Abstract
Induced by climate change associated disturbances and further promoted by changes in forest management, the proportion of European beech, a highly competitive climax species, is increasing in Central Europe. However, its climatic stability is questioned by the recent loss of vitality. In this regard, the evaluation of seed sources used for reforestation receives great attention. Here, we present a multi-trait evaluation of height, basal area and stem quality assessed over 25 years for 85 provenances grown at three sites in northern, western and eastern Germany. Considerable provenance differentiation exists, explaining 21% (proportion of trees with acceptable stem form) to 45% (basal area) of the variance on single sites and 20 to 39% across sites, while provenance by environment interactions are absent. On the landscape level, this results in distinct patterns with spatially adjacent provenances showing a similar trait expression. These patterns are highly similar for height and basal area but divergent for stem form. They could be directly linked to geographic variables with multivariate regression tree analysis that captured 58% of the phenotypic variation, delineating eleven ‘ecotypes’ shaped by local adaptation. A selection based on two multi-trait indices gives highly concordant results. Particularly, the intermediate elevations of the central highlands in Western Germany host highly suitable provenances. Lower elevation provenances from continental climates in the south-east profit from the transfer to favorable growing conditions. Since the majority of provenances was already exposed to elevated temperatures compared to their origin during the observation period, this study gives a first indication for the potential benefits of assisted migration facing climate change.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Extreme weather events have become more frequent across Europe, greatly impacting the functioning of forest ecosystems and their ability to provide their various services to society (Lindner et al. 2010; Ding et al. 2016). The transformation of pure and even-aged conifer stands to mixed forests, particularly by enhancing the proportion of broadleaved tree species has become a frequent management prescription to increase future stand resistance and resilience (Knoke et al. 2008; Jactel et al. 2017). This is often not possible by promoting natural regeneration alone, e.g., when seed bearing adults of target species are absent, and requires planting of trees that are adaptable and powerful enough to meet expected environmental changes.
European beech (Fagus sylvatica L.), hereafter named beech, is considered a major tree species to improve stand stability and resilience. It is the most abundant broadleaf species in the temperate forests of Central Europe, with a potential natural habitat covering 910,000 km2 (Bohn and Gollub 2010). In the northern part of its range it is a species of the plains, while it is the typical broadleaf of the mountains in the southern part (Alexandrov et al. 2011). Beech is a highly competitive climax species, that is not constrained by soil acidity, soil nutrition or humus type (Bolte et al. 2007). However, it has high requirements in terms of water availability and generally prefers a maritime, temperate climate with mild winters and moist summer conditions (Ellenberg 1988; Bolte et al. 2007). High summer temperatures and drought pose immanent stress to this species and are the limiting factors towards the xeric limits of its distribution (Czúcz et al. 2011). These effects are, however, not only limited to the southern regions. Dendroecological studies report strong growth declines for the south, but likewise for the core of the distribution that began already in the 1980s (Knutzen et al. 2017; Diers et al. 2022; Martinez del Castillo et al. 2022). Most recently, the severe heatwave and drought in 2018, which was even further exacerbated for some regions in 2019, resulted in unforeseen vitality loss and severe damage (Rukh et al. 2023). These range from early leaf senescence and leaf browning to dieback in adult stands (Arend et al. 2022; Frei et al. 2022; Rukh et al. 2023), and from partial to complete failure of regeneration as well as initial phases of forest development (BMEL 2019), having severe consequences for future forest generations.
In regard of the current situation, information on provenance suitability is urgently needed to provide forest practitioners with planting recommendations. It appears simple and easy to rely on the assumption that ‘local is best’ presuming that the local population shaped by natural selection is adapted to the specific environmental conditions (Langlet 1971). However, a great wealth of provenance trials, has shown that this is not always the case (Boshier et al. 2015; Rau et al. 2015). Parallel to the rising concerns about the sensitivity and resistance of beech to climatic change, it is debated, whether an assisted migration of non-local provenances originating from warmer and drier climates appears an appropriate option of active adaptation (Bolte et al. 2009). The idea behind is to introduce genes pre-adapted to expected future conditions, when the local populations by themselves are not able to adapt or migrate fast enough to keep pace with these changes (Aitken and Bemmels 2016).
The most suitable tool to address the performance and adaptability of beech populations and to identify those relevant for reforestation is the great wealth of already existing provenance trials. While early trials mainly concentrated on the national level, both in terms of provenances and sites (e.g. Krahl-Urban 1958; Madsen 1985; Teissier du Cros 1993), the range-wide evaluation of the genetic resources became the focus of an international cooperation starting in the 1980s (Muhs and von Wühlisch 1993). Six international provenance trial series of beech were established in 1986, 1987, 1988, 1995, 1998 and 2007. In total, these include 465 provenances, planted at 75 trials in 23 European countries with a variable number of 14–100 provenances per site (Liesebach et al. 2023). Various studies evaluated individual or few locally adjacent trials of these series (see a list of references for the first five series in Liesebach 2015). Most recently, patterns of range-wide local adaptation and phenotypic plasticity (Gárate-Escamilla et al. 2019) or lags in climate adaptation (Fréjaville et al. 2020) were modeled based on a large data base with phenotypic data of two series assembled by the BeechCOSTe52 data consortium (Robson et al. 2018).
Still, the enormous potential that these trials offer for decision making has not yet been investigated thoroughly. Provenance by environment interactions (P × E), which greatly affect the stability and adaptability of provenances against the stressors that prevail at given environments, were scarcely investigated for beech. Only two studies exist focusing on data of juvenile age: Alia et al. (2011) did a comprehensive analysis of P × E in survival, height and diameter (as well as some further derived variables) for 57 provenances planted in different representation at 20 trial sites across the species range. And Stojnić et al. (2015a) evaluated stability and performance in height (5 and 6 years old) of a rather small set of 15 provenances planted on four sites in south-eastern Europe. A comparable study concentrating on a more mature age and with a regional focus to the center of the species distribution, however, is missing.
The high relevance of P × E was shown for many commercially important species (e.g. Douglas fir (Zas et al. 2003), Radiata pine (Gapare et al. 2015), Northern red oak (Kormann et al. 2023)). Still, most studies of P × E concentrate on stability in single traits, while tree breeding programs generally consider several traits as being of economic and/or ecologic value which are affecting each other either positively or negatively (White et al. 2007; Pâques 2013). Traits relevant for breeding of beech are, for example, height and diameter growth, stem straightness, spiral grain, forking and branch habit, flushing and late frost damage (de Cuyper et al. 2011). Simultaneous selection accounting for multiple traits at the same time has been difficult so far, since existing methods are facing problems with multicollinearity (Christophe and Birot 1983; Olivoto and Nardino 2021). Two new developed multi-trait selection indices (multi-trait genotype-ideotype distance matrix—MDIGI (Olivoto and Nardino 2021) and multi-trait stability index—MTSI (Olivoto et al. 2019)) developed for agricultural field trials, are free from multicollinearity issues. Recently, these have been applied for the first time in forest trees, successfully combining growth, quality and survival of Norway spruce (Alexandru et al. 2023). For beech, none of the selection indices has been used so far.
Here, we selected the most suitable provenances of beech considering three traits of adaptive, economic and qualitative value that are highly correlated with the breeding objective: tree height, which is commonly used as main proxy for fitness, basal area, which combines individual tree dimension and survival to the economically important overall yield per unit area, and stem form as quality parameter relevant for wood processing and subsequent use. The objectives of this study were to (1) quantify the differentiation among provenances, (2) examine provenance by environment interactions, and (3) illustrate spatial structures present in trait variation across the species range. Further, (4) provenances of high performance were carefully chosen through multiple trait selection at each individual trial site and by considering their stability across sites.
Material and methods
Experimental design
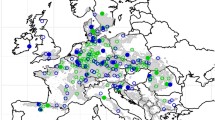
For the 4th international beech trial series 1993/95, seed from a total of 158 stands were collected across Europe in 1992 (Fig. 1). It was the first series covering almost the entire distribution, except for the Apennine peninsula and a large part of the Balkans. All seed lots were stratified for 13 weeks prior to sowing in spring 1993 at the nursery of the Thünen Institute of Forest Genetics in Grosshansdorf, Germany. Seedlings were transplanted after one growing season and finally lifted after 2 years. During winter, plants were stored as bare root seedlings in cold storage and prepared for shipment to 17 countries to be planted at a total of 23 trial sites. All trial sites were set up in the same experimental design, a randomized complete block design with three replications of multiple tree plots with 50 individuals at a spacing of 2 × 1 m.
Sampled provenances (circles) and trial sites (triangles) of the 4th series of the International Beech Network. 85 provenances (dark blue circles) were planted at all three German sites Schädtbek, Malter and Wesel (red triangles). The natural distribution of beech is shaded in dark grey (EUFORGEN 2008)
While most of the trial sites were established with a set of 49 provenances, five included 100 provenances, with three of them—Schädtbek, Wesel and Malter—being located in Germany. Here, we concentrate on 85 overlapping provenances that were planted at all three of them (Fig. 1). A list of provenances is provided in Supplementary Table S1. Located on the lower Rhine middle terrace, Wesel is the warmest, Schädtbek in Schleswig–Holstein represents the intermediate, and Malter in the Eastern Ore Mountains the coldest site (Table 1). All three sites were established on former agricultural land.
Measurement of traits
The three trial sites were measured at recurring time intervals, coinciding at age 10, 20 and 25. At age 10, all individual tree heights were recorded, except for Wesel, where a subsample of 20 trees (every 2nd and 4th row per plot, and if a tree was missing an adjacent tree in the neighbouring row) was measured. As an indicator of stem quality, stem form was rated at age 15 (Malter) to 20 (Schädtbek and Wesel) using a 4-step-scale (1 = straight, 2 = slight to moderate bends, 3 = strong bends, 4 = no straight stem, including stems with forking below 1.30 m). The latest assessment was conducted during winter 2017/2018, 25 years after planting, measuring diameter at breast height (DBH) of all living trees. Up until this measurement, all trials still remained unthinned.
For statistical analysis, traits were summarized at the plot level (Ræbild et al. 2002; Williams et al. 2002; Alia et al. 2011). For tree height, means per plot were derived. Within plot variances were closely investigated, ranging from 0.18 to 1.15 in Schädtbek, 0.12 to 1.23 in Wesel and 0.32 to 1.68 in Malter. For stem form, class 1 and 2 were summarized as ‘acceptable stem form’ to report the proportion of trees with an acceptable stem form per plot (Fig. S1). Basal area (m2) per tree was calculated using the formula \({\text{BA}} = \frac{\pi }{4}*{\text{DBH}}^{2}\), and then summed for each plot and expressed in m2/ha. The basal area sum per area accounts for survival and by that is a suitable estimate of the real productivity of a provenance (Ræbild et al. 2002; Kormann et al. 2023).
Quantification of provenance and site effects
To determine the effect of provenance, site and experimental design factors on the observed phenotypic expression, analyses of variance (ANOVA) were performed at two levels. First, we applied the following mixed linear model for each individual trial site in order to estimate the effect of provenance and spatial blocking for each trait and site:
where \(Y_{ij}\) is the phenotypic observation of a trait made for the \(i\) th provenance (\(P\)), located in the \(j\) th block (B), \(\mu\) is the overall experimental mean, and \(e\) is the experimental error (residual). Provenance was treated as random effect and the spatial blocks as fixed effect. Model assumptions for homoscedasticity and normal distribution were checked by visual inspection of diagnostic plots (Residuals vs. Fitted plot and Normal Q–Q plot). (Fig. S2 in the supplementary material shows the distribution of plot means per trait used as response variables.)
To investigate differences among sites and quantify the effect of provenance by environment interaction, we then applied a second linear mixed model across sites:
where \(Y_{ijk}\) is the phenotypic observation of a trait made for the \(i\) th provenance (\(P\)), grown at the \(j\) th environment (\(E\)), located in the \(k\) th block (B) within environment \(E\). \(P \times E\) represents the provenance by environment interaction, \(\mu\) is the overall experimental mean, and \(e\) is the experimental error (residual). In this model, provenance and \(P \times E\) were treated as random effects and environment as fixed effect.
Using restricted maximum likelihood (REML) estimation of variance parameters, best linear unbiased predictors (BLUPs) were extracted for each provenance from the individual-site and across-site linear mixed models; these were further used in the subsequent steps. To address the relationships among traits, phenotypic correlations were calculated for each site.
All statistical analyses were conducted in R (R Core Team 2019). For mixed models, we used the package metan (Olivoto and Lúcio 2020).
Spatial interpolation of trait variation
To illustrate the provenance differentiation spatially, predicted BLUPs for provenances were assigned to their corresponding geographic origin and displayed across the species distribution. To ease the comparison among traits, they were rescaled to deviation from the mean value (e.g. Ujvári-Jármay et al. 2016). That means a performance < 100% represents a trait being poorer than average, while > 100% represents a trait being higher than average.
An inverse distance weighted (IDW) interpolation was used to graphically illustrate the commonality in the phenotypic expression of spatially adjacent provenances. This interpolation method explicitly implements the assumption that things, here provenances, that are close to one another are more alike than those that are farther apart. Expressing this by a formula (Hartmann et al. 2018), the IDW approach estimates the value of \(z\) at a location \(x\) by weighting the mean of nearby observations:
where \(w_{i} = \left| {x - x_{i} } \right|^{ - \beta }\), \(\beta \ge 0\) and \(\left| \cdot \right|\) corresponds to the Euclidean distance. The inverse distance power \(\beta\) determines the degree to which the nearer point(s) are preferred over more distant points. It was set to default with \(\beta = 2\) corresponding to an inverse squared relationship. The number of surrounding points \(n\) to be included decides whether a global or local weighting is applied, here it was set to \(n = 5\). If the point \(x\) coincides with an observation location (\(x = x_{i}\)), then the observed value, \(x\), is returned to avoid infinite weights. The IDW interpolation was conducted in ArcMap 10.3, the output was delimited within the range of the species (EUFORGEN 2008).
Multi-variate grouping of provenances
To cluster geographically adjacent provenances into groups of homogeneous phenotypic expression, we used multivariate regression tree (MRT) analysis (cf. Liepe et al. 2016). MRT is a constrained clustering method (De’ath 2002) that partitions the variance in one dataset (BLUPs for provenances) based on criteria of another data set, here geographic variables of provenance origin. Two provenances with a divergent growth behaviour were excluded from this analysis, as suggested by Müller et al. (2023). They conclude based on genomic analysis that one population from Bulgaria (provenance 158) likely corresponds to the related species Fagus orientalis Lipsky and one from Northern Germany (provenance 32) represents an admixed population, that means this provenance represents the offspring of a stand established with reproductive material transferred from different parts of the range.
The MRT was conducted with R package mvpart (Therneau et al. 2013). The variance explained by each split of the regression tree was extracted with the corresponding wrapper MVPARTwrap (Ouellette and Legendre 2013).
Calculation of multi-trait indices for provenance selection
The stability and adaptability of beech provenances of different origin were estimated based on multiple traits using two recently proposed stability indices, the multi-trait genotype–ideotype distance index (MGIDI) for individual sites (Olivoto and Nardino 2021) and the multi-trait stability index (MTSI) across sites (Olivoto et al. 2019) implemented in the R package metan (Olivoto and Lúcio 2020).
The calculation of the MGIDI relies on four main steps: (i) rescaling the predicted trait means (BLUPs) so that all have a 0–100 range, (ii) using factor analysis (with varimax rotation by Kaiser 1958) to account for the correlation structure and dimensionality reduction of data, (iii) planning an ideotype based on desired values of traits and (iv) computing the distance between each genotype to the planned ideotype (Olivoto and Nardino 2021). Since the goal of tree improvement with beech is the selection for increasing adaptability and growth rates, we have designed the ideotype as having a larger height, a higher basal area, and a better quality. The selection intensity was 20%, i.e., 17 of the total 85 provenances were selected. The final MGIDI index is calculated with this formula:
where \({\text{MGIDI}}_{i }\) is the multi-trait genotype–ideotype distance index for the \(i\) th provenance, \(y_{ij}\) is the score of the \(i\) th provenance in the \(j\) th factor, and \(y_{j}\) is the \(j\) th score of the ideotype. The lower the MGIDI of a provenance, the closer it is to the ideotype and has desired values for the analysed traits.
The calculation of the MTSI relies basically on the same stepwise procedure, but requires an additional step at the beginning. First, the stability of each provenance across environments is quantified with the weighted average of the absolute scores (WAASB) (Olivoto and Lúcio 2020). Here, performance is prioritized over stability for the growth traits height and basal area (75:25), while equal weights are assigned for stem form (50:50). Scaled values for WAASB and mean performance of individual traits are then combined for simultaneous selection in the WAASBY index, which enters the factor analysis and subsequent steps as described above (ii to iv) (Olivoto and Lúcio 2020). The ideotype and the selection intensity are assigned the same as in the single site analysis. The final MTSI index is calculated with this formula:
where the \(MTSI_{i}\) is the multi-trait stability index for the \(i\) th genotype, \(F_{ij}\) is the \(j\) th score of the \(i\) th genotype and \(F_{j}\) is the \(j\) th score of the ideotype.
Juvenile height, basal area and stem form entered the calculation of these indices. To quantify the benefits of simultaneous selection for each trait and site, we finally computed the selection differentials for the selected 20% best provenances both for the selection at individual sites and across sites.
Results
Differentiation among provenances at individual sites
Significant differences among provenances were found at the three sites and for all examined traits (Table 2). The proportion of phenotypic variance explained by provenance varied between 21 and 45%, with largest effects on basal area and lowest on stem form. The experimental design effect block was significant for most traits, except for basal area in Malter, reflecting a substantial spatial heterogeneity present within the three trials that each cover an area greater than 3 ha.
On average, juvenile height was largest in Wesel (3.9 m) and lowest in Schädtbek, while highest basal area at age 25 was recorded in Schädtbek (29.8 m2/ha), and lowest in Wesel (Table 2). Malter ranged intermediate in both growth traits but had the highest proportion of acceptable stem form (58%).
The differentiation among provenances is highest for basal area, ranging between 68 and 132% compared to the corresponding site means (100%) (Fig. 2). Many provenances grow better than average across sites, while others are consistently worse than average (e.g., provenances from Sweden (SE), Denmark (DK) and Spain (ES)). The close distance between means of basal area at the three sites for the majority of provenances indicates a highly stable performance. The two provenances Seelzerturm (39) from Germany and F.D. Planoise (13) from France are particularly outstanding with a basal area > 120% at all sites.
Provenance differentiation in basal area by individual trial sites. Predicted trait means for provenances are expressed as % deviation from the site mean (100%, horizontal dashed line). That means a performance < 100% represents a basal area being poorer than average, while > 100% represents a basal area being higher than average. To account for the large sampling density in Germany, provenances are grouped by regions of provenance (810XX, Anonymous 1994), while the foreign provenances are grouped by countries (abbreviations according to ISO 3166-1 Alpha 2 code). Provenance names corresponding to the numbers are given in Table S1
Provenance means for height and basal area are highly positively correlated at all trial sites (r = 0.69 in Schädtbek, 0.73 in Wesel, 0.80 in Malter), and are likewise positively correlated with DBH and survival (Fig. S3). Growth traits and stem form, however, show a negative correlation. Particularly in Malter, the productive provenances have a smaller proportion of trees with an acceptable stem form, while the less productive provenances have a higher proportion of trees with an acceptable stem form (r = − 0.56 for stem form vs. height and − 0.45 vs. basal area).
Provenance by environment interactions
For none of the traits, provenance by environment interactions were significant. They accounted for rather small proportions of the phenotypic variance with only 2% (basal area) to 5% (stem form). Provenance had a significant effect on phenotypic trait expression of all traits in the analysis across sites (Table 3), similar to the magnitudes in the single site analysis (Table 2). This effect accounted for 20% (stem form) to 39% (basal area) of the phenotypic variation. Fixed effects for site and block within site were significant across traits as well.
Spatial patterns in provenance performance
There are distinct spatial patterns among provenances that highly correspond between juvenile height and basal area (Fig. 3a, b) but are different for stem form (Fig. 3c). Overall, provenance differentiation appears structured in large patches with spatially adjacent provenances showing a similar trait expression as highlighted by the interpolated distribution in the background (limited by the geographic extent of provenance representation). Several provenances from the central uplands of Germany, basically the center of the species distribution, particularly stand out with a good height and basal area, and likewise do some from the east of the distribution (Fig. 3a, b, labelled points representing the 20% best provenances of each trait). Concentrating on stem form, however, it is a completely different set of provenances showing the highest proportion of trees with an acceptable stem form (Fig. 3c, labelled points). None of the good growing provenances reside among those that scored a superior stem form, being in line with the described negative correlation among growth traits and stem form (Fig. S3).
Provenance differentiation at spatial scale. Trait means for height (a), basal area (b) and stem form (c) extracted for provenances from the model across sites are visualized at their corresponding location of origin (points). Multivariate regression tree analysis was used to group provenances based on their phenotypic expression constrained by geographic variables (d), the resulting groups are displayed spatially (e). Trait means are represented as % deviation from the overall mean (100%). That means a performance < 100% represents a trait being poorer than average, while > 100% represents a trait being higher than average. For a, b, c, the species range in the background represents an interpolated surface created by IDW interpolation and is colored by the same color scheme. The 20% best provenances for each individual trait are labelled with the corresponding provenance number (No in Table S1). The dendrogram (d) represents the result of the iterative clustering procedure. Red numbers indicate the variance explained by a particular split. Bar plots at the end of the tree branches illustrate group means of phenotypic variation for each trait expressed as deviation from the overall mean, which is here set equal to the horizontal line. The provided color coding of the 11 groups (G1 to G11) is used in e and provenances selected by MTSI are labelled. The two provenances in white (32 and 158) were excluded prior to MRT analysis
Multivariate regression tree analysis constrained by geographic variables revealed 11 groups explaining 58% of the total variance (Fig. 3d). The resulting groups, reflecting regional tendencies in growth performance and quality, contain two to 16 provenances. The first and second split separate the poor performing provenances towards the northern and southern margins from the core of the species distribution, with each split accounting for more than 10% of the variance. The leaf charts at the nodes of the dendrogram show the means for groups with similar phenotypic expression in height, basal area and stem form. The first group to the left (G1 in Fig. 3d, e), for example, representing 13 provenances from the north (≥ 51.82°N), is characterized by height and basal area worse than average and an intermediate stem form. Within the southern partition (< 46.44°N), two provenances from elevations > 1025 m in Italy and Slovenia further segregate (G3), performing better than the six provenances originating from southern France and Spain (G2). Subsequent splits within the central belt between 46.4 to 51.8°N are primarily longitudinal, first segregating at 9.77°E, approximately the central meridian of Germany. The group east of 9.77°E splits further, resulting in four groups, where five provenances of particularly good stem form, but poor growth from the Bavarian Forest and the Ore Mountains (G5) segregate from an intermediate group between 9.77°E and 11.59°E (G4). The group expanding from 13.91°E towards the east is further split at an elevation of < 375 m. Provenances sourced from the lower elevations (G7) grow better than those from higher elevations (G6). Towards the right of the dendrogram three provenances (39, 75, 71) combining outstanding growth with a stem form better than average (G11) split apart from a relatively large group compromising western Germany and northern France. This again divides into three groups, first at an elevation of 425 m resulting in a group of average performance and low quality from the lower elevations (G8), while the better performing higher elevation provenances again segregate, approximately at border between Germany (G9) with Belgium and France (G10).
Selection of suitable provenances
Using the defined selection intensity of 20%, 17 out of 85 provenances are selected. Concentrating at individual sites (selection by MGIDI), this leads to a positive selection differential in Schädtbek, i.e. an improvement of the mean of the selected provenances compared to the site mean for all traits, while it is not possible to improve height, basal area and stem form at the same time in Wesel and Malter (Table 4). For the sake of better growth potential, the proportion of trees with an acceptable stem form is reduced by 4.5% (Malter) and 9.5% (Wesel).
Twelve of the provenances selected in Schädtbek, thirteen in Wesel and nine in Malter originate from Germany, mainly concentrating in the Central uplands (Table S4). Five of them are selected at all sites, namely Malchin (32), Seelzerthurm (39), Monschau (48), Schmallenberg (54) and Spangenberg (75), and eight on at least two sites. In Wesel and Malter provenances from North-Rhine Westphalia are frequently selected.
Combining performance and stability of multiple traits—height, basal area and stem form—in the MTSI index, the following 17 provenances are selected as most suitable across sites: nine German provenances, namely Malchin (32), Seelzerthurm (39), Monschau (48), Eitorf (52), Schmallenberg (54), Wünnenberg Glashütte (58), Neuenheerse (61), Spangenberg (75), Osburg (87), Elmstein-Süd, Rfö.Wolfsgrube (91) and Münsingen Brente (95); the French F.D. de Planoise (13); two Polish Bnerków (116) and Henryków (118), two Slovakian Trenčín (130) and Zamutov (124) and the Ukrainian Svaljava Polana (141) (see their spatial location in Fig. 3e). These provenances primarily belong to the MRT groups with a high growth performance towards the right of the dendrogram: G7, G10 and G11 (Fig. 3d, e). Particularly outstanding and stable is the provenance Seelzerthurm (39) with the lowest MTSI of 0.065. The rank order of all 85 provenances according to the MTSI index is displayed in Figure S7.
Applying this selection at individual sites, the trade-off between growth traits and stem form is prevalent at all of them: height and basal area have a positive selection differential (5.2 to 17.9%), while stem form is negatively affected at all sites (Table 5). In Schädtbek and Malter, the group of provenances selected across sites achieves a higher average performance in basal area (Table 5) than those selected on each individual site (Table 4).
Discussion
Absence of provenance by environment interaction
For none of the assessed traits, significant P × E interactions were detected among the 85 provenances planted at three trial sites in Germany (Table 3). The absence of P × E interactions contrasts results of Alia et al. (2011), who found significant interactions for survival, height as well as DBH (measures of age 9 and 10), when considering 8, 12 or 20 trials of the International Beech Trial Series 1995/98. However, their sites showed considerably more environmental variation since they were distributed from the Pyrenees in Spain across Central Europe to the Carpathians in Romania. At a smaller spatial scale Stojnić et al. (2015a), also found significant interactions examining height growth at four sites located in the fragmented south east of the distribution (Bosnia, Croatia, Serbia). However, their site conditions ranged from 6.6 to 11.0°C in mean annual temperatures ranging and from 624 to 1240 mm in annual precipitation. Our three German environments experience rather low levels of abiotic stress and a good water availability throughout the year, with highly concordant amounts of precipitation (Table 1). The climatic variability among them is apparently too small to induce considerable rank changes in trait expression. We conclude from the absence of interactions that the three sites can be summarized to one mega-environment and extracted provenance means of trait expression simultaneously.
Accordingly, provenance performance is highly similar among sites (Fig. 2), with the better yielding provenances growing better across environments, and the poor performers growing consistently badly. Those provenances showing an environmental-independent growth are generalists with a highly consistent trait expression (Williams et al. 2002). Those with a large range among sites appear adapted to specific environmental conditions and may be considered specialists. However, only few of them, for example provenances 84, 93, 94, 97 (Fig. 2) show a basal area above average at one or two sites and a poor performance on the remaining. The effect of these specialists appears too small to result in an overall significant interaction term.
The high frequency of generalists in forest field trials appears a common observation for growth traits, which Matheson and Raymond (1986) explained by the longevity of trees. Forest trees are required to grow for many years, and successful genotypes must be well-adapted to changing seasons and variable selective forces. In their thorough review on tropical tree species, they concluded that provenances growing well on favourable sites do likewise cope well with unfavorable sites, as long as there was no major environmental stress. This was recently shown in an extensive provenance trial series with Norway spruce. Transfer functions for height growth had a highly consistent shape at over 30 environments across Europe with outstanding provenances originating from the Hercyno-Carpathian domain, despite for the northernmost harsh environment in Sweden, where local provenances did perform best (Liepe et al. 2022). The reason lays in the considerable phenotypic plasticity allowing trees to physiologically or morphologically respond to changing environments (Kramer 1995).
Ecotypic variation across species range
Considerable variation exists between provenances of beech ranging from 21% (stem form) to 45% (basal area) on single sites and from 20 to 39% across sites (Tables 2 and 3). On the landscape level, this results in distinct patterns with spatially adjacent provenances showing a similar trait expression, which could be segregated into meaningful groups using MRT analysis constrained by geographic predictor variables (Fig. 3). We interpret these 11 groups as ‘ecotypes’ shaped by local adaptation to environmental conditions at provenance origin, a term first proposed by Turesson (1922), referring to groupings of populations in relation to a type of habitat or climate. Mátyás (2004) further specified it as a population adapted to local site conditions that occur in patches rather than as clinal variation, which was highlighted by various studies on intraspecific adaptation of beech (Jazbec et al. 2007; Ivanković et al. 2008; Mátyás et al. 2009; von Wühlisch 2012; Popović et al. 2021).
From the left to the right, the 11 ecotypes at the final leaves of the dendrogram represent the following major European regions: the North European Plains (G1) at the northern range margin; Basque Mountains, Pyrenees, Massif Central and Eastern Slovenia at the southern margin (G2); high elevation sources from the southern Alps experiencing particularly high precipitation (G3); a transition zone in southern Germany ranging from the Harz Mountains down south to Bavaria, separating subatlantic (west) and subcontinental (east) climate influences (G4); higher elevation sources from Ore Mountains and Bavarian Forest (G5); two groups covering Sudetes and Carpathians, one > 375 m (G6), the other < 375 m (G7); a low elevation ecotype covering south-western Germany and northern France (G8); and in the same area two ecotypes from > 425 m, one being concentrated in Germany (G10), basically the center of the species distribution and the other ranging approximately from the border between Germany with Belgium and France to the Mountains of Morvan (G9); and finally the three most outstanding provenances located in central Germany (G11).
Drawing a conclusive picture of the adaptive portfolio that each ecotype developed in response to the climate at their origin (Table S5), requires additional traits, including bud phenology and ecophysiological traits. These traits were not directly considered here, but all together cumulate in the economically important growth traits, representing the most frequently measured proxies for fitness (Alberto et al. 2013). The length of the available growing season, for example, defined by the timing of budbreak and leaf senescence, represents the major driver of photosynthetic activity and as result growth (Gömöry and Paule 2011; Garate-Escamilla et al. 2020). As a trait of strong genetic control (Kramer et al. 2017), budbreak determines provenance response to climatic change, here induced by the transfer to the testing environments, representing a shift to warmer climates for the majority of ecotypes (Fig. S8). The chilling and forcing requirements of budbreak are relatively well understood (Garate-Escamilla et al. 2020), e.g. low elevation provenances from Atlantic climates in the north west developed high heat sum requirements to avoid an early flushing imposed by frequent occurrence of forcing temperatures above 0 °C, as opposed to those from the continental south east with a low heat sum requirement to react quickly to the fast seasonal change in spring. The high heat sum requirements are reflected in the poor performance of the ecotype from the Northern plains (G1), while the ecotype compromising the low elevations of Sudetes and Carpathians (G7), profits from an extended growing season under the favorable conditions in Germany due to its early budbreak (von Wühlisch et al. 1995). If a consideration of bud phenology in the MRT were possible, this could aid to further refine the presented grouping. For example, by ruling out the misleading grouping of the provenance from Eastern Slovenia together with those from the south western range margin. All together, they have a highly comparable growth behaviour, but the provenance from Eastern Slovenia breaks bud early, while those from Southern France and Spain break bud late (von Wühlisch et al. 1995).
The ecotypes from central highlands in Germany (G9, G10, G11) possess a highly suitable adaptive portfolio for the growing conditions at the three trial sites, resulting in particularly good growth performance. Interestingly, G9 and G10 split at an elevation of 425 m, which closely resembles the altitudinal limit between the German regions of provenance 810 07/08 and 810 09/10 (Anonymous 1994). Geographically, each pair is assigned one specific area, but within applies an altitudinal border set to 400 or 500 m, depending on the ecological unit.
Considering the expected increase in drought frequency and severity, investigations of ecophysiological traits (anatomic, hydraulic and foliar traits) are emerging to better understand the strategies of water use. The number of studied traits is extensive, however, limited to a much smaller number of provenances (e.g. 6 by Robson et al. 2012, 10 by Hajek et al. 2016, 11 by Stojnić et al. 2015b). At our site in Schädtbek, Hajek et al. (2016) investigated 22 ecophysiological traits, of which they found only three to differ significantly among provenances, namely the xylem pressure inducing 88% loss of hydraulic conductance, leaf size and vessel density. The high adaptive capacity in xylem function of beech appeared to be predominantly a consequence of vessel density, which closely correlates with growth (height, as well as above ground biomass) (Hajek et al. 2016), underlining the suitability of growth performance for provenance selection.
As to be expected from the absence of provenance by environment interactions, the spatial patterns derived from single trait analysis, show similar overall tendencies as described here (Figs. S4–S6). The theorem that ‘local is best’, frequently assumed as a priori for the delineation of seed zones (O’Neill and Gómez-Pineda 2021), does not apply for the three beech trials analysed here. Locally adjacent provenances in Schädtbek (Fig. S4) and Malter (Fig. S6) are clearly outperformed by others, while it may to some extent be confirmed for the site in Wesel (Fig. S5), with various good performing provenances at close proximity.
Trade-off between growth and stem form
The main goals of tree breeding are to increase growth, adaptability and economic value of the wood finally to be harvested. Simultaneous selection for multiple traits is essential to produce desired responses for all traits of interest. In the case of beech, presented here, this is, however, counteracted by the trade-off between growth (height, basal area and also DBH, Fig. S3) and stem form (Fig. 3). Considering each individual trait separately, the best provenances (20%) for height and basal area are highly concordant, but almost completely different to the best in stem form (Fig. 3a, b vs. c). Combining the three traits in a multi-trait selection index to select the most suitable provenances across sites, growth will be improved but to the disadvantage of a poorer stem form (Table 5). When concentrating the selection at individual trial sites rather than all three at the same time, it appears possible to improve all traits in Schädtbek, while the trade-off is prevalent in Wesel and Malter (Table 3).
Such trade-offs between growth and quality traits are actually not uncommon. Schermann et al. (1997) observed that a selection for fast growth in 9-year-old families of coastal Douglas fir indirectly increased the frequency of stem defects and Woeste et al. (2021) reported a trade-off between volume and the retention of large limbs for red oak. It may be argued that a high growth potential to some extent outweighs a minor quality, since a purposeful silvicultural tending harbours substantial means of improvement towards stem straightness.
Stem form quality, generally of particular interest in tree breeding, is a rather difficult trait. First, it is based on a qualitative assessment with a scoring scheme rather than a quantitative measurement. That said, the given scores are largely dependent on the subjective view of the observer, which were different persons at each site. Second, there seems to be a tendency that trees from higher elevations, mostly being thinner, are given a better score, which has been described before for other provenance trials with beech (Rau et al. 2015). Larger, pre-growing trees occupy more space to make use of the available resources. They tend to have larger crowns, thicker branches and likely split into more than one main axis.
Apart from the potential observer bias, the preconditions of the trial sites need to be stressed, since they are likely a reason for the overall poor quality. All three trials were established on former agricultural land. Such sites show substantial differences in chemical and physical soil properties compared to forest sites (e.g. higher nutrient content, absence of humus layer), with negative effects on young beech stands (Cukor et al. 2022). Further, the wider spacing may have a negative effect on individual tree quality. With 5000 trees/ha the planting density was lower than the high stocking recommended for comparable afforestations with 7000 to 10,000 trees/ha (according to forest funding guidelines of federal states in Germany, e.g. Anonymous 2020) to create conditions to develop good quality and minimize the risk of wolf trees (Liziniewicz 2009). Due to the absence of a shelter, be it a remaining overstory or a the planting of fast growing nurse species, the sites were prone to late frost damage, which represents a frequent threat to young beech cultivations and can significantly alter tree quality (Wagner et al. 2010). Such silvicultural elements are frequent management praxis for beech, but difficult to implement in the experimental setup with the goal to assess provenance differentiation.
Concluding remarks
Our study highlights the potential benefits in productivity that can be expected using the most suitable reproductive material for reforestations with beech in the central-northern part of the range. The environmental variation among the three sites investigated appears too small to induce provenance by environment interactions resulting in a great advantage for forest practitioners: the three trials may be summarized to one mega-environment and provenance recommendations developed jointly. Using multivariate regression tree analysis, we identified eleven ecotypes of similar trait expression, being much larger than the currently used delineations of provenance regions by the European member states. Still, they closely resemble some aspects of these delineations, e.g. an elevational segregation between 400 and 500 m in Western Germany.
Concentrating on the individual provenance level, the best 20% with high value for tree breeding and deployment have been identified. Several highly suitable seed sources concentrate in the Central Uplands of Hesse, North-Rhine Westphalia and Rhineland-Palatine in Germany and herein in the national provenance regions 810 07, 810 08 and 810 09 (Anonymous 1994). Since these provenances have proven to be stable across all three sites, they appear suitable to be planted across a wider range of environments in the Northern half of Germany that are silviculturally appropriate for beech. In addition, provenances from the Western Carpathians (< 325 m) appear suitable candidates for assisted migration.
Data availability
Data will be made available upon request.
Code availability
Code will be made available upon request.
References
Aitken SN, Bemmels JB (2016) Time to get moving: assisted gene flow of forest trees. Evol Appl 9:271–290. https://doi.org/10.1111/eva.12293
Alberto FJ, Aitken SN, Alía R et al (2013) Potential for evolutionary responses to climate change—evidence from tree populations. Glob Chang Biol 19:1645–1661. https://doi.org/10.1111/gcb.12181
Alexandrov A, Giannini R, Parnuţa G et al (2011) Ecology of European beech, its phyto-sociological characteristics, silviculture and conservation strategy. In: von Wühlisch G, Alia R (eds) Genetic resources of European Beech (Fagus sylvatica L.) for sustainable forestry. Proceedings of the COST E52 Final Meeting (4–6 May 2010, Burgos, Spain). Monografias Inia: Serie Forestal–Madrid: Torrejen de Ardoz. No. 22-2011. INIA, pp 19–26
Alexandru A-M, Mihai G, Stoica E, Curtu AL (2023) Multi-trait selection and stability in Norway Spruce (Picea abies) Provenance trials in Romania. Forests 456:14. https://doi.org/10.3390/f14030456
Alia R, Bozic G, Gömöry D et al (2011) The survival and performance of beech provenances over a Europe-wide gradient of climate. In: von Wühlisch G, Alia R (eds) Genetic Resources of European Beech (Fagus sylvatica L.) for Sustainable forestry. Proceedings of the COST E52 final meeting (4–6 May 2010, Burgos, Spain). Monografias Inia: Serie Forestal–Madrid: Torrejen de Ardoz. No. 22–2011. INIA, pp 115–126
Anonymous (1994) Forstvermehrungsgut-Herkunftsgebietsverordnung (FoVHgV) [Regulation on provenance regions for forest reproductive material]. Verordnung über Herkunftsgebiete für forstliches Vermehrungsgut vom 7.10.1994 (BGBl. I S. 3578), geändert durch die Verordnung vom 15.1.2003 (BGBl. I S. 238)
Anonymous (2020) Richtlinie über die Gewährung von Zuwendungen zur Förderung forstwirtschaftlicher Maßnahmen im Land, Anlage 4 - Pflanzenzahlen je Hektar Pflanzfläche Niedersachsen. [Funding guidelines to promote forest management, Annex 4 - Plant numbers per hectare in Lower Saxony]. RdErl. d. ML v. 1. 12. 2020 - 406–64030/1–2.6/2–1 -
Arend M, Link RM, Zahnd C et al (2022) Lack of hydraulic recovery as a cause of post-drought foliage reduction and canopy decline in European beech. New Phytol 234:1195–1205. https://doi.org/10.1111/nph.18065
BMEL (2019) Deutschlands Wald im Klimawandel - Eckpunkte und Maßnahmen [German forest under climate change - Key points and actions]. Berlin, Germany
Bohn U, Gollub G (2010) Buchenwälder als natürliche Vegetation in Europa [Beech forests as natural vegetation in Europe]. Natur Landsch 82:391–397
Bolte A, Czajkowski T, Kompa T (2007) The north-eastern distribution range of European beech—a review. Forestry 80:413–429. https://doi.org/10.1093/forestry/cpm028
Bolte A, Ammer C, Löf M et al (2009) Adaptive forest management in central Europe: climate change impacts, strategies and integrative concept. Scand J For Res 24:473–482. https://doi.org/10.1080/02827580903418224
Boshier D, Broadhurst L, Cornelius J et al (2015) Is local best? Examining the evidence for local adaptation in trees and its scale. Environ Evid 4:1–10. https://doi.org/10.1186/s13750-015-0046-3
Christophe C, Birot Y (1983) Genetic structures and expected genetic gains from multitrait selections in wild populations of Douglas fir and Sitka spruce. II Practical application of index selection on several populations. Silvae Genet 32:173–181
Cukor J, Vacek Z, Vacek S et al (2022) Biomass productivity, forest stability, carbon balance, and soil transformation of agricultural land afforestation: a case study of suitability of native tree species in the submontane zo. CATENA 210:105898. https://doi.org/10.1016/j.catena.2021.105893
de Cuyper B, Bastien J, Paques L et al (2011) D2.1-Common protocols and reference standards for selected traits and species TREES4FUTURE-Designing Trees for the Future
Czúcz B, Gálhidy L, Mátyás C (2011) Present and forecasted xeric climatic limits of beech and sessile oak distribution at low altitudes in Central Europe. Ann For Sci 68:99–108. https://doi.org/10.1007/s13595-011-0011-4
De’ath G (2002) Multivariate regression trees: a new technique for modeling species-environment relationships. Ecology 83:1105–1117. https://doi.org/10.1890/0012-9658(2002)083[1105:MRTANT]2.0.CO;2
Diers M, Weigel R, Leuschner C (2022) Both climate sensitivity and growth trend of European beech decrease in the North German Lowlands, while Scots pine still thrives, despite growing sensitivity. Trees. https://doi.org/10.1007/s00468-022-02369-y
Ding H, Chiabai A, Silvestri S, Nunes PALD (2016) Valuing climate change impacts on European forest ecosystems. Ecosyst Serv 18:141–153. https://doi.org/10.1016/j.ecoser.2016.02.039
Ellenberg H (1988) Vegetation ecology of Central Europe, 4th edn. Cambridge University Press, Cambridge
EUFORGEN (2008) Distribution map of beech (Fagus sylvatica). www.euforgen.org/species/fagus-sylvatica/
Frei ER, Gossner MM, Vitasse Y et al (2022) European beech dieback after premature leaf senescence during the 2018 drought in northern Switzerland. Plant Biol 24:1132–1145. https://doi.org/10.1111/plb.13467
Fréjaville T, Vizcaíno-Palomar N, Fady B et al (2020) Range margin populations show high climate adaptation lags in European trees. Glob Chang Biol 26:484–495. https://doi.org/10.1111/gcb.14881
Gapare WJ, Ivković M, Liepe KJ et al (2015) Drivers of genotype by environment interaction in radiata pine as indicated by multivariate regression trees. For Ecol Manage 353:21–29. https://doi.org/10.1016/j.foreco.2015.05.027
Gárate-Escamilla H, Hampe A, Vizcaíno-Palomar N et al (2019) Range-wide variation in local adaptation and phenotypic plasticity of fitness-related traits in Fagus sylvatica and their implications under climate change. Glob Ecol Biogeogr 28:1336–1350. https://doi.org/10.1111/geb.12936
Garate-Escamilla H, Brelsford C, Hampe A et al (2020) Greater capacity to exploit warming temperatures in northern populations of European beech is partly driven by delayed leaf senescence. Agric For Meteorol 284:107908. https://doi.org/10.1016/j.agrformet.2020.107908
Gömöry D, Paule L (2011) Trade-off between height growth and spring flushing in common beech (Fagus sylvatica L.). Ann For Sci 68:975–984. https://doi.org/10.1007/s13595-011-0103-1
Hajek P, Kurjak D, von Wühlisch G et al (2016) Intraspecific variation in wood anatomical, hydraulic, and foliar traits in ten European beech provenances differing in growth yield. Front Plant Sci 7:1–14. https://doi.org/10.3389/fpls.2016.00791
Hartmann K, Krois J, Waske B (2018) E-Learning Project SOGA: statistics and geospatial data analysis. Freie Universitaet Berlin, Department of Earth Sciences
Ivanković M, Bogdan S, Božič G (2008) Varijabilnost visinskog rasta obične bukve (Fagus sylvatica L.) u testovima provenijencija u hrvatskoj i sloveniji [European beech (Fagus sylvatica L.) height growth variability in Croatian and Slovenian provenance trials]. Sumar List 132:529–541
Jactel H, Bauhus J, Boberg J et al (2017) Tree diversity drives forest stand resistance to natural disturbances. Curr For Reports 3:223–243. https://doi.org/10.1007/s40725-017-0064-1
Jazbec A, Šegotić K, Ivanković M et al (2007) Ranking of European beech provenances in Croatia using statistical analysis and analytical hierarchy process. Forestry 80:151–162. https://doi.org/10.1093/forestry/cpm007
Kaiser HF (1958) The varimax criterion for analytic rotation in factor analysis. Psychometrika 23:187–200
Knoke T, Ammer C, Stimm B, Mosandl R (2008) Admixing broadleaved to coniferous tree species: a review on yield, ecological stability and economics. Eur J For Res 127:89–101. https://doi.org/10.1007/s10342-007-0186-2
Knutzen F, Dulamsuren C, Meier IC, Leuschner C (2017) Recent climate warming-related growth decline impairs European beech in the center of its distribution range. Ecosystems 20:1494–1511. https://doi.org/10.1007/s10021-017-0128-x
Kormann JM, Liesebach M, Liepe KJ (2023) Provenances from introduced stands of Northern Red Oak (Quercus rubra L.) outperform those from the natural distribution. For Ecol Manage 531:120803. https://doi.org/10.1016/j.foreco.2023.120803
Krahl-Urban J (1958) Vorläufige Ergebnisse der Buchenprovenienzversuche [Preliminary results of beech provenance trials]. Allg Forst Jagd 129:242–251
Kramer K (1995) Phenotypic plasticity of the phenology of seven European tree species in relation to climatic warming. Plant Cell Environ 18:93–104. https://doi.org/10.1111/j.1365-3040.1995.tb00356.x
Kramer K, Ducousso A, Gömöry D et al (2017) Chilling and forcing requirements for foliage bud burst of European beech (Fagus sylvatica L.) differ between provenances and are phenotypically plastic. Agric For Meteorol 234–235:172–181. https://doi.org/10.1016/j.agrformet.2016.12.002
Langlet O (1971) Two hundred years genecology. Taxon 20:653–721
Liepe KJ, Hamann A, Smets P et al (2016) Adaptation of lodgepole pine and interior spruce to climate: implications for reforestation in a warming world. Evol Appl 9:409–419. https://doi.org/10.1111/eva.12345
Liepe KJ, van der Maaten E, van der Maaten-Theunissen M, Liesebach M (2022) High phenotypic plasticity, but low signals of local adaptation to climate in a large-scale transplant experiment of Picea abies (L.) Karst. in Europe. Front for Glob Chang 5:1–15. https://doi.org/10.3389/ffgc.2022.804857
Liesebach M (2015) The international network of European beech trials—the base to study intra-specific variation. Dendroflora of Central Europe–utilization of knowledge in research, education and practice. Technicka Univerzita vo Zvolene, Zvolen, pp 169–187
Liesebach M, Liepe KJ, Šeho M et al (2023) Ergebnisse aus dem Internationalen Buchenherkunftsversuch 1996/98 von den Versuchsflächen in Deutschland [Results from German test sites of the international beech provenance trial 1996/98]. Thünen Rep 105:97–127
Lindner M, Maroschek M, Netherer S et al (2010) Climate change impacts, adaptive capacity, and vulnerability of European forest ecosystems. For Ecol Manage 259:698–709. https://doi.org/10.1016/j.foreco.2009.09.023
Liziniewicz M (2009) The development of beech in monoculture and mixtures. Thesis, Swedish University of Agricultural Sciences
Madsen S (1985) Progeny trials with approved Danish beech seed stands. Mitteilungen BFH 150:92–98
Marchi M, Castellanos-Acuña D, Hamann A et al (2020) ClimateEU, scale-free climate normals, historical time series, and future projections for Europe. Sci Data 7:1–10. https://doi.org/10.1038/s41597-020-00763-0
Martinez del Castillo E, Zang CS, Buras A et al (2022) Climate-change-driven growth decline of European beech forests. Commun Biol 5:1–9. https://doi.org/10.1038/s42003-022-03107-3
Matheson A, Raymond C (1986) A review of provenance x environment interaction: its practical importance and use with particular reference to the tropics. Commonw For Rev 65:283–302
Mátyás C (2004) Genetics and genetic resources/population, conservation and ecological genetics. In: Evans J, Burley J, Youngquist JA (eds) Encyclopedia of Forest Sciences. Academic Press, pp 188–197
Mátyás C, Božič G, Gömöry D et al (2009) Transfer analysis of provenance trials reveals macroclimatic adaptedness of european beech (Fagus sylvatica L.). Acta Silv Lignaria Hungarica 5:47–62
Müller NA, Geßner C, Mader M et al (2023) Genomic variation of a keystone forest tree species reveals patterns of local adaptation and future maladaptation. bioRxiv 2023.05.11.540382. https://doi.org/10.1101/2023.05.11.540382
O’Neill GA, Gómez-Pineda E (2021) Local was best: sourcing tree seed for future climates. Can J For Res 51:1432–1439. https://doi.org/10.1139/cjfr-2020-0408
Olivoto T, Lúcio ADC (2020) metan: an R package for multi-environment trial analysis. Methods Ecol Evol 11:783–789. https://doi.org/10.1111/2041-210X.13384
Olivoto T, Nardino M (2021) Genetics and population analysis MGIDI: toward an effective multivariate selection in biological experiments. Bioinformatics 37:1383–1389. https://doi.org/10.1093/bioinformatics/btaa981
Olivoto T, Lúcio ADC, da Silva JAG et al (2019) Mean performance and stability in multi-environment trials II: Selection based on multiple traits. Agron J 111:2961–2969. https://doi.org/10.2134/agronj2019.03.0221
Ouellette M-H, Legendre P (2013) MVPARTwrap: Additional features for package mvpart. R package version 0.1–9.2. https://CRAN.R-project.org/package=MVPARTwrap
Pâques LE (ed) (2013) Forest tree breeding in Europe current state-of-the-art and perspectives. Springer, Dordrecht
Popović V, Lučić A, Rakonjac L (2021) Variability of morphological traits of European beech (Fagus sylvatica L.) seedlings in Serbia. South-East Eur For 12:83–89. https://doi.org/10.15177/SEEFOR.21-06
R Core Team (2019) A language and environment for statistical computing. R Foundation for Statistical Computing. https://www.r-project.org/
Ræbild A, Hansen CP, Kjaer ED (2002) Statistical analysis of data from provenance trials. Guidel Tech Notes No 63:27
Rau HM, Rumpf H, Schönfelder E (2015) Neue Ergebnisse aus den Buchen-Herkunftsversuchen von Krahl-Urban [New results from the Krahl-Urban provenance trials with European beech]. Forstarchiv 86:27–41. https://doi.org/10.4432/0300-4112-86-27
Robson TM, Sánchez-Gómez D, Cano FJ, Aranda I (2012) Variation in functional leaf traits among beech provenances during a Spanish summer reflects the differences in their origin. Tree Genet Genomes 8:1111–1121. https://doi.org/10.1007/s11295-012-0496-5
Robson MT, Benito-Garzón M, Miranda RA et al (2018) Phenotypic trait variation measured on european genetic trials of Fagus sylvatica L. Sci Data 5. https://doi.org/10.1038/sdata.2018.149
Rukh S, Sanders TGM, Krüger I et al (2023) Distinct responses of European beech (Fagus sylvatica L.) to drought intensity and length—a review of the impacts of the 2003 and 2018–2019 drought events in Central Europe. Forests 14:1–24. https://doi.org/10.3390/f14020248
Schermann N, Adams WT, Aitken SN, Bastien JC (1997) Genetic parameters of stem form traits in a 9-year-old coastal Douglas-fir progeny test in Washington. Silvae Genet 46:166–170
Stojnić S, Orlović S, Ballian D et al (2015a) Provenance by site interaction and stability analysis of European beech (Fagus sylvatica L.) provenances grown in common garden experiments. Silvae Genet 64:133–147. https://doi.org/10.1515/sg-2015-0013
Stojnić S, Orlović S, Miljković D et al (2015b) Provenance plasticity of European beech leaf traits under differing environmental conditions at two Serbian common garden sites. Eur J For Res 134:1109–1125. https://doi.org/10.1007/s10342-015-0914-y
Teissier du Cros E (1993) Study of beech variability in France. In: Muhs H-J, von Wühlisch G (eds) The scientific basis for the evaluation of the genetic resources of beech (Proceedings), pp 131–143
Muhs H, von Wühlisch G (eds) (1993) The scientific basis for the evaluation of the genetic resources of beech. Proceedings of an EC workshop. Ahrensburg, 1993
Therneau TM, Atkinso B, Ripley B et al (2013) Package “mvpart”. R package version 1.6–0
Turesson G (1922) The species and the variety as ecological units. Hereditas 3:100–113
Ujvári-Jármay É, Nagy L, Mátyás C (2016) The IUFRO 1964/68 inventory provenance trial of Norway spruce in Nyírjes, Hungary - results and conclusions of five decades. Acta Silv Lignaria Hungarica 12:177. https://doi.org/10.1515/aslh-2016-0001
von Wühlisch G (2012) Anpassungsfähigkeit von Buchen and den Klimawandel [Adaptability of beech to climate change]. AFZ Wald 13:22–24
von Wühlisch G, Krusche D, Muhs HJ (1995) Variation in temperature sum requirement for flushing of beech provenances. Silvae Genet 44:343–346
Wagner S, Collet C, Madsen P et al (2010) Beech regeneration research: from ecological to silvicultural aspects. For Ecol Manage 259:2172–2182. https://doi.org/10.1016/j.foreco.2010.02.029
White TL, Adams WT, Neale DB (2007) Forest genetics. CABI Publishing, Oxfordshire UK, Cambridge USA
Williams ERER, Matheson ACAC, Harwood CECE (2002) Experimental design and analysis for tree improvement, 2nd edn. CSIRO Publishing, Collingwood, Australia
Woeste KE, Pike CC, Warren JC, Coggeshall MV (2021) Characterization of stem volume and form tradeoffs in a northern red oak (Quercus rubra) breeding population in early stages of selection. Ann For Sci 78. https://doi.org/10.1007/s13595-021-01084-x
Zas R, Merlo E, Díaz R, Fernández-López J (2003) Stability across sites of Douglas-fir provenances in Northern Spain. For Genet 10:71–82
Acknowledgements
We gratefully acknowledge the continuous measurements conducted by our technicians and nursery staff. The presented analysis is only possible through the long-term maintenance of the sites for which we thank Ralf Boettcher (forest district Ostholstein, Bundesforsten), Georg Cuppenbender (forest district Hochwald), Martin Rogge and Marius Erley (Department of Forest Genetics and Forest Reproductive Material), all three of Wald & Holz North-Rhine Westfalia and Steffen Seyfert (forest district Baerenfels, Karsdorf).
Funding
Open Access funding enabled and organized by Projekt DEAL. This research was in part conducted during the project ‘BucheTIG’ funded by the German Federal Ministry of Food and Agriculture and the Federal Ministry of the Environment, Nature Conservation, Nuclear Safety and Consumer Protection following a decision by the German Bundestag (Waldklimafonds, Grant No. 2219WK60A4 and 2219WK60B4). Seed source collection and establishment of the trial sites were funded by the Concerted Action of the Commission of the European Communities (AAAIR3 Programme, Grant No. CT94-2091).
Author information
Authors and Affiliations
Contributions
KJL and ML conceived and designed the study and carried out data curation. KJL performed the analysis and prepared the original draft. EM and MM provided critical feedback and helped to shape research and manuscript. JMK contributed to the analysis. HW provided curated data for one site. All authors reviewed the article and gave their approval to the final version to be published.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Communicated by Oliver Gailing.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Heino Wolf: Retired.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liepe, K.J., van der Maaten, E., van der Maaten-Theunissen, M. et al. Ecotypic variation in multiple traits of European beech: selection of suitable provenances based on performance and stability. Eur J Forest Res 143, 831–845 (2024). https://doi.org/10.1007/s10342-024-01656-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-024-01656-2