Abstract
Aromatic side chains are often key residues in enzyme active sites and protein binding sites, making them attractive probes of protein dynamics on the millisecond timescale. Such dynamic processes can be studied by aromatic 13C or 1H CPMG relaxation dispersion experiments. Aromatic 1H CPMG relaxation dispersion experiments in phenylalanine, tyrosine and the six-ring moiety of tryptophan, however, are affected by 3J 1H–1H couplings which are causing anomalous relaxation dispersion profiles. Here we show that this problem can be addressed by site-selective 1H/2H labeling of the aromatic side chains and that artifact-free relaxation dispersion profiles can be acquired. The method has been further validated by measuring folding–unfolding kinetics of the small protein GB1. The determined rate constants and populations agree well with previous results from 13C CPMG relaxation dispersion experiments. Furthermore, the CPMG-derived chemical shift differences between the folded and unfolded states are in excellent agreement with those obtained directly from the spectra. In summary, site-selective 1H/2H labeling enables artifact-free aromatic 1H CPMG relaxation dispersion experiments in phenylalanine and the six-ring moiety of tryptophan, thereby extending the available methods for studying millisecond dynamics in aromatic protein side chains.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aromatic amino acids are an important subgroup of amino acids. They are bulky and responsible for a significant proportion of the protein hydrophobic core, where they typically form pairs or clusters making specific aromatic–aromatic interactions (Burley and Petsko 1985, 1989). They are overrepresented in protein binding interfaces where they contribute significantly to the binding free energy (Birtalan et al. 2010; Bogan and Thorn 1998; Lo Conte et al. 1999) and, in the form of His and Tyr, are key residues in enzyme catalysis (Bartlett et al. 2002). Even more, Phe and Tyr undergo frequent 180° rotations of the χ2 angle (‘ring flips’) and thereby provide unique information of transient ‘breathing’ processes of proteins (Li et al. 1999; Wagner 1980; Wagner et al. 1976; Weininger et al. 2014b). All of these reasons make aromatic side chains interesting and useful probes for studying protein dynamics on the millisecond time scale.
Conformational transitions on this time scale are often linked to biological functions (Mittermaier and Kay 2009) and transiently populated high-energy states play important roles in enzyme catalysis (Boehr et al. 2006; Cole and Loria 2002; Eisenmesser et al. 2002) or ligand binding (Demers and Mittermaier 2009; Malmendal et al. 1999). Such transitions between different conformations generally lead to a modulation of NMR parameters as the chemical shift (Gutowsky and Saika 1953) or residual dipolar couplings (Igumenova et al. 2007; Vallurupalli et al. 2007), resulting in exchange contributions to transverse relaxation rate constants. This can be probed by NMR relaxation dispersion methods from which one can gain unique information on the structures, thermodynamics and kinetics of the underlying processes (Palmer 2004; Palmer et al. 2001).
Protein dynamics on the millisecond time scale in aromatic side chains can be studied by 13C (Weininger et al. 2012) and 1H (Raum et al. 2018) CPMG relaxation dispersion experiments. The key requirement for 13C relaxation dispersion experiments is site-selective 13C labeling of aromatic side chains (Lundström et al. 2012; Schörghuber et al. 2018; Weininger 2019), which eliminates 1J 13C–13C couplings. To date, there are several well established labeling strategies, that achieve this goal (Kasinath et al. 2013; Lichtenecker 2014; Lichtenecker et al. 2013; Lundström et al. 2007; Milbradt et al. 2015; Schörghuber et al. 2015, 2017a, b; Teilum et al. 2006; Weininger 2017a, b). 13C relaxation dispersion experiments for the study of dynamics on the ms (Weininger et al. 2012) and µs (Weininger et al. 2014a) time scale have been developed and applied on the characterization of ring flips (Weininger et al. 2013, 2014b) and transient histidine tautomerization (Weininger et al. 2017). In contrast, sole site-selective 13C labeling is not sufficient to enable artifact-free 1H CPMG relaxation dispersion experiments. It eliminates artifacts from possible strong 13C–13C couplings, but sizeable 3J 1H–1H couplings (that exist in Phe, Tyr, and the 6-ring moiety of Trp) cause severe artifacts (Raum et al. 2018). Therefore, additional site-selective 1H/2H labeling is required.
Here we demonstrate that in site-selective 1H/2H labeled aromatic side chains the artifact caused by 3J 1H–1H couplings is gone and flat relaxation dispersion profiles can be measured in the absence of exchange. Artifact-free relaxation dispersion profiles can be acquired if chemical exchange is present, and meaningful (exchange rates and populations) and correct (chemical shift differences) parameters of this process can be determined. This has been demonstrated on the folding/unfolding of protein GB1 at high temperatures.
Materials and methods
Protein samples
Uniformly 1H and site-selective 13C labeled, using 2 g/L 2-13C1 glucose (Lundström et al. 2007), as well as site-selective 1H and 13C labeled, using specific synthesized precursors (Lichtenecker 2014; Lichtenecker et al. 2013; Schörghuber et al. 2015) (80 mg/L for Phe and Tyr, 10 mg/L for Trp), GB1 (QDD variant) was expressed and purified as described in (Lindman et al. 2006). Samples contained 990 µM (uniformly 1H labeled) or 440 µM (site-selective 1H labeled) protein in 20 mM HEPES and 10% (v/v) D2O at pH 7.0. Small amounts of NaN3 were added.
NMR spectroscopy
All experiments were acquired on a Bruker Avance III spectrometer at a static magnetic field of 14.1 T or 18.8 T and 298 K or 313 K. 1H CPMG relaxation dispersion experiments were performed using a relaxation compensated approach as published before (Raum et al. 2018). A constant relaxation period of 20 ms was chosen in all experiments, except for 18.8 T and 313 K where 40 ms was chosen. Refocusing frequencies are between 100 (50 for 18.8 T) and 1000 Hz and B1 field strengths for the CPMG pulses are 16 kHz (14.1 T) and 18.5 kHz (18.8 T). During the CPMG period the 1H carrier was put in the aromatic region (6.7 ppm). No 2H decoupling was used for the experiments. Experiments were typically performed with 72 number of scans resulting in a experimental time of 2 days. This was done to assure the highest data quality possible for the establishment of the method. Further the dispersion step is quite moderate and there are only three positions for the global fit. Spectra were processed with NMRPipe (Delaglio et al. 1995) and analyzed with PINT (Ahlner et al. 2013) or NMRView (Johnson 2004). The spectra have not been referenced to DSS.
Data analysis
Measurement uncertainties of relaxation rates were estimated as the average standard deviation of double measurements. CPMG relaxation dispersion experiments were fitted globally to the Carver-Richards equation (Carver and Richards 1972; Davis et al. 1994). Data modeling utilized the Levenberg–Marquardt (Press et al. 2002) nonlinear least-squares optimization algorithm implemented in MATLAB. For error estimation, Monte-Carlo simulations with 1000 steps were executed. Derived Δδ values were compared with 1H shift differences between native and unfolded signals at 40 °C.
Results and discussion
GB1 is a small, 56 residue protein containing 6 aromatic amino acids: 1 Trp (W43), 2 Phe (F30 and F52) and 3 Tyr (Y3 and Y33 and Y45).
Site-selective 1H/2H labeling in the aromatic side chains of GB1
Both labeling methods result in site-selective 13C labeling in Phe ε* (F30 and F52), Tyr ε* (Y3 and Y33 and Y45) and Trp ζ3 (W43). In the uniformly 1H labeled sample (based on 2-13C1 glucose) Trp δ1 and ζ2 (Fig. 1) are labeled additionally. The resulting spectra are highly comparable, signals from the site-selective 1H labeled sample showed reduced linewidths in 1H (about 25% reduced). If normalized by the protein concentration the site-selective 1H labeled sample will display 5.5 times the signal strength for Phe ε* (F30 and F52) and three times the signal strength for Trp ζ3 (W43). In case of Phe ε*, with about 20% 13C incorporation from 2-13C1 glucose (Weininger 2017a), this translates to an apparent 110% 13C incorporation in the site-selective 1H labeled sample. This can be explained by a close to 100% 13C incorporation, 5% gain because losses from 3J 1H–1H couplings during the INEPT transfer periods are suppressed and additional gain from improved 1H relaxation. In case of Trp ζ3, with about 25% 13C incorporation from 2-13C1 glucose (Weininger 2017a) this translates to an apparent 75% 13C incorporation in the site-selective 1H labeled sample. Applying the same reasoning for Trp ζ3, one can estimate around 70% incorporation in case of the site-selective 1H labeled sample (at 10 mg/L precursor). In case of Tyr the site-selective labeling did not work well (around 4% 13C incorporation). The reason for this is not clear. As a direct consequence of this only a limited number of experiments could be performed on Tyr. On the other hand this highlights the selective incorporation of the Phe precursor in Phe. Results of 13C incorporation are summarized in Table 1.
Aromatic side chains (Trp, Phe and Tyr) with 1H/2H and 13C labeling resulting from 2-13C1 glucose (a) and specific synthesized precursors (b). 13C with an attached proton are colored red. 2H in the aromatic ring is colored blue. 13C incorporation from glucose is 22% (Fε*), 19% (Yε*), 24% (Wζ3) and 12% (Wζ2). 13C incorporation (together with 2H incorporation at adjacent positions) from the specific precursors is 100% (Fε*) and > 75% (Wζ3). The same should be expected for Yε* (but did not in this attempt)
In order to evaluate the amount of vicinal protons (3J) to the 13C bound proton we performed 2D 1H13C-HSQC-1H1H-TOCSY experiments. For the uniformly 1H labeled samples we observe strong cross signals to vicinal (and for Trp ζ3 4J) protons (Fig. 2, red). These are completely absent (Phe ε*) or strongly reduced (Trp ζ3) in the site-selective 1H labeled sample, indicating complete (> 98%, Phe ε*) or a high amount of (about 90%, Trp ζ3) deuteration of vicinal protons, that are believed to cause artifacts in aromatic 1H CPMG relaxation dispersion experiments (Raum et al. 2018). In summary, labeling by synthetic 1H and 13C selective precursors can be seen as perfect (Phe ε*) or pretty good (Trp ζ3) in terms of producing high amounts of isolated 1H–13C spin pairs in aromatic side chains. Labeling for Tyr ε* are shown to be on the same level in previous attempts (Lichtenecker et al. 2013), but did not work properly in this work.
1D 1H (constant 13C chemical shift) slices from 1H13C HSQC-TOCSY spectra from uniformly 1H labeled (red) and site-selective labeled (black) GB1. The spectra at the top (a) are from the slice along the 13C chemical shift of F30Cε* (128.21 ppm) and the spectra at the bottom (b) are from the slice along the 13C chemical shift of W43Cζ3 (118.15 ppm). Each spectrum is normalized to the maximum intensity of the corresponding peak of a 1H13C HSQC spectrum measured under same conditions. Signals are labeled. Ratios of auto to (vicinal) cross signals in the uniformly 1H labeled cases are 27/73 (a) and 8/92 (b), in the site-selective 1H cases 100/0 (a) and 90/10 (b)
Artifact-free 1H CPMG relaxation dispersion profiles by site-selective 1H/2H labeling
Next, we recorded 1H CPMG relaxation profiles on both GB1 samples at 25 °C (no exchange conditions) and 40 °C (exchange conditions) (Weininger 2019). At 25 °C, we observe anomalous relaxation dispersion profiles for the uniformly 1H labeled sample that are caused by 3J 1H–1H couplings (Fig. 3a, c, e, red). In contrast, we observe flat relaxation dispersion profiles for the site-selective labeled sample (Fig. 3a, c, e, black), as one would expect for conditions with no chemical exchange. Resulting RMSD values for the flat dispersions are: 0.18 s−1 (F30), 0.29 s−1 (F52) and 0.39 s−1 (W43). Furthermore, R2 values at high refocusing frequencies are around 15 s−1 higher in the uniformly 1H labeled sample. 8 s−1 can be directly attributed to the 3J 1H–1H coupling (Raum et al. 2018), the rest can be interpreted as improved 1H relaxation in the site-selective 1H labeled sample. In case of Tyr (Fig. 3g), the observation was the same. Because of the low labeling yield in case of site-selective 1H/2H labeling, the recorded relaxation dispersion profile (Fig. 3e, black) is noisier. Additionally, since contribution from uniformly 1H labeled protein at 13C natural abundance are comparable to the site-selective 1H/2H labeled protein (1–4%), the artifact is reduced but still there. Under exchange conditions at 40 °C one can record pronounced relaxation dispersion profiles for the site-selective labeled sample (Fig. 3b, d, f, black). In contrast, relaxation dispersion profiles appear to be flat (Fig. 3b, red) or heavily perturbed (Fig. 3d, f, red), for the uniformly 1H labeled samples. This can be visualized as a superposition of the artifact caused by the 3J 1H–1H couplings and the relaxation dispersion profiles caused by chemical exchange. In summary, we have confirmed earlier findings, that 3J 1H–1H couplings are causing artifacts in aromatic 1H CPMG relaxation profiles (if they are larger than 2 Hz), and established that this problem can be eliminated by the use of site-selective 1H labeled precursors.
Aromatic 1H CPMG relaxation dispersion profiles of uniformly 1H labeled (colored in red) and site-selective labeled (black) GB1 at 298 K (a, c, e and g, no exchange) and 313 K (b, d and f, exchange) at a static magnetic field of 14.1 T. For F30ε* (a), F52ε* (c) and W43ζ3 (e) flat relaxation dispersion profiles are obtained with the site-selective labeled sample at 298 K and unperturbed relaxation dispersion profiles are obtained at 313 K (b, d, f). The uniformly labeled sample shows artificial profiles affected by 3J 1H–1H couplings in all cases. F30ε* at 298 K is additionally affected by ring flips that cause a constant increase in R2 values. For Y33ε* (g), the artifact is significantly reduced with the site-selective labeled sample
Unfolding of GB1 by 1H CPMG relaxation dispersion
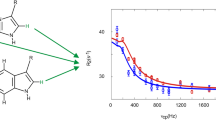
Finally, we applied aromatic 1H CPMG relaxation dispersion experiments using site-selective 1H labeled samples on a known exchanging system, GB1 at 40 °C (Weininger 2019). Relaxation dispersion profiles obtained at two magnetic field strengths can be fitted globally (for F30, F52 and W43) to a two-state exchange model (Fig. 4), resulting in an exchange rate constant (kex) of (94 ± 5) s−1 and a population of the unfolded state (pu) of (2.8 ± 0.1)%. The derived chemical shift differences from the relaxation dispersion profiles are in excellent agreement with shift differences directly derived from spectra at 40 °C, that show the characteristics of an unfolded protein (Fig. 5). The population of the unfolded state is somewhat lower than for 13C CPMG relaxation dispersion experiments (Weininger 2019). These have, however, been measured at an older sample containing large amounts of salt, and salt is known to destabilize GB1 (Lindman et al. 2006). The derived populations at low salt concentration in this work are in good agreement to unfolding transitions under these conditions.
Aromatic 1H CPMG relaxation dispersion profiles acquired on a 1H selective-labeled 440 µM sample of GB1 in 20 mM HEPES, pH 7, at 40 °C and static magnetic field strengths of 14.1 (black) and 18.8 (blue). Data are shown for F30ε* (a), F52ε* (b) and W43ζ3 (c). Solid lines represent the global fit of a two-state exchange model to the experimental data. The resulting exchange rate kex is (94 ± 5) s−1, the population of the unfolded state pu (2.8 ± 0.1)%. Differences in chemical 1H shift were estimated to (0.22 ± 0.01) ppm for F30ε*, (0.14 ± 0.01) ppm for F52ε* and (0.48 ± 0.01) ppm for W43ζ3 and are in good agreement with 1H shift differences between native and unfolded signals in the spectra, which are 0.2 ppm, 0.135 ppm and 0.475 ppm
Correlation of 1H chemical shift differences between the folded and unfolded states of GB1 derived from CPMG relaxation dispersion experiments under native conditions and measured directly from an aromatic 1H–13C HSQC spectrum. The solid grey line represents the ideal correlation. Points are labeled. Pairwise RMSD of the points is 0.012 ppm
Conclusions
We have demonstrated that artifact-free aromatic 1H CPMG relaxation dispersion profiles can be obtained using site-selective 1H labeled precursors, which produce isolated 1H–13C spin pairs in aromatic side chains. In contrast, relaxation dispersion profiles in a uniformly 1H labeled samples are heavily perturbed by high (7–8 Hz) 3J 1H–1H coupling constants. Correct parameters of unfolding of GB1 could be derived by 1H CPMG relaxation dispersion experiments on a site-selective 1H and 13C labeled sample. By site-selective 1H/2H labeling one can therefore extend the positions suitable for aromatic 1H CPMG relaxation dispersion experiments to Fε and Wζ3 specifically and Yε in principal (this work) and Fδ, Fζ, Yδ, Wε3, Wη2 and Wζ2 (using suitable precursors), thereby extending the arsenal of aromatic probes for the study of ms dynamics.
References
Ahlner A, Carlsson M, Jonsson BH, Lundström P (2013) PINT: a software for integration of peak volumes and extraction of relaxation rates. J Biomol NMR 56:191–202. https://doi.org/10.1007/s10858-013-9737-7
Bartlett GJ, Porter CT, Borkakoti N, Thornton JM (2002) Analysis of catalytic residues in enzyme active sites. J Mol Biol 324:105–121
Birtalan S, Fisher RD, Sidhu SS (2010) The functional capacity of the natural amino acids for molecular recognition. Mol Biosys 6:1186–1194. https://doi.org/10.1039/b927393j
Boehr DD, McElheny D, Dyson HJ, Wright PE (2006) The dynamic energy landscape of dihydrofolate reductase catalysis. Science 313:1638–1642
Bogan AA, Thorn KS (1998) Anatomy of hot spots in protein interfaces. J Mol Biol 280:1–9
Burley SK, Petsko GA (1985) Aromatic-aromatic interaction: a mechanism of protein structure stabilization. Science 229:23–28
Burley SK, Petsko GA (1989) Electrostatic interactions in aromatic oligopeptides contribute to protein stability trends. Biotech 7:354–359
Carver JP, Richards RE (1972) General 2-site solution for chemical exchange produced dependence of T2 upon Carr-Purcell pulse separation. J Magn Reson 6:89. https://doi.org/10.1016/0022-2364(72)90090-x
Cole R, Loria JP (2002) Evidence for flexibility in the function of ribonuclease A. Biochemistry 41:6072–6081
Davis DG, Perlman ME, London RE (1994) Direct measurements of the dissociation-rate constant for inhibitor-enzyme complexes via the T-1-rho and T-2 (Cpmg) methods. J Magn Reson B 104:266–275. https://doi.org/10.1006/jmrb.1994.1084
Delaglio F, Grzesiek S, Vuister GW, Zhu G, Pfeifer J, Bax A (1995) Nmrpipe - a multidimensional spectral processing system based on unix pipes. J Biomol NMR 6:277–293
Demers JP, Mittermaier A (2009) Binding mechanism of an SH3 domain studied by NMR and ITC. J Am Chem Soc 131:4355–4367. https://doi.org/10.1021/ja808255d
Eisenmesser EZ, Bosco DA, Akke M, Kern D (2002) Enzyme dynamics during catalysis Science 295:1520–1523
Gutowsky HS, Saika A (1953) Dissociation, chemical exchange, and the proton magnetic resonance in some aqueous electrolytes. J Chem Phys 21:1688–1694
Igumenova TI, Brath U, Akke M, Palmer AG (2007) Characterization of chemical exchange using residual dipolar coupling. J Am Chem Soc 129(144):13396–13397
Johnson BA (2004) Using NMRView to visualize and analyze the NMR spectra of macromolecules. Methods Mol Biol 278:313–352
Kasinath V, Valentine KG, Wand AJ (2013) A C-13 labeling strategy reveals a range of aromatic side chain motion in calmodulin. J Am Chem Soc 135:9560–9563
Li H, Yamada H, Akasaka K (1999) Effect of pressure on the tertiary structure and dynamics of folded basic pancreatic trypsin inhibitor. Biophys J 77:2801–2812
Lichtenecker RJ (2014) Synthesis of aromatic C-13/H-2-alpha-ketoacid precursors to be used in selective phenylalanine and tyrosine protein labelling. Org Biomol Chem 12:7551–7560. https://doi.org/10.1039/c4ob01129e
Lichtenecker RJ, Weinhaupl K, Schmid W, Konrat R (2013) alpha-Ketoacids as precursors for phenylalanine and tyrosine labelling in cell-based protein overexpression. J Biomol NMR 57:327–331
Lindman S, Xue WF, Szczepankiewicz O, Bauer MC, Nilsson H, Linse S (2006) Salting the charged surface: pH and salt dependence of protein G B1 stability. Biophys J 90:2911–2921. https://doi.org/10.1529/biophysj.105.071050
Lo Conte L, Chothia C, Janin J (1999) The atomic structure of protein-protein recognition sites. J Mol Biol 285:2177–2198
Lundström P et al (2007) Fractional C-13 enrichment of isolated carbons using [1-C-13]- or [2-C-13]-glucose facilitates the accurate measurement of dynamics at backbone C-alpha and side-chain methyl positions in proteins. J Biomol NMR 38:199–212
Lundström P, Ahlner A, Blissing AT (2012) Isotope labeling methods for relaxation measurements isotope labeling in biomolecular NMR 992:63–82
Malmendal A, Evenas J, Forsen S, Akke M (1999) Structural dynamics in the C-terminal domain of calmodulin at low calcium levels. J Mol Biol 293:883–899
Milbradt AG, Arthanari H, Takeuchi K, Boeszoermenyi A, Hagn F, Wagner G (2015) Increased resolution of aromatic cross peaks using alternate C-13 labeling and TROSY. J Biomol NMR 62:291–301
Mittermaier AK, Kay LE (2009) Observing biological dynamics at atomic resolution using NMR. Trends Biochem Sci 34:601–611
Palmer AG (2004) NMR characterization of the dynamics of biomacromolecules. Chem Rev 104:3623–3640
Palmer AG, Kroenke CD, Loria JP (2001) Nuclear magnetic resonance methods for quantifying microsecond-to-millisecond motions in biological macromolecules. Meth Enzymol 339:204–238
Press WH, Teukolsky SA, Vetterling WT, Flannery BP (2002) Numerical recipes in C++: the art of scientific computing, 2nd edn. Cambridge University Press, New York
Raum HN, Dreydoppel M, Weininger U (2018) Conformational exchange of aromatic side chains by (1)H CPMG relaxation dispersion. J Biomol NMR 72:105–114. https://doi.org/10.1007/s10858-018-0210-5
Schörghuber J, Sara T, Bisaccia M, Schmid W, Konrat R, Lichtenecker RJ (2015) Novel approaches in selective tryptophan isotope labeling by using Escherichia coli overexpression media. ChemBioChem 16:746–751
Schörghuber J, Geist L, Bisaccia M, Weber F, Konrat R, Lichtenecker R (2017a) Anthranilic acid, the new player in the ensemble of aromatic residue labeling precursor compounds. J Biomol NMR 69:13–22. https://doi.org/10.1007/s10858-017-0129-2
Schörghuber J, Geist L, Platzer G, Konrat R, Lichtenecker RJ (2017b) Highly selective stable isotope labeling of histidine residues by using a novel precursor in E. coli-based overexpression systems. ChemBioChem 18:1487–1491. https://doi.org/10.1002/cbic.201700192
Schörghuber J et al (2018) Late metabolic precursors for selective aromatic residue labeling. J Biomol NMR 71:129–140. https://doi.org/10.1007/s10858-018-0188-z
Teilum K, Brath U, Lundstrom P, Akke M (2006) Biosynthetic C-13 labeling of aromatic side chains in proteins for NMR relaxation measurements. J Am Chem Soc 128:2506–2507
Vallurupalli P, Hansen DF, Stollar E, Meirovitch E, Kay LE (2007) Measurement of bond vector orientations in invisible excited states of proteins. Proc Natl Acad Sci USA 104:18473–18477
Wagner G (1980) Activation volumes for the rotational motion of interior aromatic rings in globular-proteins determined by high-resolution H-1-Nmr at variable pressure. FEBS Lett 112:280–284
Wagner G, Demarco A, Wuthrich K (1976) Dynamics of aromatic amino-acid residues in globular conformation of basic pancreatic trypsin-inhibitor (Bpti). 1. H-1 Nmr-studies. Biophys Struct Mech 2:139–158
Weininger U (2017a) Site-selective 13C labeling of proteins using erythrose. J Biomol NMR 67:191–200. https://doi.org/10.1007/s10858-017-0096-7
Weininger U (2017b) Site-selective C-13 labeling of histidine and tryptophan using ribose. J Biomol NMR 69:23–30. https://doi.org/10.1007/s10858-017-0130-9
Weininger U (2019) Optimal isotope labeling of aromatic amino acid side chains for NMR studies of protein dynamics. Methods Enzymol 614:67–86. https://doi.org/10.1016/bs.mie.2018.08.028
Weininger U, Respondek M, Akke M (2012) Conformational exchange of aromatic side chains characterized by L-optimized TROSY-selected C-13 CPMG relaxation dispersion. J Biomol NMR 54:9–14
Weininger U, Respondek M, Low C, Akke M (2013) Slow aromatic ring flips detected despite near-degenerate NMR frequencies of the exchanging nuclei. J Phys Chem B 117:9241–9247
Weininger U, Brath U, Modig K, Teilum K, Akke M (2014a) Off-resonance rotating-frame relaxation dispersion experiment for C-13 in aromatic side chains using L-optimized TROSY-selection. J Biomol NMR 59:23–29
Weininger U, Modig K, Akke M (2014b) Ring flips revisited: C-13 relaxation dispersion measurements of aromatic side chain dynamics and activation barriers in basic pancreatic trypsin inhibitor. Biochemistry 53:4519–4525
Weininger U, Modig K, Geitner AJ, Schmidpeter PAM, Koch JR, Akke M (2017) Dynamics of aromatic side chains in the active site of FKBP12. Biochemistry 56:334–343. https://doi.org/10.1021/acs.biochem.6b01157
Acknowledgements
This research was supported by the Deutsche Forschungsgemeinschaft (WE 5587/1-1).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Raum, H.N., Schörghuber, J., Dreydoppel, M. et al. Site-selective 1H/2H labeling enables artifact-free 1H CPMG relaxation dispersion experiments in aromatic side chains. J Biomol NMR 73, 633–639 (2019). https://doi.org/10.1007/s10858-019-00275-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10858-019-00275-z