Abstract
Biodiversity loss is a significant problem at a global scale and may be amplified by climate change. In recent years, coniferous forests have had substantial dieback across Europe due to drought and subsequent bark-beetle outbreaks. As many studies on the consequences of disturbance and subsequent management have focused on natural stands, management implications for managed spruce stands are not well understood, even though such stands are widespread throughout Europe. In this study, beetle taxonomy, conservation value, and community composition are compared among spruce plantations and four post-disturbance management approaches: standing deadwood, lying deadwood, clear cuts, and long-term succession. Diversity and community composition differed significantly among management categories, while different beetle families responded similarly. Intact spruce stands harbored the lowest beetle diversity while the highest taxonomic diversity and conservation value was on clear cuts and stands with lying or standing deadwood. The proportion of forest specialists was highest in successional forests. In summary, different forest management categories harbored distinct beetle communities at the family-, species-, and ecological guild levels. Therefore, post-disturbance management should consider the landscape scale and include different management types. This enhances landscape heterogeneity and thus overall biodiversity but could also mitigate negative impacts of natural disturbances on ecosystem services.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The increasing loss of biodiversity receives considerable attention and, worldwide, is critical (Barnosky et al. 2011; Cardinale et al. 2012; Dirzo et al. 2014). Various studies have shown substantial decreases in taxonomic diversity and biomass of different taxa (Dirzo et al. 2014; Newbold et al. 2015; Hallmann et al. 2017; Sánchez-Bayo and Wyckhuys 2019; Seibold et al. 2019). Particularly under climate change, habitat loss and fragmentation are important drivers of biodiversity loss (Fahrig 2003; Bellard et al. 2012). These also affect European forests, which have experienced massive diebacks especially in spruce stands in recent years. Forests cover ca. 30% of the world’s terrestrial area (FAO 2020) and support a substantial proportion of global biodiversity (Brodribb et al. 2020). Moreover, they provide important ecosystem services and resources for human societies (Seidl et al. 2014; Morris et al. 2016; Muys et al. 2022).
Trees have comparatively long lifespans, and consequently forests can only slowly adapt to environmental changes (Brodribb et al. 2020). Anthropogenic climate change will thus substantially affect forest ecosystems through increasing frequencies of wildfires, storms, droughts, and pest outbreaks (Seidl et al. 2017). This is predicted to have drastic impacts (IPCC 2018, 2022), for example on timber production in Europe (Seidl et al. 2014). However, the effects on biodiversity may be beneficial or detrimental, depending on the nature of the disturbance, management interventions, and the organisms under consideration (Thorn et al. 2016). Disturbance may promote ecosystem renewal, create habitat heterogeneity, and thus enhance biodiversity (White and Pickett 1985; Müller et al. 2008; Swanson et al. 2011; Senf et al. 2020). Accordingly, various studies have reported positive effects of natural disturbances in forests on different taxa (Moretti et al. 2004; Winter et al. 2015; Beudert et al. 2015; Kortmann et al. 2021, 2022). However, at least partially negative impacts may be expected in the future due to changes in disturbance amplitudes, frequencies, or interactions between different disturbance types (Seidl et al. 2014, 2017; Thom and Seidl 2016; Thom et al. 2017). Post-disturbance interventions in managed forests may also affect forest biodiversity (Thorn et al. 2014, 2018; Seibold 2015; Uhl et al. 2022).
In recent years, coniferous forests of the northern hemisphere have suffered severe diebacks. Germany is heavily affected by spruce (Picea abies, (L.) Karst) dieback as the species accounts for one quarter of the national forest area (BMEL 2021). Norway spruce is planted here far beyond its natural range, which makes it even more vulnerable to natural disturbances (Overbeck and Schmidt 2012; Hlásny et al. 2017). Such stands are often salvage-logged to mitigate economic losses and the potential for further damage (Sessions et al. 2004; Prestemon et al. 2006; Kausrud et al. 2012; Müller et al. 2019; Muys et al. 2022). However, the consequences of different management interventions on biodiversity remain largely unknown (Hlásny et al. 2017; Thorn et al. 2018; López-Bedoya et al. 2021).
Within the current biodiversity crisis, insect declines may be particularly problematic due to their multiple ecosystem functions (Hallmann et al. 2017; Sánchez-Bayo and Wyckhuys 2019). Since beetles and their ecological functions are highly diverse, they make good bioindicators (Gerlach et al. 2013). In forests, they are particularly important for ecosystem functioning and resilience (Lange et al. 2014; Homburg et al. 2019). For example, beetles serve as prey, seed dispersers, decomposers, predators, and ecosystem engineers (Müller et al. 2008; López-Bedoya et al. 2021). Depending on their ecological niche, they may respond differently to forest management. In addition, managed spruce stands are generally considered to have rather low biodiversity (Magura et al. 2000; Morris et al. 2016; Hlásny et al. 2017; Zumr et al. 2021). For example, saproxylic species typically decline in managed forests due to the removal of deadwood (Grove 2002; Cobb et al. 2011; Seibold 2015; Thorn et al. 2015; Zumr et al. 2021). Overall, conventional forest management and salvage logging often result in habitat homogenization and may therefore lead to biodiversity declines (Thorn et al. 2014, 2018). On the other hand, beetle species independent of deadwood might be less affected or even benefit from forest management (Pearce and Venier 2006; Koivula and Spence 2006; Thorn 2016). Thus, forest management also may substantially affect insect diversity in the current period of forest dieback (Stephens and Wagner 2007; Thorn et al. 2018; Horák et al. 2019; López-Bedoya et al. 2021).
In this study, the effects of forest management on beetle diversity in spruce stands in western Germany were examined. Under consideration were stands with (1) live spruce trees, (2) standing deadwood (i.e., left unmanaged after dieback), (3) lying deadwood (bark beetle infested trees were felled but left on site), (4) clear cuts where spruce trees were cut and removed, and (5) secondary succession stands. The latter were left unmanaged (no deadwood-removal, no planting) after a windthrow in 1990 to assess long-term trends of set-aside stands. Note however, that the disturbance type (windthrow) differed from the other categories (bark beetle outbreak). We focus on the following questions: (1) How do different post-disturbance management approaches affect the alpha-diversity of beetles in general as well as of different taxonomic groups? (2) How are endangered species affected by post-disturbance management and what kind of management yields the highest benefits for conservation? (3) How does community composition (species composition and functional niches) differ among management categories? By investigating these three questions, the aim is to better understand which management is best suited to maintain beetle diversity in post-disturbance spruce plantations.
Materials and methods
Study area
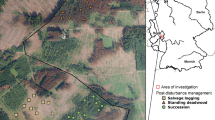
For this study, managed forest stands of the forestry office Hachenburg (50° 65′ N, 7° 82′ E) in the Westerwald mountain range in western Germany were investigated (Fig. 1). The sites were located 3–7 km northwest of the town Hachenburg at 237 to 381 m a.s.l. This area is dominated by forests (45.7%) and agricultural lands (37.3%). Settlements (8.1%) and infrastructure (5.2%) play only a minor role (Statistisches Landesamt Rheinland-Pfalz 2020). The soils mainly consist of brownsoils and pseudogleys on devonian clay-, silt-, and sandstones (Landesamt für Geologie und Bergbau Rheinland-Pfalz 2013a, b). The annual mean temperature is 9.5 °C with an average precipitation of 855 mm per year (averages since 1994 and 2011, respectively; Agrarmeteorologie Rheinland-Pfalz 2022a, b). Due to the unusually dry years 2018–2020, the area was strongly affected by spruce diebacks (Ministerium für Umwelt Energie Ernährung und Forsten Rheinland-Pfalz 2020, 2021).
Field methods
Beetle (Coleoptera) diversity was evaluated within (1) stands with live spruce trees (hereafter ‘spruce’) as well as four post-disturbance management categories: (2) standing deadwood (stands with dead spruce that had not been managed after tree death caused by bark beetle outbreaks), (3) lying deadwood (stands in which dead trees were felled but left in place without further management, and no bark-treatment was applied and no timber removed), (4) clear cuts (former spruce stands that were felled and cleared after bark beetle outbreaks, all timber removed, no reforestation), and (5) succession areas (former spruce stands left unmanaged, no deadwood removed, no reforestation after a windthrow in 1990, thus allowing for natural succession; hereafter termed ‘succession’). The latter category was chosen to assess the long-term development of set-aside stands, even though the disturbance type differed from the other post-disturbance management categories. Thus all categories consisted of either former or current commercially managed spruce plantations. For each management category, ten replicate sites were chosen (Fig. 1; Table S1, Supplementary material).
Beetles were caught with two different trap types. To assess ground beetle diversity, pitfall traps were established using 125 mL PET cups filled with 60 mL of a 70% water and 30% monopropylene glycol mixture. Two traps were buried on each site 10 m apart, each with a protective plastic roof, for a total of 100 traps. In addition, a flight-interception trap was installed in the center of each plot to cover the diversity of flight-active beetles (Leather 2005). Traps were built according to instructions in Schmidl (2005) and consisted of two transparent 60 cm × 40 cm plastic discs at a right angle, a cover as rain protection, and a funnel at the bottom connected to a 1000 mL PET-bottle filled half with saline solution as trapping liquid. Flight-interception traps were installed 110 cm above the ground. Traps were emptied every two weeks between 16th of April and 20th of July (pitfall traps) and between 28th of April and 20th of July 2021 (flight interception traps). After sorting, all beetles were stored in 70% ethanol until identification using taxonomic keys (Freude et al. 1983; Lompe 2002).
Data collection
Several parameters comparing alpha diversity, species conservation and community composition among management categories were calculated (Table 1). To assess alpha diversity, total numbers of individuals and species were used as well as the effective species number for each site. The latter represents the actual species diversity, being a derivate of the Shannon–Wiener index (Jost 2006). To account for potential differences in detection probability among sites, the rarefied species richness was further estimated for a sample coverage of 90% using the iNEXT package (Hsieh et al. 2016) in R (version 4.0.3; RStudio Team 2020).
For species conservation, the ‘conservation value’ was calculated (Görn and Fischer 2011) for each site by assigning a point score to each species according to its status in the Red List of Germany (Geiser 1998; least concern = 1, data deficient/rare = 2, near-threatened = 4, vulnerable = 16, endangered = 25, critically endangered = 36). The conservation value was calculated as the sum of all point scores per site. The numbers of Red List individuals and species were also calculated for each site as the sum of all individuals or species belonging to the Red List categories.
To investigate differences in community composition, Bray–Curtis dissimilarities among different management types were calculated based on abundance data (vegan-package, R). In addition, the distributions of the five most abundant beetle families and species among management categories were compared by calculating the respective percentages of individuals for every site. To assess different distributions in ecological niches among management types, all species were allocated to categories with regard to their preference for moisture (hygrophilic, hemihygrophilic, mesophilic, xerophilic), food (xylophagous, phytophagous, mycetophagous, zoophagous, omnivorous), habitat type (forest stenotopic, forest eurytopic, open stenotopic, open eurytopic, ubiquitous), and habitat niche (deadwood, living wood, herb-layer, litter-layer, ground-dwelling) (Koch 1996). The relative abundance of each ecological guild was used for every site for comparison. Additionally, species preferences for specific management types were assessed for species with ≥ 50 individuals. A preference was assumed if ≥ 50% of all specimens were found in a specific management category.
Because of its overabundance within the pitfall traps (13,297 individuals), Anoplotrupes stercorosus (Geotrupidae) was excluded from the total number of individuals, effective number of species, rarefied species richness, and relative abundances of ecological guilds of this trap type.
Statistical analyses
To test for significant variation across management categories with regards to the total numbers of individuals and species, the effective number of species, rarefied species richness, and conservation values for both trap types, as well as the Red List individuals and species of flight-interception traps, linear mixed effect models (LME; nlme-package in R; Pinheiro and Bates 2020) with management type as fixed and forest area (Fig. 1) as random factor were performed. Forest areas were included to account for possible similarities among sites within the same forest area, possibly leading to more similar beetle communities. Due to multiple testing, a sequential Bonferroni-Holm correction was performed. To ensure normal distribution, 16 parameters had to be LN-transformed and seven square-root-transformed (Table 2 and 3). Subsequently, Tukey HSD tests were used to identify significant differences among categories (glht function; multcomp-package in R; Hothorn et al. 2008). Moran’s I tests (ape-package in R; Paradis and Schliep 2019) were also performed to account for spatial clustering among sampling sites (Dormann et al. 2007; Bivand and Wong 2018), but were non-significant throughout.
To determine differences in community composition among management categories, permutational multivariate analyses of variance (PERMANOVA, permutations = 999) were used based on the Bray–Curtis dissimilarity index and subsequent pairwise PERMANOVAS to locate significant differences. PERMANOVA rather than LME was used due to difficulties in normalizing data. Results were visualized with non-metric multidimensional scaling (NMDS) analyses. If not otherwise stated, analyses were conducted using the vegan-package (Oksanen et al. 2020) in R (R Core Team 2020).
Results
Sampling
A total of 29,862 beetles belonging to 495 species and 77 families were collected (Table S2). Of these, 21,221 individuals of 188 species and 43 families were caught with pitfall traps, and 8,641 individuals of 395 species and 74 families with flight-interception traps. By far the most abundant species was A. stercorosus, the dor beetle, a species of dung beetle, accounting for 13,303 individuals (44.6% of all individuals). In total, the most abundant families with more than one species were Carabidae (11.8% of all individuals), Elateridae (10.2%), Scolytidae (8.5%), Staphylinidae (5.9%), and Curculionidae (3.5%).
Alpha-diversity
Considering all beetles, management had a significant influence on all alpha-diversity measures (Fig. 2; Table 2). However, patterns differed among trap types. For flight-interception traps, clear cuts had the highest values for all parameters, followed by sites with lying deadwood. Spruce stands had the lowest values (except from individual numbers), while standing deadwood and succession sites had an intermediate position for most parameters. For pitfall traps, clear cuts and sites with standing deadwood had the highest values except for the number of individuals where spruce stands had the highest number. Spruce or lying deadwood had the lowest values here.
a Number of individuals, b number of species, c effective number of species and d rarefied species richness across post-disturbance management categories (colors) for all beetles caught with pitfall traps (solid colored boxes) and flight-interception traps (hatched boxes). Boxplots display the interquartile range (25–75%; box), median (line within box), as well as maximum and minimum values (whiskers); different letters above boxplots indicate significant differences among management types (Tukey HSD after linear mixed effect models; lower case letters for pitfall traps, upper case letters for flight-interception traps)
Similar patterns were found when individual families were analysed separately, even though the specific differences among management types differed slightly between trap types and beetle families (Table S3). Flight-interception traps gave significant results for four out of five families (Table 2), largely consistent with the overall pattern with highest scores on clear cuts and lying deadwood sites. Curculionoidea (Fig. S1) were the only family for which only pitfall traps showed significant results. The highest number of individuals was found in spruce stands while the effective number of species and rarefied species richness were higher on clear-cut sites. The diversity of ground-living Carabidae (Fig. S2) was also highest on clear cuts, followed by stands with lying or standing deadwood. Elateridae (Fig. S3) caught with pitfall traps had the lowest values in spruce stands throughout, while all other management categories showed enhanced diversity values. For Staphylinidae (Fig. S4), only the number of individuals found in pitfall traps differed significantly and was lowest on clear-cut sites.
Species conservation
Twenty-five species noted in the German Red List (Geiser 1998) were sampled, including one rare, three data-deficient, eleven near-threatened, seven vulnerable, two endangered (Hydroporus cf. rufifrons, one in spruce stands, one on clear cuts; Ampedus cinnabarinus, one on a lying deadwood site), and one critically endangered species (Melolontha pectoralis, two individuals on lying deadwood sites; Table S2, Supplementary material).
The type of forest management had a significant effect on species conservation (Table 3; Fig. 3). The conservation value found with pitfall traps was highest in standing deadwood sites followed by clear cuts, and lowest in sites with lying deadwood. In contrast, the conservation value of flight-interception traps was significantly higher on clear-cut and lying deadwood sites compared to other management categories (Fig. 3a). Red List individuals (Fig. 3b) and species (Fig. 3c) from flight-interception traps also significantly increased on clear cuts and sites with lying deadwood compared to live spruce and standing deadwood stands.
a Conservation value, b Red List individuals, and c Red List species across post-disturbance management categories (colors) for all beetles caught with pitfall traps (solid boxes) and flight-interception traps (hatched boxes). Boxplots display the interquartile range (25–75%; box), median (line within box), as well as maximum and minimum values (whiskers); different letters above boxplots indicate significant differences among management types (Tukey HSD after linear mixed effect models; lower case letters for pitfall traps, upper case letters for flight-interception traps)
Community composition
Species assemblages strongly differed among forest management categories (Table 4), as revealed by Bray–Curtis dissimilarities (Fig. 4; Table 5). For pitfall traps, all assemblages differed significantly among each other except those from lying deadwood which only differed from spruce stands (Table 5a). For flight-interception traps, all assemblages differed significantly (Table 5b).
Non-metric multidimensional scaling (NMDS) analysis of beetle assemblages for five management categories: live spruce, standing deadwood, lying deadwood, clear cut, and succession (n = 10 plots each, 2 dimensions, Bray Curtis distance); divided according to pitfall traps (a; tries = 54; ordination based on 7925 individuals of 187 species) and flight-interception traps (b; tries = 20; ordination based on 8641 individuals of 395 species)
Forest management significantly affected the occurrence of the most common families and species of beetles (Table 4; Fig. 5). Spruce stands had the highest proportions of Geotrupidae and Curculionidae (pitfall traps) as well as of Scolytidae (flight-interception traps). The highest abundances of A. stercorosus, Exomias araneiformis (pitfall traps), and Ips typographus (flight-interception traps) were found in live spruce stands. Standing deadwood sites harbored most family members of the Cholevidae and most individuals of Sciodrepoides watsoni (pitfall traps) or Athous subfuscus (Elateridae; flight-interception traps). Sites with lying deadwood had the highest shares of Staphylinidae (pitfall traps) as well as most specimens of Crypturgus subcribosus (Scolytidae; flight-interception traps). Clear cuts were inhabited mostly by Carabidae (pitfall traps), as well as by Elateridae, Staphylinidae and Cerambycidae (flight-interception traps). At species level, clear cuts also had the highest relative abundance of Pityogenes chalcographus (Scolytidae; flight-interception traps). Succession stands had the highest number of Abax parallelepipedus and A. parallelus (both Carabidae; pitfall traps) and the highest proportions of Trixagus leseigneuri and its family Throscidae (flight interception traps).
Relative abundance of the five most common families caught with a flight-interception traps and c pitfall traps as well as the five most abundant species (b, d; respectively) across post-disturbance management categories. Different letters behind bars indicate significant differences among categories (PERMANOVA pairwise comparisons)
The type of forest management significantly affected the relative abundance of beetles with different preferences for moisture and habitat type (Table S4). Additionally, preferences for food and habitat niches differed significantly for flight-interception traps (Table 4; Table S5, Fig. 6). Succession sites harboured the highest proportion of hygrophilic individuals, while standing deadwood sites showed the highest number of hemihygrophilic beetles (flight-interception traps; Fig. 6a). Standing deadwood sites and clear cuts showed a comparatively high proportion of xerophilic beetles (pitfall traps; Fig. 6c). Food preferences differed strongly among management categories, with the proportion of xylophagous and mycetophagous beetles being highest in spruce stands, phytophagous beetles on clear cuts, and zoophagous beetles on succession sites (flight-interception traps; Fig. 6b). Habitat type preferences also differed significantly among management types (Fig. 6g,e). Eurytopic forest species were the dominant ecological group in all management types. On clear cut sites, open landscape species became more common (20–30% of all individuals). Forest management also shaped the composition of individuals with different habitat niche preferences for flight-interception traps (Fig. 6f). Deadwood-dependent beetles were most abundant in spruce and succession stands, tree layer beetles in spruce stands, and herb layer, litter, and ground layer beetles on clear cuts.
Relative abundance of species with different preferences for moisture, food, habitat type, and habitat niche (Koch 1989–1996) across management categories caught with flight interception traps (a, b, e, f; respectively) and pitfall traps (c, d, g, h; respectively). Different letters behind bars indicate significant differences among categories (PERMANOVA pairwise comparison)
In addition, there were habitat preferences of seven species for spruce stands, three for sites with standing and one with lying deadwood, nine for clear cuts and three for succession areas (Table S6, Supplementary material).
Discussion
Our results show that beetle taxonomic diversity (Fig. 2), conservational parameters (Fig. 3), and community composition (Figs. 4–6) differ strongly among management categories. In addition, there were differences between trap types, while differences between specific beetle families were small (Table S3; Figs. S1–S5).
Alpha-diversity
Overall, the highest taxonomic diversity was in clear cuts as well as lying deadwood or, depending on trap type, standing deadwood sites. Live spruce plantations, in contrast, showed the lowest beetle diversity which agrees with other studies (Horák et al. 2019; López-Bedoya et al. 2021). Spruce forests mainly consist of row-planted trees of the same age and thus lack habitat heterogeneity (Lange et al. 2014). They are therefore often species-poor and sometimes referred to as ‘green deserts’ (Horák et al. 2019). Natural spruce forests, on the other hand, support high levels of biodiversity (Thorn et al. 2016), and dieback of large coniferous forest patches may have severe cascading effects on higher trophic levels (Conner et al. 1991; Koprowski et al. 2005). Moreover, such diebacks are often perceived negatively in society (Kortmann et al. 2021) and the concomitant deforestation of large areas raised debates among conservationists, politicians and foresters in recent years (Morris et al. 2016; Hlásny et al. 2017, 2021; Kortmann et al. 2021).
By comparing intact spruce plantations with several post-disturbance management approaches, we show that spruce dieback outside its natural range can be an opportunity for enhanced biodiversity, by substantially increasing alpha diversity. In fact, natural disturbances have been reported to promote habitat heterogeneity and concomitantly, biodiversity (White and Pickett 1985; Swanson et al. 2011; Thom and Seidl 2016; Thorn et al. 2016; Senf et al. 2020; Uhl et al. 2023). By creating so-called disturbance legacies, uniform structures are disrupted, light penetration is enhanced, and the amount of dead wood increased (Müller et al. 2008). The latter is especially a rare resource in most managed forests and limits the occurrence of saproxylic beetles (Seibold et al. 2016b; Doerfler et al. 2018; Uhl et al. 2022). By creating forest gaps with more ground vegetation, higher solar-radiation, and deadwood supply, I. typographus may act as a valuable keystone species (Grove 2002; Müller et al. 2008; Thorn et al. 2014).
Nevertheless, managed spruce stands that are affected by bark beetles are frequently salvage-logged to mitigate economic losses and the potential for further damage (Sessions et al. 2004; Prestemon et al. 2006; Kausrud et al. 2012; Müller et al. 2019; Muys et al. 2022). In contrast to other studies (e.g., Thorn et al. 2018), our results showed that clear cuts, where salvage logging was performed, harboured a rich beetle diversity regardless of trap type or beetle family. Salvage logging is often considered to have negative effects on biodiversity because it removes disturbance legacies like deadwood and habitat heterogeneity, which is especially detrimental for saproxylic organisms (Cobb et al. 2011; Thorn et al. 2014, 2016, 2018; Leverkus et al. 2018; García-Tejero et al. 2018; Hagge et al. 2019a). On the other hand, clear cuts were found to enhance the diversity of carabid beetles (De Warnaffe and Lebrun 2004; Pearce and Venier 2006; Vele et al. 2011) and other arthropods (Purchart et al. 2013). Particulary highly mobile species may forage on open patches, indicated by the comparatively high proportions of phytophagous and herb-layer associated individuals caught with flight-interception traps on clear cuts. In general, however, numerous forest species depend on forest edges (Wermelinger et al. 2007; Müller et al. 2008) and may therefore benefit from salvage logging. Our study area consisted of rather small forest patches of different management forms and composition, such that the high diversity found on our clear-cut sites may be partly due to edge and spill-over effects (Magura 2002; Lacasella et al. 2015).
In contrast to salvage logging, deadwood is often reported to enhance biodiversity (e.g., Seibold et al. 2016a; Doerfler et al. 2018; Thorn et al. 2020), but to date, few studies have investigated how different types of deadwood affect biodiversity in managed forests (Vogel et al. 2020). Studying European beech (Fagus sylvatica L.) forests, Rothacher et al. (2023) found positive effects of both deadwood types for saproxylic as well as non-saproxylic beetles, and Uhl et al. (2022) observed an increased diversity of saproxylic beetles on lying deadwood compared to standing deadwood. Our study suggests that these findings can also be applied to spruce forests. Accordingly, beetle diversity in general (Fig. 2), as well as that of different families (Figs. S1–S5, Supplementary material) was significantly elevated in stands with standing (pitfall traps) and especially lying (flight-interception traps) deadwood. Thus, our results emphasize that canopy openness, along with deadwood enrichment are very important drivers for beetle diversity (Vele et al. 2011; Seibold et al. 2016a, 2016b; Vogel et al. 2020; Uhl et al. 2022; Rothacher et al. 2023).
In addition, deadwood can also have positive effects on forest restoration, for example, by protecting young seedlings from browsing (Hagge et al. 2019b). This may facilitate natural succession on disturbed sites, potentially resulting in species-rich and climate-resilient forests (Swanson et al. 2011; Lehnert et al. 2013; Pukkala 2018; Sommerfeld et al. 2021). Surprisingly though, the positive effect of succession areas on alpha-diversity was not as pronounced as expected, which might be explained by the fact that the positive effects of natural disturbances vanish over time as canopies close again (Swanson et al. 2011; Lehnert et al. 2013). Nevertheless, succession sites had a higher taxonomic diversity compared to managed spruce stands especially for Elateridae (Fig. S3), but also for other beetles, even though the results did not always differ significantly.
Species conservation
Forest management also significantly affected species conservation, with clear cuts and deadwood sites performing best (Fig. 3a). Although salvage logging normally results in the loss of deadwood resources that are particularly important for the survival of saproxylic organisms (Thorn et al. 2018; Hagge et al. 2021), tree stumps left behind after salvage logging may promote beetle diversity (Rothacher et al. 2023). However, they may only promote saproxylic species for a short time and the relatively high value of clear-cut sites might also be due to spill-over effects. Thus, sites with larger deadwood ressources could provide more permanent habitats for saproxylic beetles (Rothacher et al. 2023). Such taxa are often classified in Red Lists due to their narrow habitat requirements (Seibold et al. 2015). Accordingly, most endangered species were caught with flight-interception traps on lying deadwood sites (Fig. 3b,c). To detect these rare species, this type of trap seems more suitable because many endangered saproxylic beetles are flight-active (Leather 2005). Many saproxylic (and often endangered) beetles prefer sun-exposed deadwood, as shown by Seibold et al. (2016b), and this was emphasized by the high number of Red List individuals and -species on our lying deadwood sites. In this study, both types of deadwood (standing and lying) were equally suitable to enhance beetle conservation (Fig. 3a). Accordingly, providing different larger deadwood types appears to be especially promising to promote (saproxylic) beetle diversity (Grove 2002; Vogel et al. 2020; Rothacher et al. 2023), as many Red List taxa prefer deadwood of large diameters and advanced decay stages to complete their life-cycles (Seibold et al. 2015; Hagge et al. 2021).
Community composition
Beetle functional diversity was also affected by forest management. Numerous studies have documented shifts in community composition from forest to open land species in stands where salvage logging was carried out (e.g., Koivula and Spence 2006; Lange et al. 2014; Thorn et al. 2018). Concomitantly, our study sites with open canopies, even when deadwood was left lying, had higher proportions of open land species compared to standing deadwood, succession, or spruce stands (Fig. 6e,g). The highest proportions of forest and hygrophilic species, however, were found on spruce and succession sites, while the highest proportion of xerophilic individuals was on clear cuts (Fig. 6a,c). In other studies, canopy cover was correlated with the proportion of forest species in carabid beetle communities (Koivula et al. 2002; Magura et al. 2006; Mullen et al. 2008). Our results suggest that this correlation might hold for beetles in general, which highlights the importance of forest patches with closed canopies as a refuge for forest and hygrophilic species. Therefore, former spruce stands could be set aside for long-term succession, which saves tree planting costs and may result in resilient forests and enhanced biodiversity in younger stages (Swanson et al. 2011; Lehnert et al. 2013; Sommerfeld et al. 2021). In summary, we found highly distinct beetle communities among management categories (Fig. 4), which reinforces the beneficial effects of management heterogeneity for promoting landscape-level biodiversity within managed forests (Schall et al. 2018, 2021; Bruun and Heilmann-Clausen 2021; Georgiev et al. 2022).
Conclusions and management implications
Spruce plantations face increasing stress from climate change and subsequent bark beetle outbreaks, especially when planted as monocultures beyond their natural range. Our study shows that the dieback of managed spruce stands can be a chance for enhanced biodiversity since overall, intact spruce sites had the lowest biodiversity compared with all other categories. Clear-cut sites supported high levels of biodiversity while allowing timber extraction. However, clear-cutting leads to a change of species composition from forest to open habitat specialists. Therefore, large-scale clear cuts should be avoided. Patches with closed canopies may serve as refugia for forest specialists and hygrophilic species. Such patches can be established by allowing long-term succession, which is cost-effective and may enhance biodiversity. Providing deadwood is an effective means to support natural succession and to increase biodiversity in managed forests, especially for saproxylic and endangered species. Further, our data suggest that different types of forest management harbor highly distinct beetle communities at the family, species, and ecological level. Thus, post disturbance management should target the landscape scale to include different management types. This not only enhances landscape heterogeneity and thus overall biodiversity, but could also mitigate negative impacts of natural disturbance on ecosystem services within managed forest landscapes.
References
Barnosky AD, Matzke N, Tomiya S, Wogan GO, Swartz B, Quental TB, Marshall C, McGuire JL, Lindsey EL, Maguire KC, Mersey B, Ferrer EA (2011) Has the earth’s sixth mass extinction already arrived? Nature 471(7336):51–57. https://doi.org/10.1038/nature09678
Bellard C, Bertelsmeier C, Leadley P, Thullier W, Courchamp F (2012) Impacts of climate change on the future of biodiversity. Ecol Lett 15(4):365–377. https://doi.org/10.1111/j.1461-0248.2011.01736.x
Beudert B, Bässler C, Thorn S, Noss R, Schröder B, Dieffenbach-Fries H, Foullois N, Müller J (2015) Bark beetles increase biodiversity while maintaining drinking water quality. Conserv Lett 8(4):272–281. https://doi.org/10.1111/conl.12153
Bivand RS, Wong DWS (2018) Comparing implementations of global and local indicators of spatial association. TEST 27(3):716–748. https://doi.org/10.1007/s11749-018-0599-x
BMEL (2021) [Bundesministerium für Ernährung und Landwirtschaft] Ergebnisse der Waldzustandserhebung 2021, 1–76 (in German)
Brodribb TJ, Powers J, Cochard H, Choat B (2020) Hanging by a thread? For Drought Sci 368(6488):261–266. https://doi.org/10.1126/science.aat7631
Bruun HH, Heilmann-Clausen J (2021) What is unmanaged forest and how does it sustain biodiversity in landscapes with a long history of intensive forestry? J Appl Ecol 58(9):1813–1816. https://doi.org/10.1111/1365-2664.13754
Cardinale BJ, Duffy JE, Gonzalez A, Hooper D, Perrings C, Venail P, Narwani A, MacE G, Timan D, Wardle D, Kinzig A, Daily G, Loreau M, Grace J, Larigauderie A, Srivastava D, Naeem S (2012) Biodiversity loss and its impact on humanity. Nature 486(7401):59–67. https://doi.org/10.1038/nature11148
Cobb TP, Morissette JL, Jacobs JM, Koivula MJ, Spence JR, Langor DW (2011) Effects of postfire salvage logging on deadwood-associated beetles. Conserv Biol 25(1):94–104. https://doi.org/10.1111/j.l523-17392010.01566.x
Conner RN, Rudolph DC, Kulhavy D, Snow A (1991) Causes of mortality of red-cockaded woodpecker cavity trees. J Wildl Manage 55(1):531–537
De Warnaffe GDB, Lebrun P (2004) Effects of forest management on carabid beetles in Belgium: implications for biodiversity conservation. Biol Conserv 118(2):219–234. https://doi.org/10.1016/j.biocon.2003.08.015
Dirzo R, Young HS, Galetti M, Ceballos G, Isaac NJB, Collen B (2014) Defaunation in the anthropocene. Science 345(6195):401–406. https://doi.org/10.1126/science.1251817
Doerfler I, Gossner MM, Müller J, Seibold S, Weiser WW (2018) Deadwood enrichment combining integrative and segregative conservation elements enhances biodiversity of multiple taxa in managed forests. Biol Conserv 228:70–78. https://doi.org/10.1016/j.biocon.2018.10.013
Dormann CF, McPherson JM, Araújo MB, Bivand R, Bolliger J, Carl G, Davies RG, Hirzel A, Jetz W, Kissling D, Kühn I, Ohlemüller R, Peres-Neto PR, Reineking B, Schröder B, Schurr FM, Wilson R (2007) Methods to account for spatial autocorrelation in the analysis of species distributional data: a review. Ecography 30:609–628. https://doi.org/10.1111/j.2007.0906-7590.05171.x
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515. https://doi.org/10.1146/annurev.ecolsys.34.011802.132419
FAO (2020) [Food and Agriculture Organization of the United Nations] Global forest resources assessment. https://fra-data.fao.org/WO/fra2020/home/ Accessed 15 Jun 2022
Freude H, Harde KW, Lohse GA (1983) Die Käfer Mitteleuropas. Vol. 1–11. Goecke & Evers Verlag, Krefeld, 3761 pages (in German)
García-Tejero S, Spence JR, O’Halloran J, Bourassa S, Oxbrough A (2018) Natural succession and clearcutting as drivers of environmental heterogeneity and beta diversity in North American boreal forests. PLoS ONE 13(11):e0206931. https://doi.org/10.1371/journal.pone.0206931
Geiser R (1998) Rote Liste der Käfer (Coleoptera). In: Bundesamt für Naturschutz (Hrsg): Rote Liste gefährdeter Tiere Deutschlands. Schriftenr Landschaftspflege Naturschutz 55: 178–179 (in German).
Georgiev KB, Bässler C, Feldhaar H, Heibl C, Karasch P, Müller J, Perlik M, Weiss I, Thorn S (2022) Windthrow and salvage logging alter β-diversity of multiple species groups in a mountain spruce forest. For Ecol Manage 520:120401. https://doi.org/10.1016/j.foreco.2022.120401
Gerlach J, Samways M, Pryke J (2013) Terrestrial invertebrates as bioindicators: an overview of available taxonomic groups. J Insect Conserv 17:831–850. https://doi.org/10.1007/s10841-013-9565-9
Görn S, Fischer K (2011) Niedermoore Nordostdeutschland bewerten. Nat Landsch 43(7):211–217 ((in German))
Grove SJ (2002) Saproxylic insect ecology and the sustainable management of forests. Annu Rev Ecol Syst 33:1–23. https://doi.org/10.1146/annurev.ecolsys.33.010802.150507
Hagge J, Leibl F, Müller J, Plechinger M, Soutinho JG, Thorn S (2019a) Reconciling pest control, nature conservation, and recreation in coniferous forests. Conserv Lett. https://doi.org/10.1111/conl.12615
Hagge J, Müller J, Bässler C, Biebl SS, Brandl R, Drexler M, Gruppe A, Hotes S, Hothorn T, Langhammer P, Stark H, Wirtz R, Zimmerer V, Mysterud A (2019b) Deadwood retention in forests lowers short-term browsing pressure on silver fir saplings by overabundant deer. For Ecol Manag. https://doi.org/10.1016/j.foreco.2019.117531
Hagge J, Müller J, Birkemoe T, Buse J, Bojesen Christensen RH, Gossner MM, Gruppe A, Heibl C, Jarzabek-Müller A, Seibold S, Siitonen J, Soutinho JG, Sverdrup-Thygeson A, Thorn S, Drag L (2021) What does a threatened saproxylic beetle look like? Modelling extinction risk using a new morphological trait database. J Anim Ecol 90(8):1934–1947. https://doi.org/10.1111/1365-2656.13512
Hallmann CA, Sorg M, Jongejans E, Siepel H, Hofland N, Schwan H, Stenmans W, Müller A, Sumser H, Hörren T, Goulson D, de Kroon H (2017) More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE. https://doi.org/10.1371/journal.pone.0185809
Hlásny T, Krokene P, Liebhold A, Montagné-Huck C, Müller J, Qin H, Raffa KJ, Schelhaas MJ, Seidl R, Svoboda M, Viiri H (2017) Living with bark beetles: impacts, outlook and management options. From Sci Policy 8:1–52. https://doi.org/10.36333/fs08
Hlásny T, König L, Krokene P, Lindner M, Montagné-Huck C, Müller J, Qin H, Raffa KF, Schelhaas M-J, Svoboda M, Viiri H, Seidl R (2021) Bark beetle outbreaks in Europe: state of knowledge and ways forward for management. Curr for Rep 7(3):138–165. https://doi.org/10.1007/s40725-021-00142-x
Homburg K, Drees C, Boutaud E, Nolte D, Schuett W, Zumstein P, von Ruschkowski E, Assmann T (2019) Where have all the beetles gone? Long-term study reveals carabid species decline in a nature reserve in Northern Germany. Insect Conserv Divers 12:268–277. https://doi.org/10.1111/icad.12348
Horák J, Brestovanská T, Mladenović S, Kout J, Bogusch P, Halda JP, Zasadil P (2019) Green desert?: Biodiversity patterns in forest plantations. For Ecol Manag 433:343–348. https://doi.org/10.1016/j.foreco.2018.11.019
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50(3):346–363. (package = "multcomp")
Hsieh TC, Ma KH, Chao A (2016) iNEXT: An R package vor interpolation and extrapolation of species diversity (Hill numbers). Methods Ecol Evol 7:1451–1456
IPCC (2018) [Intergovernmental Panel on Climate Change]. Summary for policymakers. In: Global warming of 1.5 °C. An IPCC special report on the impacts of global warming of 1.5 °C above pre-industrial levels and related global greenhouse gas emission pathways, in the context of strengthening the global response to the threat of climate change, sustainable development, and efforts to eradicate poverty.
IPCC (2022) [Intergovernmental Panel on Climate Change]. Climate change 2022: impacts, adaptation, and vulnerability. Contribution of working group II to the sixth assessment report of the intergovernmental panel on climate change.
Jost L (2006) Entropy and diversity. Oikos 113(2):363–375. https://doi.org/10.1111/j.2006.0030-1299.14714.x
Kausrud K, Økland B, Skarpaas O, Grégoire J-C, Erbilgin N, Stenseth NC (2012) Population dynamics in changing environments: the case of an eruptive forest pest species. Biol Rev 87(1):34–51. https://doi.org/10.1111/j.1469-185X.2011.00183.x
Koch K (1996) Die Käfer Mitteleuropas - Ökologie, Vol. 1–3. Goecke & Evers Verlag, Krefeld, 1211 pages (in German)
Koivula M, Spence JR (2006) Effects of post-fire salvage logging on boreal mixed-wood ground beetle assemblages (Coleoptera, Carabidae). For Ecol Manag 236(1):102–112. https://doi.org/10.1016/j.foreco.2006.09.004
Koivula M, Kukkonen J, Niemela J (2002) Boreal carabid-beetle (Coleoptera, Carabidae) assemblages along the clear-cut originated succession gradient. Biodivers Conserv 11:1269–1288
Koprowski JL, Alanen MI, Lynch AM (2005) Nowhere to run and nowhere to hide: response of endemic Mt. Graham red squirrels to catastrophic forest damage. Biol Conserv 126(4):491–498. https://doi.org/10.1016/j.biocon.2005.06.028
Kortmann M, Müller JC, Baier R, Bässler C, Buse J, Cholewińska O, Förschler MI, Georgiev KB, Hilszczański J, Jaroszewicz B, Jaworski T, Kaufmann S, Kuijper D, Lorz J, Lotz A, Łubek A, Mayer M, Mayerhofer S, Meyer S, Morinière J, Popa F, Reith H, Roth N, Seibold S, Seidl R, Stengel E, Wolski GJ, Thorn S (2021) Ecology versus society: impacts of bark beetle infestations on biodiversity and restorativeness in protected areas of Central Europe. Biol Conserv. https://doi.org/10.1016/j.biocon.2020.108931
Kortmann M, Roth N, Buse J, Hilszczański J, Jaworski T, Morinière J, Seidl R, Thorn S, Müller J (2022) Arthropod dark taxa provide new insights into diversity responses to bark beetle infestations. Ecol Appl. https://doi.org/10.1002/eap.2516
Lacasella F, Gratton C, De Felici S, Isaia M, Zapparoli M, Marta S, Sbordoni V (2015) Asymmetrical responses of forest and “beyond edge” arthropod communities across a forest–grassland ecotone. Biodivers Conserv 24:447–465. https://doi.org/10.1007/s10531-014-0825-0
Landesamt für Geologie und Bergbau Rheinland-Pfalz (2013a) Kartenviewer - Fachanwendungen und Fachthemen: Boden - BFD 50 - Bodenformengesellschaft. https://mapclient.lgb-rlp.de/?app=lgb&view_id=19. Accessed 28 Jun 2022 (in German)
Landesamt für Geologie und Bergbau Rheinland-Pfalz (2013b) Kartenviewer - Fachanwendungen und Fachthemen: Geologie: Geologische Übersichtskarte 1:300,000. https://mapclient.lgb-rlp.de/?app=lgb&view_id=19. Accessed 28 Jun 2022] (in German)
Lange M, Türke M, Pašalić E, Boch S, Hessenmöller D, Müller J, Prati D, Socher SA, Fischer M, Weisser WW, Gossner MM (2014) Effects of forest management on ground-dwelling beetles (Coleoptera; Carabidae, Staphylinidae) in Central Europe are mainly mediated by changes in forest structure. For Ecol Manage 329:166–176. https://doi.org/10.1016/j.foreco.2014.06.012
Leather SR (2005) Insect sampling in forest ecosystems. Blackwell Publishing, Oxford, p 303
Lehnert LW, Bässler C, Brandl R, Burton PJ, Müller J (2013) Conservation value of forests attacked by bark beetles: highest number of indicator species is found in early successional stages. J Nat Conserv 21(2):97–104. https://doi.org/10.1016/j.jnc.2012.11.003
Leverkus AB, Lindenmayer DB, Thorn S, Gustafsson L (2018) Salvage logging in the world’s forests: interactions between natural disturbance and logging need recognition. Global Ecol Biogeogr 27(10):1140–1154. https://doi.org/10.1111/geb.12772
Lompe A (2002) Die Käfer Europas - ein Bestimmungswerk im Internet. http://coleonet.de/coleo/index.htm. Accessed 12 Jan 2022 (in German)
López-Bedoya PA, Magura T, Edwards FA, Edwards DP, Rey-Benayas JM, Lövei GL, Noriega JA (2021) What level of native beetle diversity can be supported by forestry plantations? Glob Synth Insect Conserv Divers 14(16):736–747. https://doi.org/10.1111/icad.12518
LVermGeo (2021) [Landesamt für Vermessung und Geobasisinformation Rheinland-Pfalz]. Luftbilder Rheinland-Pfalz DOP40. In: Open Data - Digitale Orthophotos (DOP), Bodenauflösung 40cm. https://www.geoportal.rlp.de/mapbender/php/mod_inspireDownloadFeed.php?id=2b009ae4-aa3e-ff21-870b-49846d9561b2&type=SERVICE&generateFrom=wmslayer&layerid=61676. Accessed 27 Nov 2022] (in German)
Magura T (2002) Carabids and forest edge: spatial pattern and edge effect. For Ecol Manag 157:23–37
Magura T, Tóthmérész B, Bordán Z (2000) Effects of nature management practice on carabid assemblages (Coleoptera: Carabidae) in a non-native plantation. Biol Conserv 93:95–102
Magura T, Tóthmérész B, Elek Z (2006) Changes in carabid beetle assemblages as Norway spruce plantations age. Community Ecol 7(1):1–12. https://doi.org/10.1556/ComEc.7.2006.1.1
Ministerium für Umwelt Energie Ernährung und Forsten Rheinland-Pfalz (2020) WALDZUSTANDS-BERICHT 2020 (in German)
Ministerium für Umwelt Energie Ernährung und Forsten Rheinland-Pfalz (2021) WALDZUSTANDS-BERICHT 2021 (in German)
Moretti M, Obrist MK, Duelli P (2004) Arthropod biodiversity after forest fires: winners and losers in the winter fire regime of the southern Alps. Ecography 27:173–186
Morris JL, Cottrell S, Fettig CJ, Hansen WD, Sherriff RL, Carter VA, Clear JL, Clement J, DeRose RJ, Hicke JA, Higuera PE, Mattor KM, Seddon AWR, Seppa HT, Stednick JD, Seybold SJ (2016) Managing bark beetle impacts on ecosystems and society: priority questions to motivate future research. J Appl Ecol 54(3):750–760. https://doi.org/10.1111/1365-2664.12782
Mullen K, O’Halloran J, Breen J, Giller P, Pithon J, Kelly T (2008) Distribution and composition of carabid beetle (Coleoptera, Carabidae) communities across the plantation forest cycle—implications for management. For Ecol Manage 256(4):624–632. https://doi.org/10.1016/j.foreco.2008.05.005
Müller J, Bußler H, Goßner M, Rettelbach T, Duelli P (2008) The European spruce bark beetle Ips typographus in a national park: from pest to keystone species. Biodivers Conserv 17(12):2979–3001. https://doi.org/10.1007/s10531-008-9409-1
Müller J, Noss RF, Thorn S, Bässler C, Leverkus AB, Lindenmayer D (2019) Increasing disturbance demands new policies to conserve intact forest. Conserv Lett. https://doi.org/10.1111/conl.12449
Muys B, Angelstam P, Bauhus J, Bouriaud L, Jactel H, Kraigher H, Müller J, Pettorelli N, Pötzelsberger E, Primmer E, Svoboda M, Thorsen BJ, Van Meerbeek K (2022) Forest biodiversity in Europe. From Sci Policy 13:1–80
Newbold T, Hudson LN, Hill SLL, Contu S, Lysenko I, Senior RA, Börger L, Bennett DJ, Choimes A, Collen B, Day J, De Palma A, Díaz S, Echeverria-Londoño S, Edgar MJ, Feldman A, Garon M, Harrison MLK, Alhusseini T, Ingram DJ, Itescu Y, Kattge J, Kemp V, Kirkpatrick L, Kleyer M, Laginha Pinto Correia D, Martin CD, Meiri S, Novosolov M, Pan Y, Phillips HRP, Purves DW, Robinson A, Simpson J, Tuck SL, Weiher E, White HJ, Ewers RM, Mace GM, Scharlemann JPW, Purvis A (2015) Global effects of land use on local terrestrial biodiversity. Nature 520(7545):45–50. https://doi.org/10.1038/nature14324
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin P, O’Hara R, Simpson G, Solymos P, Stevens M, Szoecs E, Wagner H (2020) Vegan: community ecology package. R-package. https://CRAN.R-project.org/package=vegan
Overbeck M, Schmidt M (2012) Modelling infestation risk of Norway spruce by Ips typographus (L.) in the Lower Saxon Harz Mountains (Germany). For Ecol Manag 266:115–125. https://doi.org/10.1016/j.foreco.2011.11.011
Paradis E, Schliep K (2019) ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35(3):526–528. https://doi.org/10.1093/bioinformatics/bty633
Pearce JL, Venier LA (2006) The use of ground beetles (Coleoptera: Carabidae) and spiders (Araneae) as bioindicators of sustainable forest management: a review. Ecol Indic 6(4):780–793. https://doi.org/10.1016/j.ecolind.2005.03.005
Pinheiro J, Bates D (2020) Linear and nonlinear mixed effect models (nlme-package). R-package. https://CRAN.R-project.org/package=nlme
Prestemon JP, Wear DN, Stewart FJ, Holmes TP (2006) Wildfire, timber salvage, and the economics of expediency. For Policy Econ 8(3):312–322. https://doi.org/10.1016/j.forpol.2004.07.003
Pukkala T (2018) Effect of species composition on ecosystem services in European boreal forest. J for Res 29(2):261–272. https://doi.org/10.1007/s11676-017-0576-3
Purchart L, Tuf IH, Hula V, Suchomel J (2013) Arthropod assemblages in Norway spruce monocultures during a forest cycle—a multi-taxa approach. For Ecol Manag 306:42–51. https://doi.org/10.1016/j.foreco.2013.06.012
QGIS (2002) QGIS Development Team: QGIS Geographic Information System. Version 3.4.7—Madeira. Open Source Geospatial Foundation Project.
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for statistical computing, Vienna
Agrarmeteorologie Rheinland-Pfalz (2022a) Langjährige Mittelwerte Wetterstation Isert. https://www.dlr.rlp.de/Internet/AM/NotesAM.nsf/cc619a07ed6c4634c1257800003dca56/07804c9e9e2baf08c1257171002e8a40?OpenDocument&TableRow=2.1.1,2.6#2.1. Accessed 1 Feb 2022 (in German)
Agrarmeteorologie Rheinland-Pfalz (2022b) Langjährige Mittelwerte Hydrometeorologische Station Hachenburg. https://www.dlr.rlp.de/Internet/AM/NotesAM.nsf/cc619a07ed6c4634c1257800003dca56/ce8964b69eb243f7c12573ef002f58b1?OpenDocument&TableRow=2.4#2. Accessed 1 Feb 2022 (in German)
Rothacher J, Hagge J, Bässler C, Brandl R, Gruppe A, Müller J (2023) Logging operations creating snags, logs, and stumps under open and closed canopies promote stand-scale beetle diversity. For Ecol Manag. https://doi.org/10.1016/j.foreco.2023.121022
Sánchez-Bayo F, Wyckhuys KAG (2019) Worldwide decline of the entomofauna: a review of its drivers. Biol Conserv 232:8–27
Schall P, Gossner MM, Heinrichs S, Fischer M, Boch S, Prati D, Jung K, Baumgartner V, Blaser S, Böhm S, Buscot F, Daniel R, Goldmann K, Kaiser K, Kahl T, Lange M, Müller J, Overmann J, Renner SC, Schulze E-D, Sikorski J, Tschapka M, Türke M, Weisser WW, Wemheuer B, Wubet T, Ammer C (2018) The impact of even-aged and uneven-aged forest management on regional biodiversity of multiple taxa in European beech forests. J Appl Ecol 55(1):267–278. https://doi.org/10.1111/1365-2664.12950
Schall P, Heinrichs S, Ammer C, Ayasse M, Boch S, Buscot F, Fischer M, Goldmann K, Overmann J, Schulze E-D, Sikorski J, Weisser WW, Wubet T, Gossner MM (2021) Among stand heterogeneity is key for biodiversity in managed beech forests but does not question the value of unmanaged forests: response to Bruun and Heilmann-Clausen (2021). J Appl Ecol 58(9):1817–1826. https://doi.org/10.1111/1365-2664.13959
Schmidl J (2005) Entomologiebedarf-Eklektoren / FIT’s Rahn/Mini. In: Bioform-entomology & equipment. https://www.bioform.de/shop.php?wg=1&action=wgstart. Accessed 12 Apr 2023 (in German)
Seibold S, Brandl R, Buse J, Hothorn T, Schmidl J, Thorn S, Müller J (2015) Association of extinction risk of saproxylic beetles with ecological degradation of forests in Europe. Conserv Biol 29(2):382–390. https://doi.org/10.1111/cobi.12427
Seibold S, Bässler C, Baldrian P, Reinhard L, Thorn S, Ulyshen MD, Weiß I, Müller J (2016a) Dead-wood addition promotes non-saproxylic epigeal arthropods but effects are mediated by canopy openness. Biol Conserv 204:181–188. https://doi.org/10.1016/j.biocon.2016.09.031
Seibold S, Bässler C, Brandl R, Büche B, Szallies A, Thorn S, Ulyshen MD, Müller J (2016b) Microclimate and habitat heterogeneity as the major drivers of beetle diversity in dead wood. J Appl Ecol 53(3):934–943. https://doi.org/10.1111/1365-2664.12607
Seibold S, Gossner MM, Simons NK, Blüthgen N, Müller J, Ambarli D, Ammer C, Bauhus J, Fischer M, Habel JC, Linsenmair KE, Nauss T, Penone C, Prati D, Schall P, Schulze E-D, Vogt J, Wöllauer S, Weisser WW (2019) Arthropod decline in grasslands and forests is associated with landscape-level drivers. Nature 574(7780):671–674. https://doi.org/10.1038/s41586-019-1684-3
Seibold S (2015) Biodiversity of dead-wood dependent insects–effects of forest management and prospects of conservation. Dissertation, Wissenschaftszentrum Weihenstephan für Ernährung, Landnutzung und Umwelt, Technical University of Munich, 1–55
Seidl R, Schelhaas MJ, Rammer W, Verkerk PJ (2014) Increasing forest disturbances in Europe and their impact on carbon storage. Nat Clim Chang 4(9):806–810. https://doi.org/10.1038/nclimate2318
Seidl R, Thom D, Kautz M, Martin-Benito D, Peltoniemi M, Vacchiano G, Wild J, Ascoli D, Petr M, Honkaniemi J, Lexer MJ, Trotsiuk V, Mairota P, Svoboda M, Fabrika M, Nagel TA, Reyer CPO (2017) Forest disturbances under climate change. Nat Clim Chang 7(6):395–402. https://doi.org/10.1038/nclimate3303
Senf C, Mori AS, Müller J, Seidl R (2020) The response of canopy height diversity to natural disturbances in two temperate forest landscapes. Landsc Ecol 35(9):2101–2112. https://doi.org/10.1007/s10980-020-01085-7
Sessions J, Bettinger P, Buckman R, Newton M, Hamann J (2004) Hastening the return of complex forests following fire: the consequences of delay. J for 102(3):38–45
Sommerfeld A, Rammer W, Heurich M, Hilmers T, Müller J, Seidl R (2021) Do bark beetle outbreaks amplify or dampen future bark beetle disturbances in Central Europe? J Ecol 109(2):737–749. https://doi.org/10.1111/1365-2745.13502
Statistisches Landesamt Rheinland-Pfalz (2020) Flächennutzung Verbandsgemeinde Hachenburg. http://infothek.statistik.rlp.de/MeineHeimat/content.aspx?id=103&l=2&g=0714302&tp=1025. Accessed 1 Feb 2022 (in German)
Stephens SS, Wagner MR (2007) Forest plantations and biodiversity: a fresh perspective. J for 105(6):307–313. https://doi.org/10.1093/jof/105.6.307
Swanson ME, Franklin JF, Beschta RL, Crisafulli CM, DellaSala DA, Hutto RL, Lindenmayer DB, Swanson FJ (2011) The forgotten stage of forest succession: early-successional ecosystems on forest sites. Front Ecol Environ 9(2):117–125. https://doi.org/10.1890/090157
Thom D, Seidl R (2016) Natural disturbance impacts on ecosystem services and biodiversity in temperate and boreal forests. Biol Rev 91(3):760–781. https://doi.org/10.1111/brv.12193
Thom D, Rammer W, Seidl R (2017) The impact of future forest dynamics on climate: interactive effects of changing vegetation and disturbance regimes. Ecol Monogr 87(4):665–684. https://doi.org/10.1002/ecm.1272
Thorn S, Bässler C, Gottschalk T, Hothorn T, Bussler H, Raffa K, Müller J (2014) New insights into the consequences of post-windthrow salvage logging revealed by functional structure of saproxylic beetles assemblages. PLoS ONE. https://doi.org/10.1371/journal.pone.0101757
Thorn S, Bässler C, Bernhardt-Römermann M, Cadotte M, Heibl C, Schäfer H, Seibold S, Müller J (2015) Changes in the dominant assembly mechanism drive species loss caused by declining resources. Ecol Lett 19(2):163–170. https://doi.org/10.1111/ele.12548
Thorn S, Bässler C, Svoboda M, Müller J (2016) Effects of natural disturbances and salvage logging on biodiversity—lessons from the Bohemian Forest. For Ecol Manag 388:113–119. https://doi.org/10.1016/j.foreco.2016.06.006
Thorn S, Bässler C, Brandl R, Burton PJ, Cahall R, Campbell JL, Castro J, Choi C-Y, Cobb T, Donato DC, Durska E, Fontaine JB, Gauthier S, Hebert C, Hothorn T, Hutto RL, Lee E-J, Leverkus AB, Lindenmayer DB, Obrist MK, Rost J, Seibold S, Seidl R, Thom D, Waldron K, Wermelinger B, Winter M-B, Zmihorski M, Müller J (2018) Impacts of salvage logging on biodiversity: a meta-analysis. J Appl Ecol 55(1):279–289. https://doi.org/10.1111/1365-2664.12945
Thorn S, Seibold S, Leverkus AB, Michler T, Müller J, Noss RF, Stork N, Vogel S, Lindenmayer DB (2020) The living dead: acknowledging life after tree death to stop forest degradation. Front Ecol Environ 18(9):505–512. https://doi.org/10.1002/fee.2252
Thorn S (2016) Effects of salvage logging on biodiversity—ecological evidence for conservation-oriented management of natural disturbances. Dissertation, Wissenschaftszentrum Weihenstephan für Ernährung, Landnutzung und Umwelt, Technical University of Munich, 1–62
Uhl B, Krah FS, Baldrian P, Brandl R, Hagge J, Müller J, Thorn S, Vojtech T, Bässler C (2022) Snags, logs, stumps, and microclimate as tools optimizing deadwood enrichment for forest biodiversity. Biol Conserv. https://doi.org/10.1016/j.biocon.2022.109569
Uhl B, Pouska V, Červenka J, Karasch P, Bässler C (2023) Response of moth communities (Lepidoptera) to forest management strategies after disturbance. Eur J Entomol 120:35–41. https://doi.org/10.14411/eje.2023.005
Vele A, Holusa J, Frouz J, Konvicka O (2011) Local and landscape drivers of ant and carabid beetle communities during spruce forest succession. Eur J Soil Biol 47(6):349–356. https://doi.org/10.1016/j.ejsobi.2011.09.003
Vogel S, Gossner MM, Mergner U, Müller J, Thorn S (2020) Optimizing enrichment of deadwood for biodiversity by varying sun exposure and tree species: an experimental approach. J Appl Ecol 57(10):2075–2085. https://doi.org/10.1111/1365-2664.13648
Wermelinger B, Flückiger PF, Obrist MK, Duelli P (2007) Horizontal and vertical distribution of saproxylic beetles (Col., Buprestidae, Cerambycidae, Scolytinae) across sections of forest edges. J Appl Entomol 131(2):104–114. https://doi.org/10.1111/j.1439-0418.2006.01128.x
White SP, Pickett ST (1985) The ecology of natural disturbance and patch dynamics. Academic Press, New York
Winter MB, Ammer C, Baier R, Donato DC, Seibold S, Müller J (2015) Multi-taxon alpha diversity following bark beetle disturbance: evaluating multi-decade persistence of a diverse early-seral phase. For Ecol Manag 338:32–45. https://doi.org/10.1016/j.foreco.2014.11.019
Zumr V, Remeš J, Pulkrab K (2021) How to increase biodiversity of saproxylic beetles in commercial stands through integrated forest management in Central Europe. Forests. https://doi.org/10.3390/f12060814
Acknowledgements
We would like to thank the employees of the forestry office Hachenburg and all forest owners who allowed us to carry out the fieldwork in their stands and for helping us to put up the traps; especially Monika Runkel and Andreas Schäfer. Thanks also to Roland Busch and Rita Seul-Plath for help with trap preparation and to Thomas Wagner for help with identifing beetles. Thanks also to David Liesenfeld for help with field work and arthropod sorting.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was supported by Forschungsanstalt für Waldökologie und Forstwirtschaft (FAWF) of Landesforsten Rheinland-Pfalz (FF5.3-01-2021).
Author information
Authors and Affiliations
Contributions
EP: conceptualization, methodology, investigation, formal analysis, visualization, writing—original draft preparation, writing—review & editing; CT, JG, LG: Investigation; KF: Conceptualization, Methodology, Writing—Review & Editing, Supervision, Project administration, Funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest and no known competing financial interests or personal relationships that could have influenced the work reported in this paper.
Human and animal participants
To obtain the data presented in the manuscript it has been necessary to involve animals. Therefore, individuals had to be killed and removed from the field. However, all applicable international, national, and institutional guidelines for the care and use of animals were followed. No human participants were involved in the work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project Funding
This study was supported by Forschungsanstalt für Waldökologie und Forstwirtschaft (FAWF) of Landesforsten Rheinland-Pfalz (FF5.3- 01-2021).
The online version is available at http://www.springerlink.com
Corresponding editor: Tao Xu
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Plath, E., Trauth, C., Gerhards, J. et al. Dieback of managed spruce stands in western Germany promotes beetle diversity. J. For. Res. 35, 48 (2024). https://doi.org/10.1007/s11676-024-01695-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11676-024-01695-w