (Kays 2018); gray fox (Roemer et al. 2016); red fox (Moehrenschlager and Sovada 2016); kit fox (Cypher and List 2014); swift fox (Moehrenschlager and Sovada 2016). Mephitidae: Eastern spotted skunk (Gompper and Jachowski 2016); western spotted skunk (Cuarón et al. 2016c); hog-nosed skunk (Helgen 2016); hooded skunk (Cuarón et al. 2016a); striped skunk (Helgen and Reid 2016a). Mustelidae: mink (Reid et al. 2016b); American badger (Helgen and Reid 2016b); river otter (Serfass et al. 2015); black-footed ferret (Belant et al. 2015); least weasel (McDonald et al. 2019); short-tailed weasel (Reid et al. 2016a); long-tailed weasel (Helgen and Reid 2016c). Felidae: Bobcat (Kelly et al. 2016); ocelot (Paviolo et al. 2015). Procyonidae: Raccoon (Timm et al. 2016); ringtail (Reid et al. 2016c); white-nosed coati (Cuarón et al. 2016b). Rangelands landcover (Olson et al. 2001)
1.1 Species and Population Statuses
Although there are many mesocarnivores in western landscapes, most are understudied, and information is therefore limited on their distribution and population status. In general, many are declining, notably up to 62% of the world’s small carnivore species (Belant et al. 2009), and there is a need for improving conservation efforts that reduce population threats (Marneweck et al. 2021). Contrasting this plight, some populations are expanding and are considered to be nuisance species. We present this information collectively by their scientific Family to highlight similarities within groups and where information remains sparse.
1.2 Family: Canidae
Five medium-sized canids occur in western rangelands; coyotes (Canis latrans) and four fox species (gray fox [Urocyon cinereoargenteus], kit fox [Vulpes macrotis], red fox [Vulpes vulpes], and swift fox [Vulpes velox]). The most widespread and largest (7–20 kg) of the five is the coyote (Kays 2018). Coyotes historically occupied a considerable portion of the western third of North America, excluding parts of northern Canada and Alaska (Hody and Kays 2018). They mostly occurred in open habitats including grasslands and shrublands. During the last century, the exploratory tendencies and generalist diet of coyotes, combined with human modifications that opened forested habitats and the historical elimination of gray wolves (Canis lupus) and cougars (Puma concolor) from much of their respective ranges, have allowed coyotes to expand their range by over 40% (Gompper 2002; Hody and Kays 2018). Coyote populations are rarely assessed even though some states have harvest seasons or bounty programs, but they are generally considered stable or increasing throughout their range.
The red fox is also widespread throughout North America; however, some populations are a mix of native and non-native individuals (e.g., Black et al. 2018). Native red foxes originated in boreal and mountainous habitats in northern North America (Kamler and Ballard 2002; Aubry et al. 2009). Non-native red foxes were likely transported to the United States from Europe in the 1700s (Kasprowicz et al. 2016); however, it is likely interbreeding was limited to the eastern parts of the United States and is not of concern for western populations (Statham et al. 2012; Sacks et al. 2018; Kuo et al. 2019). Their omnivorous diet and ability to thrive near human habitation enabled red foxes to occupy much of North America (Kamler and Ballard 2002; Hoffmann and Sillero-Zubiri 2016). Red foxes occur in higher densities near agriculture, towns, or dry and patchy landscapes with shrubs and woodlands, and occur at lower densities in grasslands (Hoffmann and Sillero-Zubiri 2016). Red fox populations are rarely assessed but there are current attempts related to threatened and endangered subpopulations, such as for the Sierra Nevada red fox (V. v. necator; e.g., Hatfield et al. 2021).
The gray fox overlaps with the red fox throughout most of its range and there is evidence that the two species can coexist with little competition, likely due to the ability of gray foxes to climb (Lesmeister et al. 2015). The range of the gray fox extends farther southwest, while the range of the red fox extends farther north. Similar to coyotes and red foxes, the diet of gray foxes is omnivorous and consists mostly of small mammals, fruits, and seeds (Larson et al. 2015). They occur in woodland, riparian forests, and dense shrublands but also at agricultural and urban edges (Roemer et al. 2016). Gray fox populations are rarely assessed but considered stable throughout their range.
The kit fox is found in arid deserts and grasslands of southwest North America, whereas the historical range of the swift fox included the Great Plains grasslands and shrublands. Kit foxes and swift foxes only overlap in the southern most extent of their ranges, in parts of Texas and New Mexico. They were considered conspecific until 2005, when they were separated into two species following genetic assessments (Cypher and List 2014; Moehrenschlager and Sovada 2016). They are similar in size (1.3–3.5 kg; swift foxes are slightly smaller) and have similar biology. The diet of both species is comprised by a high proportion of rodents and insects (Hines and Case 1991; Pechacek et al. 2000). Kit foxes are considered stable throughout the southern portions of their range, and declining in the northern third of their range (Lonsinger et al. 2020). Swift foxes were eliminated from ~ 90% of their range by the 1950s, and today occupy about 40% of their historical range, with lower densities in the northern portion of their range (Zimmerman 1998; Sovada et al. 2009). Intraguild predation by coyotes on swift foxes and kit foxes has been documented in many areas throughout the foxes’ range and is linked to local extinctions (Nelson et al. 2007; Thompson and Gese 2007; Karki et al. 2007; Lonsinger et al. 2017).
1.3 Family: Felidae
There are three felid mesocarnivores found in North America’s rangelands: bobcat (Lynx rufus), ocelot (Leopardus pardalis), and jaguarundi (Herpailurus yagouaroundi). Only bobcats and ocelots are found within the United States today. Bobcats are common and widespread across North America, whereas, the ocelot’s range is primarily through Central and South America (Paviolo et al. 2015; Kelly et al. 2016). Both species can withstand certain degrees of anthropogenic disturbance, albeit ocelots are significantly less tolerant of human disturbances. In the United States, ocelots are considered endangered (Paviolo et al. 2015); they require unfragmented thornscrub habitat, which has limited availability (Jackson et al. 2005; Horne et al. 2009; Janečka et al. 2016). The majority of ocelots in the United States are currently on private rangelands in Texas (Lombardi et al. 2020b) and experience high mortality from road collisions (Haines et al. 2005; Blackburn et al. 2021). Bobcat populations are stable or increasing throughout most of their range (Roberts and Crimmins 2010). Even so, they are closely regulated due to inclusion in Appendix II of the Convention on International Trade in Endangered Species of Wild Flora and Fauna (CITES) as the only spotted cat legally traded worldwide. Local bobcat densities are dependent on prey availability (CITES 2021). Bobcats are generalists and adapt to changes in prey composition (Newbury and Hodges 2018). In southern rangelands, ocelot and bobcat diets overlap, consisting of small rodents, lagomorphs, and birds (Booth-Binczik et al. 2013). Overlap in diet and competition with bobcats may affect the ocelot’s recovery, however, further research is needed to underline the ecological mechanisms of co-occurrence of these species (Lombardi et al. 2020a).
1.4 Family: Procyonidae
Three procyonids are found in North America’s rangelands, raccoons (Procyon lotor), ringtail (Bassariscus astutus), and white-nosed coati (Nasua narica). All are generalist omnivores occupying diverse habitats. Raccoons are highly flexible and cohabitate with humans. They inhabit grasslands and shrublands and are known to prey on grassland birds and nests. Raccoons are limited by water resources and tend to select sites near streams and riparian forests (Timm et al. 2016; Berry et al. 2017). Their expansion to arid rangelands is partially attributed to anthropogenic water resources (Kamler et al. 2003b). Raccoons thrive on anthropogenic resources and have flexible social organization, from solitary in natural habitats to social in urban habitats. Interactions among raccoons and with other species are a concern for the transmission of pathogens (Hirsch et al. 2013). Raccoon populations are rarely assessed but they are considered to be stable or increasing.
Ringtail and the white-nosed coati are semi‐arboreal species, although ringtails may be more appropriately described as scansorial because they primarily occur in areas with little or no tree cover by exploiting canyons and similar orographic features. Ringtails are found in diverse habitats that include forests, deserts, rocky cliffs, and tropical areas and withstand low levels of disturbance and human habitation (Reid et al. 2016c), from southern Oregon south into Mexico. There are no data on ringtail populations (Reid et al. 2016c), and their status varies from furbearer to fully protected in states within the United States where they are found.
White-nosed coatis occur in low densities in southwestern rangelands of the United States. They are common in tropical habitats, and are also found in hardwood riparian forests of deserts in the southwestern United States and Mexico (Cuarón et al. 2016b). White-nosed coati are decreasing globally, although abundant in some areas (Cuarón et al. 2016b).
1.5 Family: Mephitidae
Five mephitids occur in western rangelands. The striped skunk (Mephitis mephitis) is the most common and widespread (Helgen and Reid 2016a). Striped skunks are opportunistic feeders, have an omnivorous diet that consists mostly of insects, but rodents, birds, and fruits are also consumed (Greenwood et al 1999). Populations are rarely assessed, but they have expanded their range in Canada (Long 2003). Eastern spotted skunks (Spilogale putorius) and western spotted skunks (Spilogale gracilis) occupy the east and west portions of the United States, respectively, and there is some overlap across a portion of the Great Plains (Cuarón et al. 2016c; Gompper and Jachowski 2016). The population of western spotted skunks is unknown but thought to be decreasing alongside decreasing prairie habitat, while eastern spotted skunks are declining throughout their range (Gompper and Hackett 2005). Spotted skunks consume invertebrates, small mammals, snakes, amphibians, birds, and plants, along with scavenging large mammalian prey (Sprayberry and Edelman 2016). Hog-nosed skunks (Conepatus leuconotus) and hooded skunks (Mephitis macroura) range from the southwestern United States through much of Central America (Cuarón et al. 2016a; Helgen 2016). Hog-nosed skunks are insectivorous (Hall and Dalquest 1963), more so than other skunks, but also an opportunistic feeder that consumes a variety of small vertebrates and fruits (Dragoo and Honeycutt 1999). The population is declining (Dragoo and Sheffield 2009). There are almost no studies on the diet of hooded skunks, considered to be omnivores, and their populations are considered stable (Cuarón et al. 2016a). Mephitids are fairly opportunistic and adaptable to differing conditions.
1.6 Family: Mustelidae
The most diverse group of mesocarnivores are the mustelids. Comprised of seven species, North America’s mustelids differ in size, specialization, habitat selection, diet composition, and activity patterns. The two semi-aquatic species, river otters (Lontra canadensis) and mink (Neovison vison) occur throughout much of North America. River otters occupy rivers and streams. Their populations were extremely reduced in the first half of the twentieth century. Thanks to habitat restoration, stricter regulations around harvest, and reintroduction programs, they now occupy about 90% of their historical range (Roberts et al. 2020). Despite their impressive recovery, otter densities and reproductive success are susceptible to heavy metals and polycyclic aromatic compound contamination in rivers and food resources (Thomas et al. 2021). River otters primarily consume fish and cetaceans (Melquist et al. 2003).
Mink are obligate carnivores that occupy areas by small streams, marshes, and dense vegetation (Reid et al. 2016b; Holland et al. 2019). Captive populations of mink are found throughout the United States and maintained for their fur. Farm minks may come into contact with wild animals through fence lines or by escaping confinement, which could spread diseases to wild animals; captive mink have transmitted COVID-19 to wildlife (e.g., Shriner et al. 2021). Mink are highly adaptable, considered generalist predators that eat fish, invertebrates, birds, amphibians, and small mammals. They primarily consume muskrats and lagomorphs in much of their native range (Dunstone 1993).
American badgers (Taxidea taxus) are widespread throughout western rangelands. Their diet mostly consists of small mammals but they also consume birds, reptiles, and insects (Helgen and Reid 2016b). Despite being characterized as generalists, badgers select for prairie dog (Cynomys spp.) colonies when available (Grassel and Rachlow 2018). Badgers are also considered ecosystem engineers where their den mounds contribute to soil nutrient patchiness that in turn affects vegetation composition (Eldridge and Whitford 2009). Additionally, badger burrows provide subterranean habitat to a wide diversity of species (Andersen et al. 2021).
Black-footed ferrets (Mustela nigripes) are one of the most endangered mammals in North America. Black-footed ferrets are obligate carnivores that feed mostly on prairie dogs (Brickner et al. 2014). The extermination of prairie dogs from North American rangelands caused the precipitous decline of black-footed ferrets (Knowles et al. 2002). Black-footed ferrets were believed to be extinct when the last known population died out in South Dakota until a small population was discovered in 1981 in Wyoming. Since then, several breeding facilities have been established. Ferrets have been reintroduced to 30 sites across the Great Plains, and as of 2019 occur in 23 sites. Reintroductions were successful as long as sufficient prairie dog acreage remained (Santymire and Graves 2019). Sylvatic plague, caused by the bacteria Yersinia pestis, is a significant threat to prairie dog persistence, can be contracted by black footed ferrets, and therefore threatens the existence of black-footed ferrets. Today, ~ 300 black-footed ferrets remain in the wild, though only 1–2 reintroduced populations are considered potentially viable (Belant et al. 2015). An additional ~ 320 captive individuals are still maintained and continue to be important to the population recovery program (Goldman 2021).
There are three weasel species in North America. The least weasel (Mustela nivalis) is the smallest mesocarnivore in the world. They are the most fossorial and subnivean in their hunting strategies of the three weasels. The least weasel, along with the short-tailed weasel (Mustela erminea), have a Circumboreal Holarctic distribution (Reid et al. 2016a; McDonald et al. 2019). They occupy diverse habitats that include grasslands, shrublands, riparian, tundra, and farmlands. The long-tailed weasel (Mustela frenata) is common throughout parts of Canada and the United States, and its range extends to northern South America (Helgen and Reid 2016c). All three weasel species feed predominantly on rodents and other small mammals. They also are all commonly regulated and harvested as a single group (i.e., weasel), despite little knowledge of population size or trends and some concern about population sizes of the least weasels, which are listed as a species of greatest concern in some states.
2 Intraguild Associations
The spatial organization of mesocarnivore communities is influenced not only by the distribution of resources (e.g., prey) and abiotic factors, but also intraguild interactions (Schoener 1974; Thompson and Gese 2007). The frequency and intensity of interspecific interactions among mesocarnivores often depends on multiple factors including dietary niche overlap, temporal activity patterns, and resource availability (Heithaus 2001; Donadio and Buskirk 2006; Atwood et al. 2011).
2.1 Dietary Overlap
Exploitative competition is is likely widespread among mesocarnivores as it is often inferred from patterns of dietary overlap. Sympatric canid species demonstrate high levels of dietary overlap and likely compete for resources. For example, red foxes have high dietary overlap with endangered San Joaquin kit foxes (V. m. mutica; Clark et al. 2005). Coyotes are considered a generalist and are the most widespread canid in western rangelands (Gompper 2002). Coyotes have high dietary overlap with sympatric red foxes (Azevedo et al. 2006), swift foxes, (Kitchen et al. 1999), kit foxes (Byerly et al. 2018), and gray foxes (Neale and Sacks 2001). Coyote diet overlaps with badgers for consumption of small mammals, but they may form hunting associations that compliment instead of compete for access to prey (Minta et al. 1992; Thornton et al. 2018). Striped skunks and raccoons are widespread generalists, and have more omnivorous diets that primarily include insects, plant materials, and eggs (rather than mammalian prey). Their diets likely put them in competition with one another but limits competition with canids, felids, and mustelids (Azevedo et al. 2006).
While many mesocarnivores have omnivorous diets, the felids are hypercarnivorous. This dietary specialization helps them secure taurine, an essential amino acid found in animal protein (Hedberg et al. 2007). Even so, bobcats and coyotes have also been shown to have high dietary overlap but may partition dietary resources. In Arizona, bobcats and coyotes both consumed rodents and lagomorphs, but bobcats consumed more rodents whereas coyotes consumed more lagomorphs; coyotes also supplemented their diet with larger prey (e.g., deer [Odocoileus spp.], javelina [Tayassu tajacu]) and plant material (e.g., fruit, seeds), which were consumed infrequently by bobcats (McKinney and Smith 2007). These examples illustrate how competition among species can vary in intensity among mesocarnivore dyads.
Dietary overlap among sympatric mesocarnivores often changes seasonally. For example, diets of gray foxes included fruit during summer and fall, creating relatively high dietary overlap with coyotes but little overlap with bobcats. In contrast, during winter and spring when fruits are not available, gray foxes shift to using rodents and therefore reduce dietary overlap with coyotes and increase dietary overlap with bobcats (Neale and Sacks 2001). Changes in the amount of dietary overlap across seasons is more likely to occur when at least one species practices seasonal shifts in their diet.
2.2 Intraguild Predation
Interference competition among heterospecific mesocarnivores often manifests as intraguild predation or interspecific killing. Intraguild predation occurs when two species compete for limited resources and also prey upon one another (Polis et al. 1989). Interspecific killing, often described as a form of intraguild predation, is an intense form of interference competition in which an individual of one species kills (but does not consume) a competitor (Lourenço et al. 2014). In mammalian predator guilds, intraguild predation is typically asymmetric with larger, and often more generalist predators (the intraguild predator), killing smaller, and often more specialized predators (the intraguild prey; Polis et al. 1989; Verdy and Amarasekare 2010). The resource-ratio hypothesis (Holt and Polis 1997; Miller et al. 2005) predicts three stable states for intraguild predation systems. Under resource-poor conditions, resources are insufficient to support the intraguild predator but are sufficient to support an intraguild prey that is a superior exploitative competitor. When resource levels are high, the intraguild predator is supported at sufficient levels to exclude the intraguild prey. Finally, only when resources are at intermediate levels can stable coexistence between the intraguild predator and prey be achieved, provided the intraguild predator benefits from the consumption of the intraguild prey. In western rangelands, coyotes and kit foxes have been used as a model system to evaluate patterns of intraguild predation. Studies found evidence for all three predicted stable states (Robinson et al. 2014; Lonsinger et al. 2017). In southern New Mexico, kit foxes persisted in resource-poor environments that excluded coyotes (Robinson et al. 2014). In western Utah, coyote abundance in resource-rich habitats was sufficiently high to competitively exclude kit foxes (Lonsinger et al. 2017). Habitats with intermediate resource levels supporting the coexistence of both species were identified in New Mexico shrublands (Robinson et al. 2014) and Utah grasslands (Lonsinger et al. 2017).
Despite predictions of the resource-ratio hypothesis (Holt and Polis 1997) and models of intraguild predation, which predict the coexistence of an intraguild predator and intraguild prey is unlikely when dietary overlap is high (Heithaus 2001), the coexistence of intraguild mesocarnivores is common. The coexistence of mesocarnivores may be facilitated by alternative prey (Holt and Huxel 2007), behavioral avoidance (Wilson et al. 2010), or increased vigilance of the intraguild prey (Kimbrell et al. 2007). Alternative prey available to the intraguild predator and outside of the handling capacity of the intraguild prey is common in mesocarnivores and likely contributes to coexistence in western rangelands. For example, coyotes kill ungulates, which are not common prey for sympatric fox species (Kitchen et al. 1999; Neale and Sacks 2001; Azevedo et al. 2006; Byerly et al. 2018). Patterns of avoidance that may stabilize coexistence include spatial or temporal resource partitioning. Spatial avoidance of intraguild predators by intraguild prey (i.e., safety matching) has reportedly facilitated the co-occurrence of coyotes with both swift foxes (Thompson and Gese 2007) and bobcats (Wilson et al. 2010). Patterns of predation risk and safety matching may also be influenced by sex. For example, avoidance of badgers (an intraguild predator) was stronger for female than male black-footed ferrets (an intraguild prey; Grassel et al. 2015). When mesocarnivores rely on similar prey resources, the temporal availability of prey may limit temporal partitioning. Even so, temporal partitioning may be important in facilitating co-occurrence between intraguild predator and prey. For example, gray foxes used water sources in Texas at times that minimized the potential for interactions with coyotes and bobcats (Atwood et al. 2011). Finally, the coexistence of intraguild predator and prey may be facilitated by increased vigilance by the intraguild prey, which decreases the foraging efficiency of the intraguild prey and may lead to decreased vigilance and increased susceptibility of shared prey (Kimbrell et al. 2007). Increased vigilance is likely common for intraguild prey, which must consider predation risk while foraging, leading to changes in behavior that can influence shared prey (Rosenheim 2004).
Patterns of higher intraguild predation may lead to reductions in mesocarnivore populations (i.e., mesocarnivore suppression), whereas lower intraguild predation may lead to increases (i.e., mesocarnivore release; Soulé et al. 1988; Crooks and Soulé 1999). Eradication of large carnivores from many rangeland systems has relaxed top-down pressures, allowing mesocarnivores to increase in abundance, ultimately increasing pressure on prey populations (Prugh et al. 2009). These increases may be further exacerbated by relaxed bottom-up constraints, particularly when synanthropic mesocarnivores (species ecologically associated with humans) benefit from anthropogenic subsidies (e.g., food via trash or crops, increased denning structures). In some cases, coyotes took the role of apex predators in areas where larger carnivores were extirpated (Cherry et al. 2016; Schuttler et al. 2017). In rangelands, coyotes limit prey species such as pronghorn (Antilocapra americana; Berger and Conner 2008). The implications of mesocarnivore suppression for species conservation and management can be complicated. For example, coyotes are a primary source of mortality across their range for smaller sympatric foxes, including swift and kit foxes (Nelson et al. 2007; Thompson and Gese 2007) and endangered San Joaquin kit foxes (Cypher and Spencer, 1998). Consequently, to induce mesocarnivore release by San Joaquin kit foxes, coyotes have been lethally controlled in some areas (Cypher and Scrivner 1992). However, despite the lethal removal of nearly 600 coyotes in five years from one site, indices of abundance and survival rates did not increase, nor did fewer foxes get killed by coyotes (Cypher and Scrivner 1992). Further, Clark et al. (2005) cautioned that coyote control could harm San Joaquin kit foxes by reducing competitive exclusion of nonnative red foxes by coyotes and increasing the potential negative interaction between red foxes and kit foxes, which have greater dietary overlap than kit foxes and coyotes.
3 Rangeland Management
3.1 Livestock Conflicts
Several mesocarnivores, especially the coyote, have been the focus of lethal and nonlethal tools to reduce livestock depredation in western rangelands for over a century. An abundant population of mesocarnivores may result in increased livestock loss through direct predation. Direct predation by abundant mesocarnivores has received the majority of attention concerning mesocarnivores on and around rangelands, and is generally discussed in terms of carnivore-livestock conflict (e.g., Scasta et al. 2017; Mosley et al. 2020). That is, mesocarnivores are suggested to negatively impact livestock through direct predation, which requires some type of lethal or non-lethal intervention to decrease losses (e.g., Andelt 1992; Knowlton et al. 1999; Mitchell et al. 2004; Scasta et al. 2017). However, the discussion and severity of mesocarnivore-livestock conflicts seem to vary across taxonomic groups and among species that are common and rare. Further, the scientific knowledge concerning mesocarnivore ecology related to livestock ranching and rangelands beyond direct predation is limited. For instance, evaluating how mesocarnivores may reduce potential forage competitors of livestock such as jackrabbits (Lepus spp.) or prairie dogs has received some mention, yet little empirical attention, despite evidence of jackrabbits consuming a substantial portion of livestock forage (Ranglack et al. 2015). The mesopredator release hypothesis (Soulé et al. 1988; Crooks and Soulé 1999) could apply to this context (e.g., Prugh et al. 2009; Prugh and Sivy 2020). We would expect abundant mesocarnivore populations to have a positive effect on livestock forage by reducing herbivore populations, which is a mechanism that has received empirical support (Henke and Bryant 1999).
Despite the possible strong role of mesocarnivores to rangelands, few studies have evaluated questions associated with how intact and working rangelands contribute to mesocarnivore habitat and conservation, or how mesocarnivores may provision ecosystem services on rangelands. As a contemporary demonstration, we performed a literature search over the last 20 years (i.e., 2000–2021) within the Web of ScienceFootnote 1 database. We searched for articles using search terms of the mesocarnivore species and the term ranch; an example for American badgers was “badger” & “ranch.” This combination of terms returned all articles that included badger and ranch (e.g., could be ranching, ranchland, etc.), which we then examined to ensure the articles were relevant. Once all articles were gathered, we determined if the main theme of articles were associated with livestock conflict, which we used to classify articles into two groups: (1) was or (2) was not associated with conflict. Results from this assessment indicated that (1) there are very few articles assessing mesocarnivore ecology and ranching, and (2) that most articles discussing coyotes on rangelands are associated with livestock conflict (Fig. 16.2). Consequently, there is vast potential to advance the state of understanding in the future regarding the role of working rangelands in the ecology and conservation of mesocarnivores.
Mesocarnivore studies referencing rangelands during 2000–2021. Data were gathered from Web of Science using search terms: mesocarnivore species* and ranch*. Thirty-seven relevant studies were gathered from search results. We determined if the main theme of the article was associated with livestock conflict or not. Studies of coyotes (Canis latrans) were generally about conflict with livestock
3.2 Canid Predation
Coyotes are the most discussed mesocarnivore concerning ranching and rangelands. Most assessments of coyotes on rangelands have been in the context of livestock predation and losses (Knowlton et al. 1999; Larson et al. 2019). Coyotes have been documented killing and consuming livestock (Knowlton et al. 1999; Blejwas et al. 2002; Sacks and Neale 2002; Palmer et al. 2010) and a suite of techniques, both lethal and non-lethal, have been implemented to reduce livestock losses. For instance, lethal removal in the form of trapping, calling and shooting, and aerial gunning have been employed to reduce livestock depredation (Knowlton et al. 1999; Blejwas et al. 2002). Non-lethal control in the form of livestock husbandry, fencing, electronic frightening devices, livestock guard dogs, and sterilization of breeding pairs have also been used to reduce livestock losses from coyotes (Andelt 1992; Bromley and Gese 2001; Knowlton et al. 1999; van Eeden et al. 2018a; Bromen et al. 2019; Mosley et al. 2020). Despite differing approaches to limit, reduce, or eliminate livestock losses from coyotes, few rigorous studies exist where the consequence of treatment have been effectively evaluated in a controlled or semi-controlled context (Eklund et al. 2017; van Eeden et al. 2018b). This pattern has also been observed in wildlife studies, where the impacts of predator management or removals on measurable outcomes (e.g., recruitment of young) of prey species is absent, muddled, and/or context-dependent (Ballard et al. 2001, but see Mahoney et al. 2018 and Seidler et al. 2014). Rigorous experimental work over longer timeframes is needed to assess which techniques may be most effective in preventing livestock losses across different landscapes and ecological communities (van Eeden et al. 2018b). No technique will be universally effective and often a variety of approaches may need to be used in an integrated and complimentary fashion (Knowlton et al. 1999).
Despite numerous studies evaluating and discussing modifications to coyote density or behavior to reduce livestock losses, very few studies have evaluated questions related to coyote behavior and demography as a consequence of management efforts. An exception was a study in southeastern Colorado that demonstrated coyote diurnal activity increased with a reduction in human persecution (i.e., shooting, trapping, and intense aerial gunning; Kitchen et al. 2000). This alteration in activity may have had carry-over effects on coyote diet or other interspecific interactions, but this was not evaluated as part of the study. As researchers and managers seek to understand the effectiveness of different treatments to reduce livestock losses, there will be additional opportunities to assess the consequences of treatments on coyote ecology.
Beyond coyotes, few studies have assessed the relationship between livestock ranching and the ecology of other medium-sized canids. However, many studies on the ecology of foxes (e.g., space use, den site selection, and movement) have occurred on rangelands and may be useful to predicting relationships between livestock ranching and canid ecology. For example, studies of space use, den site selection, and movement of swift foxes has often occurred on rangelands (Nicholson et al. 2006, 2007; Sasmal et al. 2015; Butler et al. 2019, 2020). Even so, many additional questions remain concerning the ecology of medium-sized canids and livestock ranching.
3.3 Felid Predation
There is less research regarding livestock ranching and rangelands and medium-sized felids relative to that of canids. Some studies have assessed livestock losses by bobcats (e.g., Scasta et al. 2017; Bromen et al. 2019), while others have evaluated bobcat occupancy or density on rangelands (Greenspan et al. 2020; Lombardi et al. 2020a). Ocelots have received more attention with specific regard to rangelands and detailed questions about their ecology. This is likely because the majority of ocelot habitat in the United States occurs on private ranchlands (Lombardi et al. 2020b), and thus successful conservation efforts require ranchland connectivity. Beyond the importance of working ranchlands for ocelot conservation, additional questions remain regarding the influence of different ranching practices on both bobcats and ocelots, and the potential interactions between these species on private ranches.
3.4 Mustelid, Mephitid, and Procyonid Predation
The majority of species within the families Mustelidae, Mephitidae, and Procyonidae have received little-to-no attention concerning their ecology on rangelands. Some notable exceptions, however, include black-footed ferrets, American badgers, and raccoons. For example, research on black-footed ferrets has examined habitat use and resource selection of reintroduced individuals on rangelands in New Mexico (Chipault et al. 2012). In Wyoming, researchers have recently examined the role of habitat provisioning wherein American badgers make subterranean habitat accessible to non-fossorial species; this work assessed the diversity and frequency of species using abandoned badger burrows on rangelands (Andersen et al. 2021). Finally, researchers have evaluated the role of raccoon fecal deposition on Escherichia coli (E. coli) distribution and concentration within Texas floodplains (Parker et al. 2013). There remain many additional opportunities to better assess the ecology of these species on working rangelands.
4 Harvest of Mesocarnivores
The harvest of mesocarnivores has a long history in North America. Humans have harvested mesocarnivores in North America for thousands of years to use for clothing, food, and religious ceremonies (Wright 1987). The Indigenous peoples of western North America harvested mesocarnivores such as badger, mink, raccoon, skunks, weasels, and wildcats (Lynx spp.) for their pelts for ceremonial decoration, clothing, quivers, baskets, and occasionally for eating before the arrival of Europeans (McGee 1987). Systematic fur trading, primarily for American beaver (Castor canadensis) pelts, between Europeans and Indigenous peoples began in the 1580s in northeastern North America (Obbard et al 1987). In the late 1700s and early 1800s, European and American fur traders expanded into the southwest and Great Plains of the United States (Ray 1987). Across North America, European and American fur trading companies kept records that have provided insights into the scale at which fur trapping occurred (Obbard et al. 1987; Ray 1987).
Harvest levels of mesocarnivores before the 1930s are difficult to decipher as fur records are incomplete, and the most continuous data sets come from the Hudson Bay Company, which predominately operated in Canada (Obbard et al. 1987). Using data from the Hudson Bay Company and other fur trading companies, Obbard et al. (1987) found that the annual mean harvest of foxes (all species grouped), raccoon, mink, and bobcat (grouped with Canada lynx; Lynx canadensis) substantially increased from a few thousand pelts to tens or hundreds of thousands of pelts annually from the 1800s to the 1980s in North America. River otter annual mean harvest also increased from a few thousand to approximately 43,000 in the 1780s, then declined to less than 16,000 between 1900 and 1909, and finally rebounded again to historically high levels starting in 1980. Ringtail and badger harvests were lower than some other species, likely because of the low utility and quality of their pelts. Annual mean harvest of ringtails and badgers largely began after 1900 and reached approximately 70,000 and 38,000 in the 1980s, respectively. Like foxes, historical harvest records of skunks encompassed all species of skunks until the 1900s, and weasel records continue to group species together. Skunk annual mean harvest increased from 113,000 in the 1920s to 1.2 million by the 1930s and 1940s, and weasel annual mean harvest increased from 490,000 to 900,000 over the same time frame. Both declined, with annual mean skunk harvest of 38,000 in the 1960s and annual mean weasel harvest of 71,000 in the 1980s.
Up until the early 1900s, coyote annual mean harvest totals were likely combined with those of wolves (C. lupus) making it difficult to identify long-term trends. Coyote annual mean harvest fluctuated between 20,000 and 107,000 pelts between the 1920s and 1950s before increasing to 500,000 by the 1980s (Obbard et al. 1987). The totals are from a time when the harvest was largely unregulated, which began to change at the beginning of the twentieth century when some species became locally scarce or extirpated (Hubert 1982).
Furbearer management began to include trapping and fur buyer licenses, and annual trapper surveys in the early 1900s (Hubert 1982). Regulations on harvest such as duration of the season, bag limits, and trapping best management practices for each of the 23 species of furbearing animals in North America were put in place to maintain a sustainable yield for perpetuity (White et al. 2015; White et al. 2021). The implementation of regulations varied by state and species during the 1900s. For example, in 1971, bobcats were unprotected in 10 of 12 western states (Faulkner 1971) but by 1987, protection was given to bobcats in all western states (Melchior et al. 1987). In contrast, raccoons changed from regulated to less regulated harvests. They were harvested during distinct hunting seasons without bag limits in eight midwestern states and year-round in one state with no bag limit in 1982 (Melchior et al. 1987). In 1993, seven midwestern states had hunting seasons with no bag limits, one state had a hunting season with a bag limit of 20, and two states allowed hunting year-round with no bag limit (Rogers 1995). In 2017, harvest was widespread with 14 species or groups of mesocarnivore in 22 midwestern and western states totaling 1,049,994 individuals (Association of Fish and Wildlife Agencies 2015, unpublished data).
In addition to records during the fur trade, harvest surveys and harvest efforts have historically been used to track population levels of mesocarnivores (Clark and Andrew 1982; Roberts and Crimmins 2010; Roberts et al. 2020). Many factors influence furbearer harvest totals beyond population abundance such as trapper experience, trapping regulations, trapper effort, pelt prices, and winter weather (Elsken-Lacy et al. 1999; Ruette et al. 2003; DeVink et al. 2011). These factors potentially confound the ability of harvest data to indicate biological changes in the population (Allen et al. 2020). Recently, many studies have evaluated the influence of different combinations of these factors simultaneously and found that the direction and relative importance of these effects on harvest varied by species (Hiller et al. 2011; Kapfer and Potts 2012; Ahlers et al. 2016; Allen et al. 2018; Bauder et al. 2020a, b). For example, winter temperature had a strong negative effect on bobcat harvest in Minnesota (Kapfer and Potts 2012), but only a weak negative effect on raccoon harvest in Illinois (Bauder et al 2020b). Raccoon, red fox, and gray fox harvests in Illinois were positively influenced by gasoline prices, while gasoline prices had a negative influence on muskrat (Ondatra zibethicus) harvest in Illinois (Ahlers et al. 2016). While the harvest of some mesopredators is not influenced by pelt prices (Hiller et al. 2011), pelt prices influenced harvest of other species, including red fox, gray fox, and muskrat (Ahlers et al. 2016). Collectively, these studies indicate biological trends can be inferred from harvest data after accounting for confounding factors. However, as these studies were conducted in the midwestern United States, it is currently unknown if their findings apply to harvest data from the western United States and Canada, which could vary in management regulations and economic and social pressures. Therefore, future research should investigate how these factors, and others, influence the harvest of mesocarnivores in rangeland-dominated jurisdictions.
Harvest of mesocarnivores is not entirely for the fur but also a source of sport for some hunters, although hunting for sport is not mutually exclusive to harvest for fur. Coyotes are often the focal animal for sport hunters. In many western rangeland systems, local communities, counties, and other organizations host coyote hunting contests, also called coyote call contests. These contests typically provide a cash reward for the most animals killed within a set time. Foxes and other mesocarnivores may also be the focus of these contests. In recent years, some western states have banned hunting contests; for example, New Mexico and Arizona banned these contests in 2019 and 2021, respectively (NM S-B76 2019, AZ AC R12-4-303 2021).
5 Predator Control
In addition to legal harvest of mesocarnivores for fur, food, and sport, mesocarnivores have also been killed for centuries to reduce human-carnivore conflicts. These events occur in response to actual or perceived threats of mesocarnivores to domestic livestock or wildlife populations (Reynolds and Tapper 1996). The earliest form of formal predator control was the bounty system. Beginning in 1800, every state in the midwestern United States has had a bounty placed on a mesocarnivore, most often coyote, red fox, and gray fox (11 states), but also bobcat (8 states) and badger (1 state; Hubert 1982). Moreover, bounty programs across North America for species such as coyote, red fox, and bobcat remained in effect through the 1970s and 1980s (Hubert 1982; Slough et al. 1987) and into the twenty-first century, with coyote bounty programs in effect in South Dakota, Texas, and Utah through 2003 (Bartel and Brunson 2003). There is currently a coyote bounty program in Utah that began in 2012, where participants can receive up to 50 USD per coyote submitted, and a nest predator bounty program in South Dakota for raccoons, badgers, striped skunks, red foxes, and Virgina opossums (Didelphis virginiana) that began in 2019, where participants can receive up to 10 USD per tail submitted.
Impacts of bounty programs are largely unclear. There is no consolidation of records to determine how many mesopredators have been killed through bounty programs but there are reports from some bounty programs to indicate high numbers of animals are taken. One report from 1972 documented that 111,569 coyotes, 17,169 bobcats, and 494,635 foxes were taken in Michigan between 1935 and 1970, accounting for 4.46 million USD in payments (Cain et al. 1972). Although the number of trappers and hunters has declined across the years, the data for number of animals submitted to current bounty programs remains high. In South Dakota, 54,471 tails were submitted in 2019 and 26,390 tails were submitted in 2020; data are available at the agency’s website: https://sdgfp.maps.arcgis.com/apps/opsdashboard/index.html#/e7bbbd6fa93b48c6a31985aa7c57c5ff. For the Utah bounty program, 7160 coyotes were submitted in its first year, 2013 (UDWR 2013). The number of coyotes submitted annually has ranged from the 7000s to 10,000s, although only 4109 coyotes were submitted in 2020. This low number may have been due to COVID-19 restrictions. Although the number of animals submitted to bounty programs appears high, it is not clear if bounty programs have the desired effect of reducing predation pressure on livestock or other wildlife. Research on the effects of removing several species of the mesocarnivore community (typically coyotes, raccoons, red foxes, and striped skunks) on waterfowl nesting success showed conflicting effects (Rohwer et al. 1995; Sargeant et al. 1995; Docken 2011; Blythe and Boyce 2020), no effect on upland bird populations (Guthery and Beasom 1977; Lawrence and Silvy 1995; Frey et al. 2003; Lyons et al. 2009; Docken 2011; Reid 2019), and no effect on rodent and lagomorph densities (Guthery and Beasom 1977; Henke and Bryant 1999). Similarly, studies on the effects of coyote removal on other mesocarnivores showed conflicting results (Henke and Bryant 1999; Kamler et al. 2003a; Karki et al. 2007). There is evidence that coyote removal has short-term positive effects on white-tailed deer (O. virginianus), mule deer (O. hemionus), and pronghorn (Guthery and Beasom 1977; Harrington and Conover 2007; Brown and Conover 2011; Mahoney et al. 2018).
In the United States, federal programs aimed at the prevention and control of wildlife damage emerged near the end of the nineteenth century and have been the primary entity responsible for reducing livestock depredation rates (Miller 2007). Known today as the United States Department of Agriculture—Animal and Plant Health Inspection Service—Wildlife Services (WS), WS became involved in predator control in 1915 when charged with reducing wolf and coyote livestock depredation. Subsequently, part of their mission was to research and develop new lethal predator control methods (Miller 2007; Feldman 2007) such as traps, M-44's, predacides, ground shooting, snares, denning, dogs, and aerial shooting (Evans and Pearson 1980). Coyote depredation continues to be the largest cause of cattle and sheep predator loss in the United States in 2015 (United States Department of Agriculture—National Agricultural Statistics Service 2015a, b), which has been confirmed by research on sheep (Palmer et al. 2010). WS killed or euthanized 68,905 coyotes in 2015 and killed or euthanized 62,002 coyotes during WS operations in 2019 (Wildlife Services 2015, 2019). These numbers are similar to the annual number killed during the 1970s (Evans and Pearson 1980) even though there are dramatically lower numbers of sheep in the United States today. Wagner and Conover (1999) found that aerial shooting of coyotes decreases the number of lambs lost to coyotes. However, this may not be a long-term solution given the financial costs and high immigration rates in exploited populations (Kilgo et al. 2017). Although an evaluation of the program’s efficacy has been called for across decades (Wagner 1988; Shivik 2014), no such evaluation has occurred. This is important because recent research has investigated the efficacy of nonlethal methods to reduce coyote depredation such as sterilization (Seidler and Gese 2012; Young et al. 2019b), livestock guard dogs (Kinka and Young 2018; Saitone and Bruno 2020), and fladry (Young et al. 2019a), which showed promising results and should continue to be part of an integrated approach and a focus of future studies.
6 Impacts of Disease
6.1 Disease Concerns for Mesocarnivore Populations
Disease enzootics in mesocarnivores can have significant implications for the dynamics of mesocarnivore communities, disease management, and human perceptions of mesocarnivores. Mesocarnivores serve as a primary reservoir for generalist pathogens (i.e., those having a wide host range), which may be transmitted among native carnivore species or spread to domestic animals (Roemer et al. 2009). Generalist pathogens maintained by mesocarnivores may also be zoonotic (i.e., transmitted between animals and humans) and, therefore, represent a public health concern. Generalist viruses and pathogens of principal concern for mesocarnivores in western rangelands include canine distemper virus (Morbillivirus sp.), parvoviruses (including feline panleukopenia virus and canine parvovirus; genus Parvovirus), rabies (genus Lyssavirus), and mange (primarily sarcoptic Sarcoptes scabiei and notoedric Notoedres cati).
Canine distemper virus is an infectious disease that impacts all families of terrestrial carnivores and has high mortality rates in some species (Deem et al. 2000). Being highly transmissible, canine distemper infections occur through contact with or inhalation of aerosolized virus, which may be shed by an individual through respiratory droplets or bodily secretions for up to 90 days after infection, and can persist in the environment for up to 14 days (Deem et al. 2000; Anis et al. 2020). The impact of canine distemper varies among species. Canine distemper is enzootic in raccoons and gray foxes, which serve as reservoirs in North America (Deem et al. 2000). Raccoons support the coinfection of multiple canine distemper strains, potentially leading to new strains (Pope et al. 2016). Among mesocarnivores, mustelids appear to be the most susceptible to canine distemper with critically high mortality rates approaching 100% (Beineke et al. 2015). For this reason, canine distemper has been considered the “most significant infectious disease” limiting endangered black-footed ferrets, which are also susceptible to canine distemper induced through a modified-live vaccine (Williams et al. 1988, 1996).
Parvoviruses impacting mesocarnivores emerged in the 1970s and have continued to evolve (Steinel et al. 2001; Hueffer et al. 2003; Allison et al. 2012). Originally described as feline panleukopenia virus, cross-species transmission to canid hosts lead to canine parvovirus (CPV) type-2 and the subsequent establishment of two antigenic strains—CPV-2a and CPV-2—which regained the ability to infect felids and became widespread (Steinel et al. 2001; Allison et al. 2013; Stuetzer and Hartmann 2014). Parvoviruses are easily transmitted from hosts through contact with their feces or contaminated objects and can persist in the environment for extended periods (weeks to months; Steinel et al. 2001). Consequently, parvoviruses can be transmitted among individuals without direct contact and are highly contagious (Allison et al. 2013). The tendency of many mesocarnivores to use latrines, olfactory cues for territorial maintenance, and shared focal resources (e.g., water resources in arid rangelands; Atwood et al. 2011) likely contributes to the high transmission capacity of parvoviruses. CPV antibodies reported in coyotes (71–100%; McCue and O’Farrell 1988; Gese et al. 1991, 2004) and adult foxes (kit and swift foxes: 60–71%; Miller et al. 2000; Gese et al. 2004) were generally high, inidicating the virus may be enzootic in these species. Evidence of CPV antibodies was higher in adults than juveniles for coyotes and foxes (Miller et al. 2000; Gese et al. 2004). This is likely explained by the fact that parvoviruses impact juvenile survival significantly more than adults, which in turn may limit recruitment and threaten populations (Gese et al. 1997).
Rabies is a zoonotic, neurotrophic virus most commonly transmitted among conspecifics through a bite from an infected animal, but interspecific transmission may also occur (Hass and Dragoo 2006; Ma et al. 2020). Rabies initially evolved in bats and later spread to mesocarnivores (Kuzmin et al. 2012). Although direct evidence of transmission between bats and mesocarnivores is rare, multiple transmission events of a bat variant of rabies to striped skunks were detected in Arizona (Leslie et al. 2006; Kuzmin et al. 2012). Mesocarnivores are reservoirs for rabies, with striped skunks and, to a lesser extent, gray foxes maintaining enzootic levels of rabies in western rangelands (Hass and Dragoo 2006; DeYoung et al. 2009; Ma et al. 2020). Rabies has also been detected in hooded, hog-nosed, and western spotted skunks (Crawford-Miksza et al. 1999; Dragoo et al. 2004), but their role in maintaining the virus is unclear. Epizootics of the skunk variant of rabies have been associated with periods following increased precipitation and may be driven by increased density of skunks resulting from bottom-up processes (e.g., increased primary productivity and prey; Hass and Dragoo 2006). While most mesocarnivore rabies cases in western rangelands are attributable to skunks, foxes, and raccoons, rabies has been detected in bobcats and coyotes, and less frequently in mustelids and other procyonids. Management of rabies in wildlife populations in western rangelands has focused largely on surveillance of wildlife populations with targeted oral vaccination programs aimed at limiting the spread and reducing the risk of transmission to pets and humans. In Texas, oral vaccination programs guided by information on dispersal tendencies and population genetic structure of gray foxes (DeYoung et al. 2009) were successful in eliminating localized gray fox and coyote-dog variants of the virus (Slate et al. 2005).
Mange is a disease caused by mites that infest a host’s epidermis leading to intense irritation and itching, hair loss, and callousing of the epidermis (Niedringhaus et al. 2019). Secondary cracking of the skin, combined with bacterial or yeast infections, may lead to severe emaciation and death (Niedringhaus et al. 2019). Sarcoptic and notoedric mange are among the most common in mesocarnivores, with sarcoptic mange being widespread (Niedringhaus et al. 2019). Transmission of mange among individuals may occur directly through contact or indirectly through shared environments, with mites being able to persist under some environmental conditions without a host (Niedringhaus et al. 2019). Species relying on dens may be at heightened risk of mange infestation, owing to the increased potential for indirect transmission (Montecino-Latorre et al. 2019). Den use, particularly when dens are used by more than one individual, family group, or species, may facilitate the spread and cross-species transmission of mange (Niedringhaus et al. 2019; Montecino-Latorre et al. 2019). In North American rangelands, coyotes are a primary host of sarcoptic mange, though other canid species may also sustain the disease (Pence et al. 1983; Niedringhaus et al. 2019). The impact of mange is likely influenced by host age and condition, with infections being more rapid in juveniles (Pence et al. 1983). Population-level impacts of mange likely vary among species and populations. For example, endangered San Joaquin kit foxes infected with mange are unlikely to recover, even with treatment, and mange can represent a significant threat to population persistence (Cypher et al. 2017). In contrast, while mange-infected coyotes had increased mortality risk, these mortalities were compensatory and did not influence population-level survival (Pence et al. 1983). Furthermore, there is evidence that some coyotes and red foxes can recover from mange infections (Chronert et al. 2007; Nimmervoll et al. 2013). Although less widespread, notoedric mange has caused acute population declines in bobcats (Serieys et al. 2013) and has been detected in endangered ocelots (Pence et al. 1995).
6.2 Disease Concerns for Other Rangeland Animals
In western rangelands, disease epizootics in mesocarnivores are of greatest concern due to the potential for transmission from the reservoir host to other species, which could include imperiled species, domestic animals, and humans. Endangered black-footed ferrets and San Joaquin kit foxes are at high risk of being impacted by diseases, and disease outbreaks in these species have demonstrated the population-level threat of pathogens. Recovery of endangered black-footed ferrets has been hampered by direct impacts of diseases that cycle in sympatric canid populations (e.g., canine distemper virus and rabies; Williams et al. 1988, 1994; Gese et al. 1991) and indirect impacts of sylvatic plague (caused by Yersinia pestis) on prairie dogs, the black-footed ferret’s primary prey. The role of disease in regulating San Joaquin kit fox populations is less clear (Cypher et al. 2017). Antibodies of CPV have been detected in San Joaquin kit foxes, but it is suspected to be enzootic and has not been implicated in population declines (McCue and O’Farrell 1988). In contrast, canine distemper virus, rabies, and sarcoptic mange have all led to mortality events or substantial declines in at least one San Joaquin kit fox population (White et al. 2000; Cypher et al. 2017; Rudd et al. 2019). Mesocarnivores tend to occur in higher densities than large carnivores and disease spillover has the potential to influence sympatric populations of imperiled large carnivores. For example, the introduction of CPV to wolves on Isle Royale from a domestic dog (C. familiaris) resulted in a precipitous decline in wolves (Wilmers et al. 2006), and similar spillover events from mesocarnivore reservoirs could threaten recovery efforts of large carnivores (e.g., Mexican gray wolves [C. l. baileyi]) in western rangelands, but these community dynamics have not been well documented or studied (Roemer et al. 2009).
The complex mesocarnivore community offers primary and secondary reservoir species for most generalist pathogens, making disease management challenging. For small, isolated populations, such as those characterizing endangered mesocarnivores, the risk of pathogen-caused local extinctions is increased when generalist pathogens can persist in sympatric species and when transmission rates are artificially increased by anthropogenic habitat changes (Smith et al. 2009). Land management practices in western rangelands may influence disease dynamics by promoting wildlife aggregation (e.g., water developments, refuge habitats) and potentially increasing contact rates. Water catchments to support game populations and livestock have greatly expanded the availability of water (Rosenstock et al. 1999) and increased habitat for some mesocarnivores (e.g., raccoons; Kamler et al. 2003b). Catchments may increase contact rates among individuals and increase the potential for intra- and inter- specific disease transmission, but there is little empirical evidence for this hypothesis (Rosenstock et al. 1999) and mesocarnivores may be able to mitigate risks in some systems through spatial and temporal partitioning of catchment usage (Atwood et al. 2011). Similarly, many mesocarnivores may experience increased spatial and temporal overlap in refuge habitats within urban (e.g., parks) or agricultural (e.g., shelterbelts) landscapes (Sévêque et al. 2020).
Many mesocarnivores have shown a remarkable ability to exploit both natural and anthropogenic landscapes (Šálek et al. 2015). The potential for disease transmission from domestic animals to mesocarnivores, and vice versa, is of great concern. Domestic dogs and cats (Felis catus) are widespread, occur in high densities, and can serve as a host for many pathogens that impact mesocarnivores (Smith et al. 2009). The ability of some mesocarnivores (e.g., striped skunks and raccoons) to be synanthropic increases the risk of spillover of generalist pathogens between domestic animals and mesocarnivores, particularly if anthropogenic subsidies associated with agriculture or urban-suburban gradients increase the mesocarnivore densities, interactions among mesocarnivores and domestic animals, or both (Ordeñana et al. 2010; Tardy et al. 2014; Theimer et al. 2015).
7 Ecosystem Threats
There are several new and continuing threats to western ecosystems that are likely to have different impacts on mesocarnivores. For example, in the last several years extreme fire events and drought have become prevalent through much of the rangelands in the western United States, yet the impacts of such fires on mesocarnivores remains unclear (e.g., Holbrook et al. 2016). Coyotes in controlled burned areas prefer recently burned habitats, where their prey are also more abundant (Stevenson et al. 2019) but in areas where fires are more severe and uncontrolled, it is likely the same responses of prey and mesocarnivores are not possible due to slower recovery of vegetation and the larger extent of recent wildfires. Instead, extreme weather events and rapid development are likely to result in more homogenous habitats that may limit the ability of mesocarnivores to co-occur because of an inability to partition habitat that currently allows for co-occurrence (e.g., Mueller et al. 2018).
The two largest ecosystem threats to mesocarnivore populations are the use of biological resources and land-use changes (Marneweck et al. 2021), both of which are increasing (Bell et al. 2004; Willcox 2020). Rangelands are being transformed into housing for the growing human population or infrastructure to support humans, such as energy extraction and irrigated croplands (Ellis et al. 2010; DuToit et al. 2017). In the Prairie Pothole Region of the United States, where mesocarnivores have occurred with irrigated crops for a longer time, there are positive relationships between mesocarnivore abundance and crops (Crimmins et al. 2016), likely related to higher abundance of prey in this landscape. Thus, we may also expect to see increasing populations of mesocarnivores in rangelands that transition to irrigated crops.
Rapid oil and gas developments are carried out with limited data about environmental impacts (Allred et al. 2015). Surprisingly, there are few publications on the effects of gas and oil development on mesocarnivore populations even though gas and oil development has experienced substantial growth in western rangelands of the United States in recent decades. One study found that mesocarnivores have unique responses to increasing levels of oilfield development (Fiehler et al. 2017). Coyotes and badgers were active in areas with high levels of development, whereas, the San Joaquin kit fox selected fields with no or medium levels of development (Fiehler et al. 2017). Swift foxes are similar to the latter and may even prefer oilfields due to lower coyote densities (Butler et al. 2020). These findings illustrate the complexities of how mesocarnivores respond to changing landscapes and the need for further studies. Studies that currently focus on species at the edge of their range (e.g., Sacks et al. 2018) or in areas where they overlap with exotic or invasive species (e.g., Moreira-Arce et al. 2015) will likely provide the best metrics to forecast population ecology and management needs for mesocarnivores.
Development of alternative energies is another rapidly growing industry, with many wind and solar energy facilities being constructed in western rangelands (Agha et al. 2020). The southwest United States has been identified as having the largest potential for solar energy (Lovich and Ennen 2011; Kabir et al. 2018), and construction of new facilities in rangelands is expected to continue. While these alternative energies may provide net benefits to ecosystems, the immediate costs to local wildlife populations are only starting to be understood. Scavengers may benefit from carcass resources under wind turbines (Smallwood et al. 2010). However, to date, almost no information is available on the response of mesocarnivores to wind and solar facilities; most studies focus on impacts to threatened and endangered or aerial species (Allison et al. 2019; Chambers et al. 2017; Lovich et al. 2011). Agha et al. (2017) found that wind energy facilities influence mesocarnivore behavior, likely by creating access to new dirt roads as travel routes and changes in prey behavior occur. Studies from Europe have shown red foxes were tolerant of wind turbines (Łopucki et al. 2017), while European badgers (Meles meles) displayed elevated stress hormones in proximity to turbines (Agnew et al. 2016). In that and another study (Smith et al. 2017), mesocarnivores appeared to avoid wind turbines, which could create micro-refuges for prey. Further studies are needed to understand how the local environmental changes associated with wind farms, such as noise, presence of humans, traffic, construction and maintenance disturbance, visual alterations to the habitat, toxins, new smells, collision threats, and more, may have short and long-term effects on mesocarnivore behavior and population dynamics.
8 Research and Management Needs
As detailed in this chapter, mesocarnivores on rangeland systems are primarily either considered problematic for human endeavors, such as livestock grazing and disease risks, or ignored and understudied. In general, there is too little scientific information and research on mesocarnivores (Marneweck et al. 2021), even for those that have garnered more attention, like coyotes. Identifying ways to fill in research gaps are needed. For example, the formation of the Eastern Spotted Skunk Cooperative Study working group has advanced research and conservation of this understudied species (Jachowski and Edelman 2021). Their efforts to engage seemingly disparate researchers and agency personnel that previously worked independently of one another created more awareness and information than previous solo efforts. A similar model of harnessing the power of multiple research groups to broaden our understanding of a species is underway by one of the authors (J.K. Young) with 16 research teams studying coyotes. Today, the ease for which researchers and agency personnel can work collaboratively despite being at distant locations from one another has created new opportunities for cooperative research, management, and conservation groups to form.
Some mesocarnivore populations are stable or increasing but many are declining, and others are too data deficient to track effectively. Greater information on population statuses and reasons for population dynamics of mesocarnivores is needed. This is especially important because rangeland landscapes are rapidly changing due to climate change and anthropogenic impacts and this guild can serve as “sentinels for global change” (Marneweck et al. 2022) but only if we understand their current status and population dynamics.
As noted in 16.7, rangeland conditions are undergoing rapid changes, primarily through climate change and rapid human development, and how mesocarnivores adapt to new challenges is largely unclear. This is important to consider because mesocarnivores provide substantial ecosystem services that could be lost with shifting populations and behavior in response to accelerated global changes (Marneweck et al. 2021). Mesocarnivores that exhibit flexible dietary and habitat requirements, such as coyotes, raccoons, and skunks, will likely adapt to landscape modifications, others with stricter habitat or dietary requirements, like ocelots and black-footed ferrets, may suffer population declines. However, how populations that currently are considered adaptive actually cope with these changes remains to be seen too.
The rapid development of urban-wildland interfaces and the infrastructure to support burgeoning human populations are also indirectly impacting mesocarnivore populations via altering connectivity. It is likely that additional roads and other structures decrease connectivity which, in turn, can impact immigration, emigration and genetic diversity of populations (e.g., Butler et al. 2020). As much of the western United States rangeland system becomes more arid, mesocarnivores and their prey are likely to shift ranges to seek moister areas. In many cases, this shift will include seeking out artificial water sources created by humans, from water guzzlers to water fountains. Thus, as cities and towns expand, so does the potential for some mesocarnivore species to interact with feral and free-roaming dogs (Young et al. 2011). Combined, these form a human footprint that mesocarnivores are likely responding to in different ways behaviorally and spatially (e.g., Carricondo‐Sanchez et al. 2019). Many opportunities exist to study these potential impacts.
9 Summary
Our chapter highlights that mesocarnivores are important to ecosystems (Roemer et al. 2009) but they remain relatively understudied, especially in rangelands. The historical and current distribution, population status (when available), and conservation status of the 22 mesocarnivores in western rangeland landscapes illustrates how some species have thrived under changing conditions in western rangeland landscapes—increasing their range and population size—while others have been negatively impacted and are declining or at least unable to recover from earlier declines. Even so, most mesocarnivores continue to be affected by direct management actions, such as persecution for real or perceived human- and livestock- conflict and fur harvest, or indirect actions associated with the ever-growing anthropogenic footprint, such as increased number of roads, traffic, and developed landscapes that are typically the cause of decreased prairie and rangeland habitats. At the same time mesocarnivores are affected by management decisions and ecosystem threats, they are also impacted by diseases and intraguild interactions.
It remains unclear how ecosystem threats will further shift mesocarnivore populations and their distributions. Mesocarnivores serve as apex predators in some western ecosystems, but as populations of large carnivores continue to recover in the West and landscapes are converted by humans or human-caused climate change, it will be important to continue to study this important guild to determine their impacts on the natural and working landscapes they occupy.
Notes
- 1.
Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
References
Agha M, Lovich JE, Ennen JR, Todd BD (2020) Wind, sun, and wildlife: do wind and solar energy development ‘short-circuit’ conservation in the western United States? Environ Res Lett 15(7):075004. https://doi.org/10.1088/1748-9326/ab8846
Agha M, Smith AL, Lovich JE, Delaney D, Ennen JR, Briggs J, Fleckenstein LJ, Tennant LA, Puffer SR, Walde A, Arundel TR (2017) Mammalian mesocarnivore visitation at tortoise burrows in a wind farm. J Wildl Mgmt 81:1117–1124
Agnew RC, Smith VJ, Fowkes RC (2016) Wind turbines cause chronic stress in badgers (Meles meles) in Great Britain. J Wildl Dis 52(3):459–467. https://doi.org/10.7589/2015-09-231
Ahlers AA, Heske EJ, Miller CA (2016) Economic influences on trapper participation and per capita harvest of muskrat. Wildl Soc Bull 40:548–553. https://doi.org/10.1002/wsb.696
Allen ML, Roberts NM, Van Deelen TR (2018) Hunter selection for larger and older male bobcats affects annual harvest demography. Roy Soc Open Sci 5:180668. https://doi.org/10.1098/rsos.180668
Allen ML, Roberts NM, Bauder JM (2020) Relationships of catch-per-unit-effort metrics with abundance vary depending on sampling method and population trajectory. PLoS ONE 15:0233444. https://doi.org/10.1371/journal.pone.0233444
Allison AB, Harbison CE, Pagan I, Stucker KM, Kaelber JT, Brown JD, Ruder MG, Keel MK, Dubovi EJ, Holmes EC, Parrish CR (2012) Role of multiple hosts in the cross-species transmission and emergence of a pandemic parvovirus. J Virology 86:865–872. https://doi.org/10.1128/JVI.06187-11
Allison AB, Kohler DJ, Fox KA, Brown JD, Gerhold RW, Shearn-Bochsler VI, Dubovi EJ, Parrish CR, Holmes EC (2013) Frequent cross-species transmission of parvoviruses among diverse carnivore hosts. J Virol 87:2342–2347. https://doi.org/10.1128/JVI.02428-12
Allison TD, Diffendorfer JE, Baerwald EF, Beston JA, Drake D, Hale AM, Hein CD, Huso MM, Loss SR, Lovich JE, Strickland MD (2019) Impacts to wildlife of wind energy siting and operation in the United States. Issues Ecol 21(1):2–18
Allred BW, Smith WK, Twidwell D, Haggerty JH, Running SW, Naugle DE, Fuhlendorf SD (2015) Ecosystem services lost to oil and gas in North America. Science 348(6233):401–402. https://doi.org/10.1126/science.aaa4785
Andelt W (1992) Effectiveness of livestock guarding dogs for reducing predation on domestic sheep. Wildl Soc Bull 20:55–62
Andersen ML, Bennett DE, Holbrook JD (2021) Burrow webs: clawing the surface of interactions with burrows excavated by American badgers. Ecol Evol 11(17):11559–11568. https://doi.org/10.1002/ece3.7962
Anis E, Needle DB, Stevens B, Yan L, Wilkes RP (2020) Genetic characteristics of canine distemper viruses circulating in wildlife in the United States. J Zoo Wildl Med 50:790–797. https://doi.org/10.1638/2019-0052
Arizona Administrative Code R12-4-303 (2021) Unlawful devices, methods, and ammunition
Association of Fish and Wildlife Agencies (2015) National fur harvest database, 1970–2018. https://www.fishwildlife.org/download_file/view/2896/1213
Atwood TC, Fry TL, Leland BR (2011) Partitioning of anthropogenic watering sites by desert carnivores. J Wildl Manage 75:1609–1615. https://doi.org/10.1002/jwmg.225
Aubry KB, Statham MJ, Sacks BN, Perrine JD, Wisely SM (2009) Phylogeography of the North American red fox: vicariance in Pleistocene forest refugia. Mol Ecol 18(12):2668–2686. https://doi.org/10.1111/j.1365-294X.2009.04222.x
Azevedo FCC, Lester V, Gorsuch W, Larivière S, Wirsing AJ, Murray DL (2006) Dietary breadth and overlap among five sympatric prairie carnivores. J Zool 269:127–135. https://doi.org/10.1111/j.1469-7998.2006.00075.x
Ballard WB, Lutz D, Keegan TW, Carpenter LH, De Vos J (2001) Deer-predator relationships: a review of recent North American studies with emphasis on mule and black-tailed deer. Wildl Soc Bull 29:99–115
Bartel RA, Brunson MW (2003) Effects of Utah’s coyote bounty program on harvester behavior. Wildl Soc Bull 31:736–743. https://doi.org/10.2307/3784593
Bauder JM, Allen ML, Ahlers AA, Benson TJ, Miller CA, Stodola KW (2020a) Identifying and controlling for variation in canid harvest data. J Wildl Manage 84:1234–1245. https://doi.org/10.1002/jwmg.21919
Bauder JM, Stodola KW, Benson TJ, Miller CA, Allen ML (2020b) Raccoon pelt price and trapper harvest relationships are temporally inconsistent. J Wildl Manage 84:1601–1610. https://doi.org/10.1002/jwmg.21928
Beineke A, Baumgärtner W, Wohlsein P (2015) Cross-species transmission of canine distemper virus-an update. One Health 1:49–59. https://doi.org/10.1016/j.onehlt.2015.09.002
Belant JL, Schipper J, Conroy J (2009) The conservation status of small carnivores in the Americas. Small Carniv Conserv 41:3–8
Belant J, Biggins D, Garelle D et al (2015) Mustela nigripes. The IUCN Red List of Threatened Species 2015: e.T14020A45200314. https://doi.org/10.2305/IUCN.UK.2015-4.RLTS.T14020A45200314.en.8235
Bell D, Roberton S, Hunter PR (2004) Animal origins of SARS coronavirus: possible links with the international trade in small carnivores. Phil Trans R Soc B: Biol Sci 359:1107–1114. https://doi.org/10.1098/rstb.2004.1492
Berger KM, Conner MM (2008) Recolonizing wolves and mesopredator suppression of coyotes: impacts on pronghorn population dynamics. Ecol Appl 8(3):599–612
Berry B, Schooley RL, Ward MP (2017) Landscape context affects use of restored grasslands by mammals in a dynamic agroecosystem. Am Midland Nat 177:165–182. https://doi.org/10.1674/0003-0031-177.2.165
Black KL, Petty SK, Radeloff VC, Pauli JN (2018) The Great Lakes Region is a melting pot for vicariant red fox (Vulpes vulpes) populations. J Mamm 99(5):1229–1236. https://doi.org/10.1093/jmammal/gyy096
Blackburn AM, Anderson CJ, Veals AM, Tewes ME, Wester DB, Young JH, DeYoung RW, Perotto-Baldivieso HL (2021) Landscape patterns of ocelot–vehicle collision sites. Landscape Ecol 36:497–511. https://doi.org/10.1007/s10980-020-01153-y
Blejwas KM, Sacks BN, Jaeger MM, McCullough DR (2002) The effectiveness of selective removal of breeding coyotes in reducing sheep predation. J Wildl Manage 66:451. https://doi.org/10.2307/3803178
Blythe EM, Boyce MS (2020) Trappings of success: predator removal for duck nest survival in Alberta Parklands. Diversity 12:119. https://doi.org/10.3390/d12030119
Booth-Binczik SD, Bradley RD, Thompson CW et al (2013) Food habits of ocelots and potential for competition with bobcats in southern Texas. Southwest Nat 58:403–410. https://doi.org/10.1894/0038-4909-58.4.403
Brickner KM, Grenier MB, Crosier AE, Pauli JN (2014) Foraging plasticity in a highly specialized carnivore, the endangered black-footed ferret. Biol Conserv 169:1–5. https://doi.org/10.1016/j.biocon.2013.10.010
Bromen NA, French JT, Walker JW, Tomeček JM (2019) Spatial relationships between livestock guardian dogs and mesocarnivores in central Texas. Human-Wildl Interact 13:29–41. https://doi.org/10.26076/0d01-xz26
Bromley C, Gese EM (2001) Surgical sterilization as a method of reducing coyote predation on domestic sheep. J Wildl Manage 65:510–519. https://doi.org/10.2307/3803104
Brown DE, Conover MR (2011) Effects of large-scale removal of coyotes on pronghorn and mule deer productivity and abundance. J Wildl Manage 75:876–882. https://doi.org/10.1002/jwmg.126
Butler AR, Bly KLS, Harris H, Inman RM, Moehrenschlager A, Schwalm D, Jachowski DS (2019) Winter movement behavior by swift foxes (Vulpes velox) at the northern edge of their range. Can J Zool 97:922–930. https://doi.org/10.1139/cjz-2018-0272
Butler AR, Bly KL, Harris H, Inman RM, Moehrenschlager A, Schwalm D, Jachowski DS (2020) Home range size and resource use by swift foxes in northeastern Montana. J Mamm 101(3):684–696. https://doi.org/10.1093/jmammal/gyaa030
Byerly PA, Lonsinger RC, Gese EM, Kozlowski AJ, Waits LP (2018) Resource partitioning between kit foxes (Vulpes macrotis) and coyotes (Canis latrans): a comparison of historical and contemporary dietary overlap. Canadian J Zool 96:497–504. https://doi.org/10.1139/cjz-2017-0246
Cain SA, Kadlec JA, Allen DL, Cooley RA, Hornocker MH, Wagner FH (1972) Predator control–1971: report to the President’s Council on Environmental Quality and the US Department of the Interior by the Advisory Committee of Predator Control. University of Michigan Press, Ann Arbor, MI, p 230
Carricondo-Sanchez D, Odden M, Kulkarni A, Vanak AT (2019) Scale-dependent strategies for coexistence of mesocarnivores in human-dominated landscapes. Biotropica 51(5):781–791. https://doi.org/10.1111/btp.12705
Chambers JC, Maestas JD, Pyke DA, Boyd CS, Pellant M, Wuenschel A (2017) Using resilience and resistance concepts to manage persistent threats to sagebrush ecosystems and greater sage-grouse. Rangeland Ecol Manage 70(2):149–164. https://doi.org/10.2737/RMRS-GTR-356
Chipault JG, Biggins DE, Detling JK, Long DH, Reich RM (2012) Fine-scale habitat use of reintroduced black-footed ferrets on prairie dog colonies in New Mexico. Western N Am Nat 72:216–227. https://doi.org/10.3398/064.072.0211
Cherry MJ, Morgan KE, Rutledge BT, Conner LM, Warren RJ (2016) Can coyote predation risk induce reproduction suppression in white‐tailed deer? Ecosphere 7:e01481
Chronert JM, Jenks JA, Roddy DE, Wild MA, Powers JG (2007) Effects of sarcoptic mange on coyotes at Wind Cave National Park. J Wildl Manage 71:1987–1992. https://doi.org/10.2193/2006-225
Clark W, Andrew RD (1982) Review of population indices applied in furbearer management. In: Sanderson GC (ed) Proceedings of the Midwest furbearer management symposium. North Central Section, Central Mountains and Plains Section, and Kansas Chapter, The Wildlife Society, 7–8 Dec 1981, Wichita, KS, USA, pp 11–22
Clark HO, Warrick GD, Cypher BL, Kelly PA, Williams DF, Grubbs DE (2005) Competitive interactions between endangered kit foxes and nonnative red foxes. Western N Am Nat 65:153–163
Convention on International Trade in Endangered Species of Wild Fauna and Flora [CITES] (2021) Lynx rufus. In: CITES 2021 Appendix II. https://cites.org/eng
Crawford-Miksza LK, Wadford DA, Schnurr DP (1999) Molecular epidemiology of enzootic rabies in California. J Clinic Virol 14:207–219. https://doi.org/10.1016/S1386-6532(99)00054-2
Crimmins SM, Walleser LR, Hertel DR, McKann PC, Rohweder JJ, Thogmartin WE (2016) Relating mesocarnivore relative abundance to anthropogenic land-use with a hierarchical spatial count model. Ecography 39(6):524–532. https://doi.org/10.1111/ecog.01179
Crooks KR, Soulé ME (1999) Mesopredator release and avifaunal extinctions in a fragmented system. Nature 400:563–566. https://doi.org/10.1038/23028
Cuarón AD, González-Maya JF, Helgen K et al (2016a) Mephitis macroura. The IUCN Red List of Threatened Species 2016a: e.T41634A45211135. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41634A45211135.en. 8235
Cuarón AD, Helgen K, Reid F et al (2016b) Nasua narica. The IUCN Red List of Threatened Species 2016b: e.T41683A45216060. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41683A45216060.en, https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41683A45216060.en
Cuarón AD, Helgen K, Reid F (2016c) Spilogale gracilis. The IUCN Red List of Threatened Species 2016c: e.T136797A45221721. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T136797A45221721.en
Cypher B, List R (2014) Vulpes macrotis. The IUCN Red List of Threatened Species 2014: e.T41587A62259374. https://doi.org/10.2305/IUCN.UK.2014-3.RLTS.T41587A62259374.en
Cypher BL, Spencer KA (1998) Competitive interactions between coyotes and San Joaquin kit foxes. J Mamm 79:204–214. https://doi.org/10.2307/1382855
Cypher BL, Scrivner JH (1992) Coyote control to protect endangered San Joaquin kit foxes at the Naval Petroleum Reserves, California. In: Proceedings of the 15th vert pest conference, pp 42–47
Cypher BL, Rudd JL, Westall TL, Woods LW, Stephenson N, Foley JE, Richardson D, Clifford DL (2017) Sarcoptic mange in endangered kit foxes (Vulpes macrotis mutica): case histories, diagnoses, and implications for conservation. J Wildl Dis 53:1–8. https://doi.org/10.7589/2016-05-098
Deem SL, Spelman LH, Yates RA, Montali RJ (2000) Canine distemper in terrestrial carnivores: a review. J Zoo Wildl Med 31:441–451. https://doi.org/10.1638/1042-7260(2000)031[0441:CDITCA]2.0.CO;2
DeVink J, Berezanski D, Imrie D (2011) Comments on Brodie and Post: Harvest effort: the missing covariate in analyses of furbearer harvest data. Pop Ecol 53:261–262. https://doi.org/10.1007/s10144-010-0241-6
DeYoung RW, Zamorano A, Mesenbrink BT, Campbell TA, Leland BR, Moore GM, Honeycutt RL, Root JJ (2009) Landscape-genetic analysis of population structure in the Texas gray fox oral rabies vaccination zone. J Wildl Manage 73:1292–1299. https://doi.org/10.2193/2008-336
Docken NR (2011) Effects of block predator management on duck and pheasant nest success in eastern South Dakota. M.S. thesis, South Dakota State University, Brookings, SD, 92pp
Donadio E, Buskirk SW (2006) Diet, morphology, and interspecific killing in Carnivora. Am Nat 167:524–536. https://doi.org/10.1086/501033
Dragoo JW, Honeycutt RL (1999) Eastern hog-nosed skunk/Conepatus leuconotus. In: Wilson DE, Ruff S (eds) The smithsonian book of North American mammals. Smithsonian Institution Press. Washington, D.C., pp 190–191
Dragoo JW, Sheffield SR (2009) Conepatus leuconotus (Carnivora: Mephitidae). Mamm Spec 827:1–8
Dragoo JW, Matthes DK, Aragon A, Hass CC, Yates TL (2004) Identification of skunk species submitted for rabies testing in the desert southwest. J Wildl Dis 40:371–376. https://doi.org/10.7589/0090-3558-40.2.371
du Toit JT, Cross PC, Valeix M (2017) Managing the livestock–wildlife interface on rangelands. In: Briske DD (ed) Rangeland systems, pp 395–425
Dunstone N (1993) The mink. UK, T. & A. D. Poyser
Eklund A, López-Bao JV, Tourani M, Chapron G, Frank J (2017) Limited evidence on the effectiveness of interventions to reduce livestock predation by large carnivores. Sci Rep 7:1–9. https://doi.org/10.1038/s41598-017-02323-w
Eldridge DJ, Whitford WG (2009) Badger (Taxidea taxus) disturbances increase soil heterogeneity in a degraded shrub-steppe ecosystem. J Arid Environ 73:66–73. https://doi.org/10.1016/j.jaridenv.2008.09.004
Ellis EC, Klein Goldewijk K, Siebert S, Lightman D, Ramankutty N (2010) Anthropogenic transformation of the biomes, 1700–2000. Global Ecol Biogeog 19(5):589–606. https://doi.org/10.1111/j.1466-8238.2010.00540.x
Elsken-Lacy P, Wilson AM, Heidt GA, Peck JH (1999) Arkansas gray fox fur price-harvest model revisited. J Arkansas Acad Sci 53:50–54
Evans GD, Pearson EW (1980) Federal coyote control methods used in the western United States, 1971–77. Wildl Soc Bull 1:34–39
Faulkner CE (1971) The legal status of wildcats in the United States. In: Jorgensen RM, Mech LD (ed) Proceedings of a symposium on the native cats of North America, their status and management. 36th American wildlife and natural resources conference. U.S. Fish and Wildlife Service, Twin Cities, Minnesota, pp 124–125
Feldman JW (2007) Public opinion, the Leopold Report, and the reform of federal predator control policy. Human-Wildl Confl 1:112–124. https://doi.org/10.26077/eh4w-3894
Fiehler CM, Cypher BL, Saslaw LR (2017) Effects of oil and gas development on vertebrate community composition in the southern San Joaquin Valley, California. Global Ecol Conserv 9:131–141. https://doi.org/10.1016/j.gecco.2017.01.001
Frey SN, Majors S, Conover MR, Messmer TA, Mitchell DL (2003) Effect of predator control on ring-necked pheasant populations. Wildl Soc Bull 31:727–735. https://doi.org/10.2307/3784592
Gese EM, Schultz RD, Rongstad OJ, Andersen DE (1991) Prevalence of antibodies against canine parvovirus and canine distemper virus in wild coyotes in southeastern Colorado. J Wildl Dis 27:320–323. https://doi.org/10.7589/0090-3558-27.2.320
Gese EM, Schultz RD, Johnson MR, Williams ES, Crabtree RL, Ruff RL (1997) Serological survey for diseases in free-ranging coyotes (Canis latrans) in Yellowstone National Park, Wyoming. J Wildl Dis 33:47–56. https://doi.org/10.7589/0090-3558-33.1.47
Gese EM, Karki SM, Klavetter ML, Schauster ER, Kitchen AM (2004) Serologic survey for canine infectious diseases among sympatric swift foxes (Vulpes velox) and coyotes (Canis latrans) in Southeastern Colorado. J Wildl Dis 40:741–748. https://doi.org/10.7589/0090-3558-40.4.741
Goldman HV (2021) Nearly extirpated by plague and distemper in the 1980s, black-footed ferrets now vaccinated for COVID-19. Small Carnivore Conserv 59. https://smallcarnivoreconservation.com/index.php/sccg/article/view/3463
Gompper ME (2002) Top carnivores in the suburbs? Ecological and conservation issues raised by colonization of north eastern North America by coyotes. Bioscience 52:185. https://doi.org/10.1641/0006-3568(2002)052[0185:TCITSE]2.0.CO;2
Gompper ME, Hackett HM (2005) The long-term, range-wide decline of a once common carnivore: the eastern spotted skunk (Spilogale putorius). Anim Conserv 8(2):195–201. https://doi.org/10.1017/S1367943005001964
Gompper M, Jachowski D (2016) Spilogale putorius. The IUCN Red List of Threatened Species 2016: e.T41636A45211474. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41636A45211474.en
Grassel SM, Rachlow JL (2018) When generalists behave as specialists: local specialization by American badgers (Taxidea taxus). Canadian J Zool 96:592–599. https://doi.org/10.1139/cjz-2017-0125
Grassel SM, Rachlow JL, Williams CJ (2015) Spatial interactions between sympatric carnivores: asymmetric avoidance of an intraguild predator. Ecol Evol 5:2762–2773. https://doi.org/10.1002/ece3.1561
Greenspan E, Anile S, Nielsen CK (2020) Density of wild felids in Sonora, Mexico: a comparison of spatially explicit capture-recapture methods. Eur J Wildl Res 66:1–12. https://doi.org/10.1007/s10344-020-01401-1
Greenwood RJ, Sargeant AB, Piehl JL et al (1999) Foods and foraging of prairie striped skunks during the avian nesting season. Wildl Soc Bull 27:823–832
Guthery FS, Beasom SL (1977) Responses of game and nongame wildlife to predator control in south Texas. J Range Manage Arch 30:404–409
Haines AM, Tewes ME, Laack LL (2005) Survival and sources of mortality in ocelots. J Wildl Manage 69:255–263. https://doi.org/10.2193/0022-541X(2005)069%3c0255:SASOMI%3e2.0.CO;2
Haines AM, Janecka JE, Tewes ME, Grassman LI, Morton P (2006) The importance of private lands for ocelot Leopardus pardalis conservation in the United States. Oryx 40:90–94. https://doi.org/10.1017/S0030605306000044
Hall ER, Dalquest WW (1963) The mammals of Veracruz. Univ Kansas Publ Mus Nat Hist 14:165–362
Harrington JL, Conover MR (2007) Does removing coyotes for livestock protection benefit free-ranging ungulates? J Wildl Manage 71:1555–1560
Hass CC, Dragoo JW (2006) Rabies in hooded and striped skunks in Arizona. J Wildl Dis 42:825–829
Hatfield BE, Runcie JM, Siemion EA, Quinn CB, Stephenson TR (2021) New detections extend the known range of the state-threatened Sierra Nevada red fox. Calif Fish Game 107:438–443
Hedberg GE, Dierenfeld ES, Rogers QR (2007) Taurine and zoo felids: considerations of dietary and biological tissue concentrations. Zoo Biol 26:517–531
Heithaus MR (2001) Habitat selection by predators and prey in communities with asymmetrical intraguild. Oikos 92:542–554
Helgen K (2016) Conepatus leuconotus. The IUCN Red List of Threatened Species 2016: e.T41632A45210809. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41632A45210809.en
Helgen K, Reid F (2016a) Mephitis mephitis. The IUCN Red List of Threatened Species 2016a: e.T41635A45211301. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41635A45211301.en. https://doi.org/10.1007/978-1-4615-6422-5_26
Helgen K, Reid F (2016b) Taxidea taxus. The IUCN Red List of Threatened Species 2016b: e.T41663A45215410. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41663A45215410.en
Helgen K, Reid F (2016c) Mustela frenata. The IUCN Red List of Threatened Species 2016c: e.T41654A45213820. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41654A45213820.en
Henke S, Bryant F (1999) Effects of coyote removal on the faunal community in western Texas. J Wildl Manage 63:1066–1081
Hiller TL, Etter DR, Belant JL, Tyre AJ (2011) Factors affecting harvests of fishers and American martens in northern Michigan. J Wildl Manage 75:1399–1405
Hines TD, Case RM (1991) Diet, home range, movements, and activity periods of swift fox in Nebraska. Prairie Nat 23:131–138
Hirsch BT, Prange S, Hauver SA, Gehrt SD (2013) Raccoon social networks and the potential for disease transmission. PLoS ONE 8:e75830
Hody JW, Kays R (2018) Mapping the expansion of coyotes (Canis latrans) across North and Central America. Zookeys 81–97.https://doi.org/10.3897/zookeys.759.15149
Hoffmann M, Sillero-Zubiri C (2016) Vulpes vulpes. The IUCN Red List of Threatened Species 2016: e.T23062A46190249. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T23062A46190249.en. IUCN Red List Threat Species 2016 8235:e.T23062A4
Holbrook JD, Arkle RS, Rachlow JL, Vierling KT, Pilliod DS, Wiest MM (2016) Occupancy and abundance of predator and prey: implications of the fire-cheatgrass cycle in sagebrush ecosystems. Ecosphere 7(6):e01307
Holland AM, Schauber EM, Nielsen CK, Hellgren EC (2019) River otter and mink occupancy dynamics in riparian systems. J Wildl Manage 83:1552–1564. https://doi.org/10.1002/jwmg.21745
Holt RD, Huxel GR (2007) Alternative prey and the dynamics of intraguild predation: theoretical perspectives. Ecology 88:2706–2712
Holt RD, Polis GA (1997) A theoretical framework for intraguild predation. Am Nat 149:745–764
Horne JS, Haines AM, Tewes ME, Laack LL (2009) Habitat partitioning by sympatric ocelots and bobcats: implications for recovery of ocelots in southern Texas. Southwest Nat 54:119–126. https://doi.org/10.1894/PS-49.1
Hubert GF (1982) History of Midwestern furbearer management and a look to the future. In: Sanderson GC (ed) Midwest furbearer management: proceedings of the symposium of the 43rd Midwest fish and wildlife conference. Kansas Chapter of the Wildlife Society, Wichita, KS, pp 175–190
Hueffer K, Parker JSL, Weichert WS, Geisel RE, Sgro J-Y, Parrish CR (2003) The natural host range shift and subsequent evolution of canine parvovirus resulted from virus-specific binding to the canine transferrin receptor. J Virol 77:1718–1726. https://doi.org/10.1128/jvi.77.3.1718-1726.2003
Jachowski DS, Edelman AJ (2021) Advancing small carnivore research and conservation: the Eastern Spotted Skunk Cooperative Study Group model. Southeast Nat 20:1–12. https://doi.org/10.1656/058.020.0sp1102
Jackson VL, Laack LL, Zimmerman EG (2005) Landscape metrics associated with habitat use by ocelots in south Texas. J Wildl Manage 69:733–738
Janečka JE, Tewes ME, Davis IA, Haines AM, Caso A, Blankenship TL, Honeycutt RL (2016) Genetic differences in the response to landscape fragmentation by a habitat generalist, the bobcat, and a habitat specialist, the ocelot. Conserv Genet 17(5):1093–1108. https://doi.org/10.1007/s10592-016-0846-1
Kabir E, Kumar P, Kumar S, Adelodun AA, Kim KH (2018) Solar energy: potential and future prospects. Renew Sustain Energy Rev 82:894–900. https://doi.org/10.1016/j.rser.2017.09.094
Kamler JF, Ballard WB (2002) A review of native and nonnative red foxes in North America. Wildl Soc Bull 30:370–379. https://doi.org/10.2307/3784493
Kamler JF, Ballard WB, Gilliland RL, Lemons PR, Mote K (2003a) Impacts of coyotes on swift foxes in northwestern Texas. J Wildl Manage 67:317–323. https://doi.org/10.2307/3802773
Kamler JF, Ballard WB, Helliker BR, Stiver S (2003b) Range expansion of raccoons in western Utah and central Nevada. West North Am Nat 63:406–408
Kapfer PM, Potts KB (2012) Socioeconomic and ecological correlates of bobcat harvest in Minnesota. J Wildl Manage 76:237–242. https://doi.org/10.2307/41418264
Karki SM, Gese EM, Klavetter ML (2007) Effects of coyote population reduction on swift fox demographics in southeastern Colorado. J Wildl Manage 71:2707–2718. https://doi.org/10.2193/2006-275
Kasprowicz AE, Statham MJ, Sacks BN (2016) The fate of the other red coat: remnants of colonial British red foxes in the Eastern United States. J Mamm 97:298–309. https://doi.org/10.1093/jmammal/gyv179
Kays R (2018) Canis latrans (errata version published in 2020). The IUCN Red List of Threatened Species 2018: e.T3745A163508579. https://doi.org/10.2305/IUCN.UK.2018-2.RLTS.T3745A163508579.en
Kelly M, Morin D, López-González CA (2016). Lynx rufus. The IUCN Red List of Threatened Species 2016: e.T12521A50655874. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T12521A50655874.en
Kilgo JC, Shaw CE, Vukovich M, Conroy MJ, Ruth C (2017) Reproductive characteristics of a coyote population before and during exploitation. J Wildl Manage 81:1386–1393. https://doi.org/10.1002/jwmg.21329
Kimbrell T, Holt RD, Lundberg P (2007) The influence of vigilance on intraguild predation. J Theor Biol 249:218–234. https://doi.org/10.1016/j.jtbi.2007.07.031
Kinka D, Young JK (2018) A livestock guardian dog by any other name: similar response to wolves across livestock guardian dog breeds. Rangeland Ecol Manage 71(4):509–517. https://doi.org/10.1016/j.rama.2018.03.004
Kitchen AM, Gese EM, Schauster ER (1999) Resource partitioning between coyotes and swift foxes: space, time, and diet. Can J Zool 77:1645–1656. https://doi.org/10.1139/cjz-77-10-1645
Kitchen AM, Gese EM, Schauster ER (2000) Changes in coyote activity patterns due to reduced exposure to human persecution. Can J Zool 78:853–857. https://doi.org/10.1139/cjz-78-5-853
Knowles CJ, Proctor JD, Forrest SC (2002) Black-tailed prairie dog abundance and distribution in the great plains based on historic and contemporary information. Great Plains Res 12:219–254
Knowlton FF, Gese EM, Jaeger MM (1999) Coyote depredation control: an interface between biology and management. Soc Range Manage 52:398–412. https://doi.org/10.2307/4003765
Kuo YH, Vanderzwan SL, Kasprowicz AE, Sacks BN (2019) Using ancestry-informative SNPs to quantify introgression of European alleles into North American red foxes. J Heredity 110(7):782–792. https://doi.org/10.1093/jhered/esz053
Kuzmin IV, Shi M, Orciari LA, Yager PA, Velasco-Villa A, Kuzmina NA, Streicker DG, Bergman DG, Rupprecht CE (2012) Molecular inferences suggest multiple host shifts of rabies viruses from bats to mesocarnivores in Arizona during 2001–2009. PLoS Pathogens 8.https://doi.org/10.1371/journal.ppat.1002786
Larson RN, Morin DJ, Wierzbowska IA, Crooks KR (2015) Food habits of coyotes, gray foxes, and bobcats in a coastal Southern California urban landscape. West North Am Nat 75:339–347. https://doi.org/10.3398/064.075.0311
Larson S, McGranahan DA, Timm RM (2019) The Marin County livestock protection program: 15 years in review. Human-Wildl Interact 13:63–78. https://doi.org/10.5070/V427110695
Lawrence JS, Silvy NJ (1995) Effect of predator control on reproductive success and hen survival of Attwater’s prairie-chicken. In: Proceedings Southeast Association of Fish Wildlife Agencies, vol 49, pp 275–282
Leslie MJ, Messenger S, Rohde RE, Smith J, Cheshier R, Hanlon C, Rupprecht CE (2006) Bat-associated rabies virus in skunks. Emerg Infect Dis 12:1274–1277.https://doi.org/10.3201/eid1208.051526
Lesmeister DB, Nielsen CK, Schauber EM, Hellgren EC (2015) Spatial and temporal structure of a mesocarnivore guild in midwestern north America. Wild Mon 191:1–61. https://doi.org/10.1002/wmon.1015
Lombardi JV, MacKenzie DI, Tewes ME et al (2020a) Co-occurrence of bobcats, coyotes, and ocelots in Texas. Ecol Evol 10:4903–4917. https://doi.org/10.1002/ece3.6242
Lombardi JV, Tewes ME, Perotto-Baldivieso HL, Mata JM, Campbell TA (2020b) Spatial structure of woody cover affects habitat use patterns of ocelots in Texas. Mamm Res 65:555–563. https://doi.org/10.1007/s13364-020-00501-2
Long JL (2003) Introduced mammals of the world: their history, distribution and influence. CABI Publishing, Wallingford, UK, xxi + 589 pp
Lonsinger RC, Gese EM, Bailey LL, Waits LP (2017) The roles of habitat and intraguild predation by coyotes on the spatial dynamics of kit foxes. Ecosphere 8:e01749. https://doi.org/10.1002/ecs2.1749
Lonsinger RC, Kluever BM, Hall LK, Larsen RT, Gese EM, Waits LP, Knight RN (2020) Conservation of kit foxes in the Great Basin Desert: Review and recommendations. J Fish Wildl Mgmt 11:679–698
Łopucki R, Klich D, Gielarek S (2017) Do terrestrial animals avoid areas close to turbines in functioning wind farms in agricultural landscapes?. Environ Monit Assess 189:1–1.
Lourenço R, Penteriani V, Rabaça JE, Korpimäki E (2014) Lethal interactions among vertebrate top predators: a review of concepts, assumptions and terminology. Biol Rev 89:270–283. https://doi.org/10.1111/brv.12054
Lovich JE, Ennen JR (2011) Wildlife conservation and solar energy development in the desert southwest, United States. Bioscience 61:982–992. https://doi.org/10.1525/bio.2011.61.12.8
Lovich JE, Ennen JR, Madrak S, Grover B (2011) Turtles, culverts, and alternative energy development: an unreported but potentially significant mortality threat to the desert tortoise (Gopherus agassizii). Chelonian Conserv Biol 10:124–129. https://doi.org/10.2744/CCB-0864.1
Lyons EK, Frost J, Rollins D, Scott C (2009) An evaluation of short-term mesocarnivore control for increasing hatch rate in Northern Bobwhites. Nat Quail Symp Proc 6:447–455
Ma X, Monroe BP, Cleaton JP, Orciari LA, Gigante CM, Kirby JD, Chipman RB, Fehlner-Gardiner C, Cedillo VG, Petersen BW, Olson V, Wallace RM (2020) Rabies surveillance in the United States during 2018. J Am Vet Med Assoc 256:195–208. https://doi.org/10.2460/javma.256.2.195
Mahoney PJ, Young JK, Hersey KR, Larsen RT, McMillan BR, Stoner DC (2018) Spatial processes decouple management from objectives in a heterogeneous landscape: predator control as a case study. Ecol Applic 28(3):786–797. https://doi.org/10.1002/eap.1686
Marneweck C, Butler AR, Gigliotti LC, Harris SN, Jensen AJ, Muthersbaugh M, Jachowski DS (2021) Shining the spotlight on small mammalian carnivores: global status and threats. Biol Conserv 255:109005. https://doi.org/10.1016/j.biocon.2021.109005
Marneweck CJ, Allen BL, Butler AR, Do Linh San E, Harris SN, Jensen AJ, Jachowski DS (2022) Middle-out ecology: small carnivores as sentinels of global change. Mamm Rev. https://doi.org/10.1111/mam.12300
McCue PM, O’Farrell TP (1988) Serological survey for selected diseases in the endangered San Joaquin kit fox (Vulpes macrotis mutica). J Wildl Dis 24:274–281. https://doi.org/10.7589/0090-3558-24.2.274
McDonald RA, Abramov AV, Stubbe M et al (2019) Mustela nivalis (amended version of 2016 assessment). The IUCN Red List of Threatened Species 2019: e.T70207409A147993366. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T70207409A147993366.en
McGee Jr HF (1987) The use of furbearers by native North Americans after 1500. In: Novak M, Baker JA, Obbard M, Malloch B (eds) Wild furbearer management and conservation in North America. Ontario Ministry of Natural Resources, Toronto, Ontario, pp 13–20
McKinney T, Smith TW (2007) Diets of sympatric bobcats and coyotes during years of varying rainfall in central Arizona. West North Am Nat 67:8–15
Melchior HR, Johnson NF, Phelps JS (1987) Wild furbearer management in the western United States and Alaska In: Novak M, Baker JA, Obbard ME, Malloch B (eds) Wild furbearer management and conservation in North America. Ontario Ministry of Natural Resources, Toronto, Ontario, pp 1117–1128
Melquist WE, Polechla PJ, Toweill D (2003) River otter, Lontra canadensis. In: Feldhamer GA, Thompson BC, Chapman JA (eds) Wild mammals of North America: biology, management, and conservation. Johns Hopkins University Press, Baltimore, Md, pp 708–734
Miller JE (2007) Evolution of the field of wildlife damage management in the United States and future challenges. Human-Wildl Confl 1:13–20. https://doi.org/10.26077/f1df-nm52
Miller DS, Covell DF, McLean RG, Adrian WJ, Niezgoda M, Gustafson JM, Rongstad OJ, Schultz RD, Kirk LJ, Quan TJ (2000) Serologic Survey for selected infectious disease agents in swift and kit foxes from the western United States. J Wildl Dis 36:798–805. https://doi.org/10.7589/0090-3558-36.4.798
Miller TE, Burns JH, Munguia P, Walters EL, Kneitel M, Richards PM, Mouquet N, Buckley HL (2005) A critical review of twenty years’ use of the resource-ratio theory. Am Nat 165:439–448. https://doi.org/10.1086/428681
Minta SC, Minta KA, Lott DF (1992) Hunting associations between badgers (Taxidea taxus) and coyotes (Canis latrans). J Mamm 73(4):814–820. https://doi.org/10.1139/cjz-2017-0234
Mitchell BR, Jaeger MM, Barrett RH (2004) Coyote depredation management: current methods and research needs. Wildl Soc Bull 32:1209–1218. https://doi.org/10.2193/0091-7648(2004)032[1209:CDMCMA]2.0.CO;2
Moehrenschlager A, Sovada M (2016) Vulpes velox. The IUCN Red List of Threatened Species 2016: e.T23059A57629306. https://doi.org/10.2305/IUCN.UK.2016-3.RLTS.T23059A57629306.en
Montecino-Latorre D, Cypher BL, Rudd JL, Clifford DL, Mazet JAK, Foley JE (2019) Assessing the role of dens in the spread, establishment and persistence of sarcoptic mange in an endangered canid. Epidemics 27:28–40. https://doi.org/10.1016/j.epidem.2019.01.001
Moreira-Arce D, Vergara PM, Boutin S, Simonetti JA, Briceño C, Acosta-Jamett G (2015) Native forest replacement by exotic plantations triggers changes in prey selection of mesocarnivores. Biol Conserv 192:258–267. https://doi.org/10.1016/j.biocon.2015.09.015
Mosley JC, Roeder BL, Frost RA, Wells SL, McNew LB, Clark PE (2020) Mitigating human conflicts with livestock guardian dogs in extensive sheep grazing systems. Range Ecol Manage 73:724–732. https://doi.org/10.1016/j.rama.2020.04.009
Mueller MA, Drake D, Allen ML (2018) Coexistence of coyotes (Canis latrans) and red foxes (Vulpes vulpes) in an urban landscape. PLoS ONE 13(1):e0190971. https://doi.org/10.1371/journal.pone.0190971
Neale JCC, Sacks BN (2001) Food habits and space use of gray foxes in relation to sympatric coyotes and bobcats. Can J Zool 79:1794–1800. https://doi.org/10.1139/cjz-79-10-1794
Nelson JL, Cypher BL, Bjurlin CD, Creel S (2007) Effects of habitat on competition between kit foxes and coyotes. J Wildl Manage 71:1467–1475. https://doi.org/10.2193/2006-234
Newbury RK, Hodges KE (2018) Regional differences in winter diets of bobcats in their northern range. Ecol Evol 8:11100–11110. https://doi.org/10.1002/ece3.4576
New Mexico Senate Bill 76 (2019) Prohibit coyote killing contests
Nicholson KL, Ballard WB, Mcgee BK, Surles J, Kamler JF, Lemons PR (2006) Swift fox use of black-tailed prairie dog towns in northwest Texas. J Wildl Manage 70:1659–1666
Nicholson KL, Ballard WB, McGee BK, Whitlaw HA (2007) Dispersal and extraterritorial movements of swift foxes (Vulpes velox) in Northwestern Texas. West North Am Nat 67:102–108. https://doi.org/10.3398/1527-0904(2007)67[102:DAEMOS]2.0.CO;2
Niedringhaus KD, Brown JD, Sweeley KM, Yabsley MJ (2019) A review of sarcoptic mange in North American wildlife. Internat J Parasit: Parasites Wildl 9:285–297. https://doi.org/10.1016/j.ijppaw.2019.06.003
Nimmervoll H, Hoby S, Robert N, Lommano E, Welle M, Ryser-Degiorgis MP (2013) Pathology of sarcoptic mange in red foxes (Vulpes vulpes): macroscopic and histologic characterization of three disease stages. J Wildl Disease 49:91–102. https://doi.org/10.7589/2010-11-316
Obbard ME, Jones JG, Newman R, Booth A, Satterthwaite AJ, Linscombe G (1987) Furbearer harvests in North America. In: Novak M, Baker JA, Obbard ME, Malloch B (eds) Wild furbearer management and conservation in North America. Ontario Ministry of Natural Resources, Toronto, Ontario, pp 1007–1034
Olson DM, Dinerstein E, Wikramanayake ED, Burgess ND, Powell GV, Underwood EC, D'amico JA, Itoua I, Strand HE, Morrison JC, Loucks CJ (2001) Terrestrial ecoregions of the world: A new map of life on Earth: A new global map of terrestrial ecoregions provides an innovative tool for conserving biodiversity. BioScience 51:933–938
Ordeñana MA, Crooks KR, Boydston EE, Fisher RN, Lyren LM, Siudyla S, Haas CD, Harris S, Hathaway SA, Turschak GM, Miles AK, Van Vuren DH (2010) Effects of urbanization on carnivore species distribution and richness. J Mamm 91:1322–1331. https://doi.org/10.1644/09-MAMM-A-312.1
Palmer BC, Conover MR, Frey SN (2010) Replication of a 1970s study on domestic sheep losses to predators on Utah’s summer rangelands. Range Ecol Manage 63:689–695. https://doi.org/10.2111/REM-D-09-00190.1
Parker ID, Lopez RR, Padia R, Gallagher M, Karthikeyan R, Cathey JC, Silvy NJ, Davis DS (2013) Role of free-ranging mammals in the deposition of Escherichia coli into a Texas floodplain. Wildl Res 40:570–577. https://doi.org/10.1071/WR13082
Paviolo A, Crawshaw P, Caso A et al (2015) Leopardus pardalis. The IUCN Red List of Threatened Species 2015: e.T11509A97212355. https://doi.org/10.2305/IUCN.UK.2015-4.RLTS.T11509A50653476.en
Pechacek PE, Lindzey FRG, Anderson STH (2000) Autumn and winter diet of the swift fox (Vulpes velox) in south-eastern Wyoming. Hystrixthe Ital J Mammal 11:83–87. https://doi.org/10.4404/hystrix-11.2-4153
Pence DB, Windberg LA, Pence BC, Sprowls R (1983) The epizootiology and pathology of sarcoptic mange in coyotes, Canis latrans, from south Texas. J Parasit 69:1100–1115
Pence DB, Tewes ME, Shindle DB, Dunn DM (1995) Notoedric mange in an ocelot (Felis pardalis) from southern Texas. J Wildl Dis 31:558–561. https://doi.org/10.7589/0090-3558-31.4.558
Polis GA, Myers CA, Holt RD (1989) The ecology and evolution of intraguild predation: potential competitors that eat each other. Ann Rev Ecol System 20:297–330. https://doi.org/10.1146/annurev.es.20.110189.001501
Pope JP, Miller DL, Riley MC, Anis E, Wilkes RP (2016) Characterization of a novel Canine distemper virus causing disease in wildlife. J Vet Diagn Invest 28:506–513
Prugh LR, Sivy KJ (2020) Enemies with benefits: integrating positive and negative interactions among terrestrial carnivores. Ecol Lett 23:902–918. https://doi.org/10.1111/ele.13489
Prugh LR, Stoner CJ, Epps CW, Bean WT, Ripple WJ, Laliberte AS, Brashares JS (2009) The rise of the mesopredator. Bioscience 59:779–791. https://doi.org/10.1525/bio.2009.59.9.9
Ranglack DH, Durham S, du Toit JT (2015) Competition on the range: science versus perception in a bison-cattle conflict in the western USA. J Appl Ecol 52:467–474. https://doi.org/10.1111/1365-2664.12386
Ray AJ (1987) The fur trade in North America: an overview from a geographical perspective. In: Novak M, Baker JA, Obbard ME, Malloch B (eds) Wild furbearer management and conservation in North America. Ontario Ministry of Natural Resources, Toronto, Ontario, pp 21–30
Reid JM (2019) Effects of predator reduction on northern bobwhite (Colinus virginianus) in the Rolling Plains of Texas. M.S. Thesis, Texas Tech University, Lubbock, TX, 64pp
Reid F, Helgen K, Kranz A (2016a) Mustela erminea. The IUCN Red List of Threatened Species 2016a: e.T29674A45203335. http://dx.doi.org/https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T29674A45203335.en Copyright: Mamm Species 8235:1. https://doi.org/10.2307/3503967
Reid F, Schiaffini M, Schipper J (2016b) Neovison vison. The IUCN Red List of Threatened Species 2016b: e.T41661A45214988. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41661A45214988.en
Reid F, Schipper J, Timm R (2016c) Bassariscus astutus. The IUCN Red List of Threatened Species 2016c: e.T41680A45215881. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41680A45215881.en
Reynolds JC, Tapper SC (1996) Control of mammalian predators in game management and conservation. Mamm Rev 26:127–155. https://doi.org/10.1111/j.1365-2907.1996.tb00150.x
Roberts NM, Crimmins SM (2010) Bobcat population status and management in North America: evidence of large-scale population increase. J Fish Wildl Manage 1:169–174. https://doi.org/10.3996/122009-JFWM-026
Roberts NM, Lovallo MJ, Crimmins SM (2020) River otter status, management, and distribution in the United States: evidence of large-scale population increase and range expansion. J Fish Wildl Manage 11:279–286. https://doi.org/10.3996/102018-JFWM-093
Robinson QH, Bustos D, Roemer GW (2014) The application of occupancy modeling to evaluate intraguild predation in a model carnivore system. Ecology 95:3112–3123. https://doi.org/10.1890/13-1546.1
Roemer GW, Gompper ME, Van Valkengurgh B (2009) The ecological role of the mammalian mesocarnivore. Bioscience 59:165–173. https://doi.org/10.1525/bio.2009.59.2.9
Roemer G, Cypher B, List R (2016) Urocyon cinereoargenteus. The IUCN Red List of Threatened Species 2016: e.T22780A46178068. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T22780A46178068.en. Mamm Species 8235:1. https://doi.org/10.2307/3503957
Rogers RE (1995) A review of state raccoon hunting and coondog training regulations. Wildl Soc Bull 23:398–406
Rohwer FC, Garrettson PR, Mense BJ (1995) Can predator trapping improve waterfowl recruitment in the Prairie Pothole region? In: Proceedings of the 7th Eastern wildlife damage management conference vol 7, pp 12–22
Rosenheim JA (2004) Top predators constrain the habitat selection games played by intermediate predators and their prey. Israel J Zool 50:129–138. https://doi.org/10.1560/K796-DMB2-546Q-Y4AQ
Rosenstock SS, Ballard WB, Devos JC (1999) Viewpoint: benefits and impacts of wildlife water developments. J Range Manage 52:302–311
Rudd J, Clifford D, Richardson D, Cypher B, Westall T, Kelly E, Foley J (2019) Hematologic and serum chemistry values of endangered San Joaquin kit foxes (Vulpes macrotis mutica) with sarcoptic mange. J Wildl Dis 55:410–415. https://doi.org/10.7589/2017-10-270
Ruette S, Stahl P, Albaret M (2003) Factors affecting trapping success of red fox Vulpes, stone marten Martes foina and pine marten M. martes in France. Wildl Biol 9:11–19. https://doi.org/10.2981/wlb.2003.003
Sacks BN, Neale JCC (2002) Foraging strategy of a generalist predator toward a special prey: coyote predation on sheep. Ecol Appl 12:299–306. https://doi.org/10.1890/1051-0761(2002)012[0299:FSOAGP]2.0.CO;2
Sacks BN, Lounsberry ZT, Statham MJ (2018) Nuclear genetic analysis of the red fox across its Trans-Pacific Range. J Heredity 109(5):573–584. https://doi.org/10.1093/jhered/esy028
Saitone TL, Bruno EM (2020) Cost effectiveness of livestock guardian dogs for predator control. Wildl Soc Bull 44:101–109. https://doi.org/10.1002/wsb.1063
Šálek M, Drahníková L, Tkadlec E (2015) Changes in home range sizes and population densities of carnivore species along the natural to urban habitat gradient. Mamm Rev 45:1–14. https://doi.org/10.1111/mam.12027
Santymire R, Graves G (2019) Black-footed ferret SAFE program action plan 2019–2021. 26
Sargeant AB, Sovada MA, Shaffer TL (1995) Seasonal predator removal relative to hatch rate of duck nests in waterfowl production areas. Wildl Soc Bull 23:507–513
Sasmal I, Honness K, Bly K, McCaffery M, Kunkel K, Jenks JA, Phillips M (2015) Release method evaluation for swift fox reintroduction at Bad River Ranches in South Dakota. Restor Ecol 23:491–498.https://doi.org/10.1111/rec.12211
Scasta JD, Stam B, Windh JL (2017) Rancher-reported efficacy of lethal and non-lethal livestock predation mitigation strategies for a suite of carnivores. Sci Rep 7:1–11. https://doi.org/10.1038/s41598-017-14462-1
Schoener TW (1974) Resource partitioning in ecological communities. Science 185:27–39. https://doi.org/10.1126/science.185.4145.27
Schuttler SG, Parsons AW, Forrester TD, Baker MC, McShea WJ, Costello R, Kays R (2017) Deer on the lookout: how hunting, hiking and coyotes affect white‐tailed deer vigilance. J Zool 301:320–327
Seidler RG, Gese EM (2012) Territory fidelity, space use, and survival rates of wild coyotes following surgical sterilization. J Ethol 30:345–354. https://doi.org/10.1007/s10164-012-0330-4
Seidler RG, Gese EM, Conner MM (2014) Using sterilization to change predation rates of wild coyotes: a test case involving pronghorn fawns. Appl Anim Behav Sci 154:83–92. https://doi.org/10.1016/j.applanim.2014.02.006
Serfass T, Evans S, Polechla P (2015) Lontra canadensis. The IUCN Red List of Threatened Species 2015: e.T12302A21936349. https://doi.org/10.2305/IUCN.UK.2015-2.RLTS.T12302A21936349
Serieys LEK, Foley J, Owens S, Woods L, Boydston EE, Lyren LM, Poppenga RH, Clifford DL, Stephenson N, Rudd J, Riley SPD (2013) Serum chemistry, hematologic, and post-mortem findings in free-ranging bobcats (Lynx rufus) with notoedric mange. J Parasit 99:989–996. https://doi.org/10.1645/12-175.1
Sévêque A, Gentle LK, López-Bao JV, Yarnell RW, Uzal A (2020) Human disturbance has contrasting effects on niche partitioning within carnivore communities. Biolog Rev 95:1689–1705. https://doi.org/10.1111/brv.12635
Shivik J (2014) The predator paradox: ending the war with wolves, bears, cougars, and coyotes. Beacon Press
Shriner SA, Ellis JW, Root JJ, Roug A, Stopak SR, Wiscomb GW, Zierenberg JR, Ip HS, Torchetti MK, DeLiberto TJ (2021) SARS-CoV-2 exposure in escaped mink, Utah, USA. Emerg Infect Dis 27(3):988. https://doi.org/10.3201/eid2703.204444
Slate D, Rupprecht CE, Rooney JA, Donovan D, Lein DH, Chipman RB (2005) Status of oral rabies vaccination in wild carnivores in the United States. Virus Res 111:68–76. https://doi.org/10.1016/j.virusres.2005.03.012
Slough BG, Jessup RH, McKay DI, Stephenson AB (1987) Wild furbearer management in western and northern Canada. In: Novak M, Baker JA, Obbard ME, Malloch B (eds) Wild furbearer management and conservation in North America. Ontario Ministry of Natural Resources, Toronto, Ontario, pp 1062–1976
Smallwood KS, Bell DA, Snyder SA, DiDonato JE (2010) Novel scavenger removal trials increase wind turbine—caused avian fatality estimates. J Wildl Manage 74(5):1089–1096. https://doi.org/10.2193/2009-266
Smith KF, Acevedo-Whitehouse K, Pedersen AB (2009) The role of infectious diseases in biological conservation. Anim Conserv 12:1–12. https://doi.org/10.1111/j.1469-1795.2008.00228.x
Smith JA, Brown MB, Harrison JO, Powell LA (2017) Predation risk: a potential mechanism for effects of a wind energy facility on Greater Prairie-Chicken survival. Ecosphere 8(6):e01835. https://doi.org/10.1002/ecs2.1835
Soulé M, Bolger D, Alberts A, Wright J, Sorice M, Hill S (1988) Reconstructed dynamics of rapid extinctions of chaparral-requiring birds in urban habitat islands. Conserv Biol 2:75–92. https://doi.org/10.1111/j.1523-1739.1988.tb00337.x
Sovada MA, Woodward RO, IGL LD (2009) Historical range, current distribution, and conservation status of the swift fox, Vulpes velox, in North America. Can Field-Nat 123:346–367. https://doi.org/10.22621/cfn.v123i4.1004
Sprayberry TR, Edelman AJ (2016) Food provisioning of kits by a female eastern spotted skunk. Southeast Nat 15(4). https://doi.org/10.1656/058.015.0417
Statham MJ, Sacks BN, Aubry KB, Perrine JD, Wisely SM (2012) The origin of recently established red fox populations in the United States: translocations or natural range expansions? J Mamm 93(1):52–65. https://doi.org/10.1644/11-MAMM-A-033.1
Steinel A, Parrish CR, Bloom ME, Truyen U (2001) Parvovirus infections in wild carnivores. J Wildl Dis 37:594–607. https://doi.org/10.7589/0090-3558-37.3.594
Stevenson ER, Lashley MA, Chitwood MC, Garabedian JE, Swingen MB, DePerno CS, Moorman CE (2019) Resource selection by coyotes (Canis latrans) in a longleaf pine (Pinus palustris) ecosystem: effects of anthropogenic fires and landscape features. Can J Zool 97(2):165–171. https://doi.org/10.1139/cjz-2018-0150
Stuetzer B, Hartmann K (2014) Feline parvovirus infection and associated diseases. Vet J 201:150–155. https://doi.org/10.1016/j.tvjl.2014.05.027
Tardy O, Massé A, Pelletier F, Mainguy J, Fortin D (2014) Density-dependent functional responses in habitat selection by two hosts of the raccoon rabies virus variant. Ecosphere 5:1–16. https://doi.org/10.1890/ES14-00197.1
Theimer TC, Clayton AC, Martinez A, Peterson DL, Bergman DL (2015) Visitation rate and behavior of urban mesocarnivores differs in the presence of two common anthropogenic food sources. Urban Ecosys 18:895–906. https://doi.org/10.1007/s11252-015-0436-x
Thomas PJ, Newell EE, Eccles K et al (2021) Co-exposures to trace elements and polycyclic aromatic compounds (PACs) impacts North American river otter (Lontra canadensis) baculum. Chemosphere 265:128920. https://doi.org/10.1016/j.chemosphere.2020.128920
Thompson CM, Gese EM (2007) Food webs and intraguild predation: community interactions of a native mesocarnivore. Ecology 88:334–346. https://doi.org/10.1890/0012-9658(2007)88[334:FWAIPC]2.0.CO;2
Thornton D, Scully A, King T, Fisher S, Fitkin S, Rohrer J (2018) Hunting associations of American badgers (Taxidea taxus) and coyotes (Canis latrans) revealed by camera trapping. Can J Zool 96(7):769–773. https://doi.org/10.1139/cjz-2017-0234
Timm R, Cuarón AD, Reid F et al (2016) Procyon lotor. The IUCN Red List of Threatened Species 2016: e.T41686A45216638. https://doi.org/10.2305/IUCN.UK.2016-1.RLTS.T41686A45216638.en
UDWR (2013) Utah’s predator control program summary. Final Report
United States Department of Agriculture—National Agricultural Statistics Service (2015a) Sheep and lamb predator and nonpredator death loss in the United States, 2015a. USDA–APHIS–VS–CEAH–NAHMS Fort Collins, CO #721.0915. www.nass.usda.gov. Accessed 25 Feb 2021
United States Department of Agriculture—National Agricultural Statistics Service (2015b) Cattle and calves death loss in the United States due to predator and nonpredator causes, 2015b. USDA–APHIS–VS–CEAH. Fort Collins, CO #745.1217. www.nass.usda.gov. Accessed 25 Feb 2021
van Eeden LM, Crowther MS, Dickman CR, Macdonald DW, Ripple WJ, Ritchie EG, Newsome TM (2018a) Managing conflict between large carnivores and livestock. Conserv Biol 32:26–34. https://doi.org/10.1111/cobi.12959
van Eeden LM, Eklund A, Miller JRB, López-Bao JV, Chapron G, Cejtin MR, Crowther MS, Dickman CR, Frank J, Krofel M, Macdonald DW, McManus J, Meyer TK, Middleton AD, Newsome TM, Ripple WJ, Ritchie EG, Schmitz OJ, Stoner KJ, Tourani M, Treves A (2018b) Carnivore conservation needs evidence-based livestock protection. PLoS Biol 16:1–8. https://doi.org/10.1371/journal.pbio.2005577
Verdy A, Amarasekare P (2010) Alternative stable states in communities with intraguild predation. J Theor Biol 262:116–128. https://doi.org/10.1016/j.jtbi.2009.09.011
Wagner FH (1988) Predator control and the sheep industry. Regina Books, Claremont, California
Wagner KK, Conover MR (1999) Effect of preventive coyote hunting on sheep losses to coyote predation. J Wildl Manage 63:606–612. https://doi.org/10.2307/3802649
White PJ, Berry WH, Eliason JJ, Hanson MT (2000) Catastrophic decrease in an isolated population of kit foxes. Southwest Nat 45:204–211. https://doi.org/10.2307/3672462
White HB, Decker T, O’Brien MJ, Organ JF, Roberts NM (2015) Trapping and furbearer management in North American wildlife conservation. Internat J Environ Stud 5:756–769. https://doi.org/10.1080/00207233.2015.1019297
White HB, Batcheller GR, Boggess EK, Brown CL, Butfiloski JW, Decker TA, Erb JD, Fall MW, Hamilton DA, Hiller TL, Hubert GF Jr (2021) Best management practices for trapping furbearers in the United States. Wildl Monog 207(1):3–59. https://doi.org/10.1002/wmon.1057
Wildlife Services (2015) Program Data Report G—animals dispersed/killed or euthanized/removed or destroyed/freed or relocated. https://www.aphis.usda.gov/aphis/ourfocus/wildlifedamage/SA_Reports/SA_PDRs. Accessed 24 Feb 2021
Wildlife Services (2019) Program Data Report G—animals dispersed/killed or euthanized/removed or destroyed/freed or relocated. https://www.aphis.usda.gov/aphis/ourfocus/wildlifedamage/SA_Reports/SA_PDRs. Accessed 24 Feb 2021
Willcox D (2020) Conservation status, ex situ priorities and emerging threats to small carnivores. Int Zoo Yb 54:1–16. https://doi.org/10.1111/izy.12275
Williams ES, Thorne ET, Appel MJG, Belitsky DW (1988) Canine distemper in black-footed ferrets (Mustela nigripes) from Wyoming. J Wildl Dis 24:385–398. https://doi.org/10.7589/0090-3558-24.3.385
Williams ES, MillsK KDR, Thorne ET, Boerger-Fields A (1994) Plague in a black-footed ferret (Mustela nigripes). J Wildl Dis 30:581–585. https://doi.org/10.7589/0090-3558-30.4.581
Williams ES, Erson SL, Cavender J, Lynn C, List K, Hearn C, Appel MJG (1996) Vaccination of black-footed ferrets (Mustela nigripes) x Siberian polecat (M. eversmanni) hybrids against domestic ferrets (M. putorius furo) against canine distemper. J Wildl Dis 32:417–423
Wilmers CC, Post E, Peterson RO, Vucetich JA (2006) Predator disease out-break modulates top-down, bottom-up and climatic effects on herbivore population dynamics. Ecol Lett 9:383–389. https://doi.org/10.1111/j.1461-0248.2006.00890.x
Wilson RR, Blankenship TL, Hooten MB, Shivik JA (2010) Prey-mediated avoidance of an intraguild predator by its intraguild prey. Oecologia 164:921–929. https://doi.org/10.1007/s00442-010-1797-8
Wright JV (1987) Archeological evidence for the use of furbearers in North America. In: Novak M, Baker JA, Obbard ME, Malloch B (eds) Wild furbearer management and conservation in North America. Ontario Ministry of Natural Resources, Toronto, Ontario, pp 3–12
Young JK, Olson KA, Reading RP, Amgalanbaatar S, Berger J (2011) Is wildlife going to the dogs? Impacts of feral and free-roaming dogs on wildlife populations. Bioscience 61(2):125–132. https://doi.org/10.1525/bio.2011.61.2.7
Young JK, Draper JP, Breck S (2019a) Mind the gap: experimental tests to improve efficacy of fladry for nonlethal management of coyotes. Wildl Soc Bull 43:265–271. https://doi.org/10.1002/wsb.970
Young JK, Draper JP, Kinka D (2019b) Spatial associations of livestock guardian dogs and domestic sheep. Human–Wildl Interact 13:6. https://doi.org/10.26076/frv4-jx12
Zimmerman A (1998) Reestablishment of swift fox in north central Montana. M.S. Thesis, Montana State University
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2023 The Author(s)
About this chapter
Cite this chapter
Young, J.K., Butler, A.R., Holbrook, J.D., Shamon, H., Lonsinger, R.C. (2023). Mesocarnivores of Western Rangelands. In: McNew, L.B., Dahlgren, D.K., Beck, J.L. (eds) Rangeland Wildlife Ecology and Conservation . Springer, Cham. https://doi.org/10.1007/978-3-031-34037-6_16
Download citation
DOI: https://doi.org/10.1007/978-3-031-34037-6_16
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-34036-9
Online ISBN: 978-3-031-34037-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)