Abstract
Cassava (Manihot esculenta Crantz) is an essential stable food crop in Sub-Saharan Africa commonly consumed amongst the low-income communities in Africa. Though cassava roots and leaf have vast economic and commercial benefits, it produces cyanogenic glycosides, which are toxic and most often responsible for the bitter taste of some cassava cultivars. The study evaluates the cassava roots and leaves’ cyanogenic potential and dry matter content of the Genetic Gain Assessment trial grown in a different environment. It establishes the association between the cyanogenic potential (CNP) and the roots and leaves dry matter (DM). Genetic Gain Assessment (GGA) cassava genotypes (N = 400) selected for the Uniform Yield Trial (UYT) breeding stage were planted under IVS (Dry season in Inland Valley Hydromorphic area) and Upland (rain-fed conditions) in two locations of IITA Research Farms, namely; Ibadan (IVS and Upland) and Mokwa (Upland) in Nigeria. The CNP content of cassava leaves in IVS, Mokwa, and Upland ranged from 3.39 to 272.16 mg/100 g, 4.28 to 228.72 mg/100 g, and 13.13 to 127.39 mg/100 g, respectively. However, the respective CNP range in root samples across IVS, Mokwa, and Upland was 0.76–76.31 mg/100 g, 0.94–136.53 mg/100 g, and 2.37–47.11 mg/100 g. Also, the mean ± SD of DM content of leaves were 27.97 ± 3.01%, 28.81 ± 4.01%, and 13.65 ± 3.69%, respectively, in IVS, Mokwa, and Upland, while the root samples had mean ± SD of DM content of 38.09 ± 4.80%, 32.69 ± ,5.93% and 24.63 ± 5.07% respectively. Furthermore, location and genotype had a highly significant effect (p < 0.001) on the CNP and DM of roots and leaves. Also, linear regressions were established between CNP and DM of root and leaf with regression equation; DM-Root = 1.1999*DM-Leaf (r = 0.956) and CNP-Root = 0.29006*CNP-Leaf (r = 0.54). The relationship between the DM (root and leaf) and CNP (root and leaf) could serve as a valuable “inter-prediction” tool for these parameters.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Cassava (Manihot esculent Crantz) is a major perennial food crop that provides food for over 800 million people worldwide1. It is an energy source in many tropical countries, including Nigeria, and supplies about 70% of daily calories for more than 50 million people2. In Nigeria, cassava is considered a smallholder farmers’ favourite because it is available all year round, is tolerant to low soil fertility, and is resistant to diseases and pests3. About half a billion people in the tropical regions of Africa, Latin America, and Asia depend heavily on cassava roots for their diets; they are starch-rich (25–30%) but low in other nutrients like protein and vitamins4. Compared with other crops, cassava contributes largely to poverty alleviation by generating income for many households in SSA3. Though cassava has vast economic and commercial benefits, it produces cyanogenic glycosides; they are poisonous and cause some cassava varieties’ bitter flavour1. Cyanogen is a by-product of enzymatic hydrolysis of specific molecules present in cassava, such as linamarin, lostaustralin, and acetone cyanohydrin5. Linamarin is synthesized in the leaves through N-hydroxylation of isoleucine and valine and then transported to the roots6. It is known to be stored in the vacuoles of the plant and concentrated more in the leaves and root cortex than in the parenchyma of the plant7. Damage to cassava flesh during harvesting allows the reaction of Linamarin and linamarase to release acetone cyanohydrin, which, after decomposition, produces cyanide6,8. Cassava cultivars are generally categorized as “bitter” or “sweet,” depending on the level of the cyanogenic glycoside. Bitter cassava cultivars have hydrogen cyanide ranging from 15 to 400 mg hydrogen cyanide per kilogram of fresh roots, while sweet cassava has values of 15–50 mg hydrogen cyanide/kg fresh cassava3.
Generally, the processing of cassava products tends to reduce the cyanide content significantly. Regrettably, careful processing usually results in the loss of some macro and micronutrients such as proteins, vitamins, and minerals, thereby reducing the product’s nutritional value. Breeding efforts to reduce the level of cyanogenic glycoside is the principal approach. Despite the efficient processing techniques, cyanide exposure from cassava products still poses a significant concern9. Conventional breeding has generated cassava cultivars with low to high cyanide but has not provided cultivars with no cyanogenic glycosides10,11. In their study, Jorgenson et al.12 obtained transgenic cassava plants with more than 99% reduction in cyanide potential and 92% tuber reduction. In IITA, more than one million cassava seedlings were evaluated for low cyanide using the picrate method. A base population of low cyanide cassava was obtained through multiple and continuous selections and recombination of the genotypes with low cyanogenic potential to improve people of low cyanogenic potential and combine high-yielding potential with pest resistance13. The Genetic Gain Assessment (GGA) trials are cassava genotypes developed and selected for the Uniform Yield Trial (UYT) breeding stage. At this stage, they were taken into multilocation On-farm practices for nomination for National Coordinated Research Project on Cassava (NCRP) trials by the National Root Crops Research Institute (NRCRI)- an agricultural research institute in Nigeria before the release. Cyanide determination was initially determined by picrate paper methods developed by Mburu et al.14. It consisted of placing picrate paper at the entry of a small transparent plastic bottle (5 × 2 cm) containing about 1 g of sample and then 1 mL of phosphate buffer at pH 8. The bottle is closed and left at ambient temperature for 24 h. The change in colour of picrate paper from yellow to chestnut–red will indicate the release of cyanide in the sample and its absorption by picrate paper. Then, the picrate paper will be removed and placed in a test tube containing 5.0 mL water, and the absorbance of the solution will be measured at 510 nm using a spectrophotometer. The picrate leaf method has been used extensively for screening many clones, but the accuracy of the technique is uncompensated. Therefore, a more accurate process, Technicon AutoAnalyzer15, was developed to accurately analyze 300 samples daily13. However, the auto-analyzer method also has certain limitations with tedious sample preparations and extractions, especially when a more significant number of cultivars are to be evaluated. A rapid method of determining cassava roots’ cyanogenic potential and dry matter while they are still underground is required to monitor the relationship between root age and cyanogenic potential.
The cyanide contents of cassava leaves and roots depend on the roots’ ages and parts and range from 189 to 2466 ppm16,17. Older leaves and roots have lower cyanide compared to the younger ones. Root parts closer to the stem end have different cyanide content than those more relative to the cortex. Also, the leaves in the lower part may have more cyanogenic potential than those in the upper part7. Hidayat et al.7, reported a significant positive correlation between the cyanogenic potential of roots (Y) and leaves (X) among the 45 Indonesian germplasm which has reasonable cyanogenic possibilities; the regression equation was Y = 36.214 + 1.3085X (r = 0.5228). This current study aims to evaluate the cyanogenic potential and dry matter content of the cassava root and leaf of the Genetic Gain Assessment trial growing in a different environment and establish the association between the cyanogenic and dry matter of the roots and leaf. This study’s findings will significantly benefit the cassava breeding program to select low cyanide genotypes using either the cyanogenic potential or dry matter of the leaves to determine the roots at different maturity periods of the plant.
Materials and methods
Genetic materials and field establishment
Four hundred cassava genotypes were planted under IVS (Dry season in Inland Valley Hydromorphic area) and Upland (rain-fed conditions) trials in 2 locations of IITA Research Farms, Ibadan (IVS and Upland) and Mokwa (Upland) in Nigeria during the 2006 and 2007 planting seasons. The climate data of the 2 locations are presented in Supplementary Table S1. An Augmented Completely Randomized Design (ACRD) with three checks, TME 1, 91/02324, and 30572, was used for the IVS and Upland trials, respectively. Planting was done on ridges (30 cm high and 1 m apart) as plots. The IVS trial consisted of one ridge and five plants per plot with 0.5 m spacing between plants and 1 m between ridges. However, for the upland trial, each plot consisted of one ridge and ten plants per plot spaced 0.5 m between plants and 1 m between ridges. There were no fertilizers or herbicides applied to both trials. Manual weeding was done as necessary.
This research field study protocols on cassava were reviewed and approved by the International Institute of Tropical Agriculture Internal Research Review Board (IITA-IRB), ensuring compliance with the relevant institutional and national guidelines and legislation (IITA-IRB-Policy-June2016.pdf).
Sampling and sample preparation
Sampling and sample preparations follow the method Alamu et al.18 described. Five plants per genotype were harvested 12 months after planting (MAP). Three cassava roots of different sizes were selected per genotype randomly and labelled appropriately. The cassava roots were washed with tap water and air-dried. The roots were peeled with a stainless-steel knife and rinsed in deionized water. Each root was quartered by dividing longitudinally into two sections, and the opposite sections were selected and cut into smaller pieces and packed into a Whirl pack bag for subsequent analysis. Before laboratory analysis, cassava leaves were collected from the field, washed, air-dried, homogenized, and packaged.
Determination of dry matter
Ten grams of the raw root and powdered leaves were weighed in a pre-weighed aluminium can and baked for 16 h at 105 °C in an air convectional oven (Memmert UN 55, GmbH) to achieve constant weight. The dry matter content was estimated as the difference between the mass before drying and the mass loss on drying19.
Determination of cyanogenic potential (CNP)
Each root was homogenized using a laboratory blender with 250 mL of 0.1 M orthophosphoric acid. Leaves were also cut and homogenized for cassava. The homogenate was centrifuged, and extract taken from the supernatant was taken as the extract; 0.1 mL of the enzyme was added to 0.6 ml of the extract. The 3.4 mL of the acetate buffer (pH 4.5) was added and mixed. Following this, 0.6 mL of colourant and 0.2 mL of 0.5% chloramines-T were added to allow the colour to develop fully, and the mixture was left to stand for 15 min. The absorbance value was measured at 605 nm compared to a blank with all the same chemicals added but with 0.1 mL of phosphate buffer instead of KCN20.
Statistical analysis
Descriptive, ANOVA, LSD mean separation, Agglomerate Hierarchical Cluster(AHC) analysis, and Pearson correlation analysis using XLSTAT statistical and data analysis solution. New York, USA https://www.xlstat.com/en)21.
Results and discussion
Assessment of CNP and DM contents of GGA cassava genotypes (leaf and root) from the different growing environments
Table 1 and Fig. 1 summarise descriptive statistics for CNP and DM contents of cassava roots and leaves across the three planting environments. CNP content of cassava leaves in IVS, Mokwa, and Upland ranged from 3.39 to 272.16 mg/100 g, 4.28 to 228.72 mg/100 g, and 13.13 to 127.39 mg/100 g, respectively. However, the respective CNP range in root samples across IVS, Mokwa, and Upland was 0.76–76.31 mg/100 g, 0.94–136.53 mg/100 g, and 2.37–47.11 mg/100 g. These values agree with previously published studies18,22. Also, the varieties in IVS and Mokwa with a minimum CNP concentration of 0.94 mg/100 g respectively in the root are within the World Health Organization limit of 1 mg/100 g (10 ppm)23. The mean ± SD of DM content of foliage were 27.97 ± 3.01%, 28.81 ± 4.01%, and 13.65 ± 3.69%, respectively, in IVS, Mokwa, and Upland, while the root samples had mean ± SD of DM content of 38.09 ± 4.80%, 32.69 ± 5.93% and 24.63 ± 5.07% in the respective locations. The DM of the root samples analyzed in this study is consistent with the values reported by Kundy et al.24, Oly-Alawuba and Agbugbaeruleke25, and Alamu et al.18. For cassava foliage, samples from IVS had the highest CNP with a mean value of 68.14 mg/100 g, while Upland had the least CNP with an average value of 26.44 mg/100 g. On the other hand, DM content was highest in foliage samples from Mokwa with a mean value of 28.81% and most diminutive in representatives from IVS with an average of 27.97%. However, for root samples, CNP was highest in samples from Mokwa (21.86 mg/100 g) and lowest in samples from IVS (12.43 mg/100 g); meanwhile, for DM content, it was highest in IVS (38.09%) and most deficient in Upland (24.63%). Cassava has been classified based on the CNP levels as sweet or nontoxic, with CNP levels below 50 ppm, while levels between 50 and 100 ppm are considered moderately harmful. Bitter cassava has CNP levels above 100 pm and is classified as poisonous and unsafe for consumption. The levels of CNP in the roots in this current study are primarily below 100 pm for the locations studied. In general, the average CNP was consistently higher in foliage than in roots across the three environments, which concurs with the study by Burns et al.22. This is a result of cyanide synthesis, which starts from the shoot apex and the leaves of the cassava plant and is then transported to the roots, thereby making cassava leaves have higher cyanide concentration than roots12,26. Burns et al.22 also reported that leaves from plants experiencing water stress tend to have high cyanide content, consistent with the observed high CNP of leaves samples from IVS.
Effect of genotypes and growing location on the CNP and DM contents of GGA cassava root and leaf
Table 2 shows the effect of genotype and growing location of the CNP and DM content of GGA cassava root and foliage. In contrast, Supplementary Table S2 shows the means of the parameters by genotypes across the three locations. Both site and genotype had a highly significant effect (p < 0.001) on the CNP of root and leaves and DM of root and leaves. However, the location did not significantly affect the mean DM of leaf samples from Mokwa and Upland. This could result from the similar environmental conditions of these two locations. The significant effects of genotype and location have been previously reported18,22,27. The effects of the environment and genotypes respond differently to changes in environmental conditions, which could be attributed to different climatic conditions such as rainfall, temperature, soil pH, and soil fertility. This significant GxE supports the importance of testing cassava across multiple locations to characterize genotype performance accurately28. Also,29 confirmed the effects of genotypes and genotype-environment interaction on cassava genotypes grown at different locations in Tanzania.
Cluster analysis of genetic gain assessment (GGA) cassava genotypes (leaf and root) CNP and DM contents
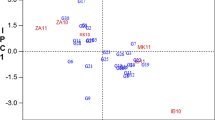
Table 3 and Fig. 2 show the hierarchical cluster analysis of the 400 GGA cassava genotypes. Cluster analysis classifies multivariate datasets into subgroups based on population similarities30. In 2021, 224 cassava genotypes were grouped into 3 clusters based on their carotenoids, vitamin C, cyanide, and dry matter compositions18 similar to what we obtained. They showed in their study that cluster 1 had 56.6% of the total population, while 2 and 3 had 42.9 and 3.57%, respectively. In this study, the dendrogram shows the association of the genotypes based on similarities in their cyanide and dry matter content across different locations. Different genotypes were put into various groups. The samples were divided into two clusters, with cluster 2 showing greater values for all parameters and models other than the leaf DM. Cluster 1 has 1510 populations, more than four times as much as Cluster 2, which only has a population of 461.
Moreover, all the check samples were found in cluster 2. It implies that all the genotypes in cluster 2 with superior dry matter (DM) roots could interest the breeder. The clustering could assist the breeders in identifying genotypes with good dry matter and low cyanide content immediately.
Pearson correlation between CNP (root and leaf) and DM (root and leaf)
Figure 2 and Table 4 present Pearson’s correlation statistics of CNP and DM parameters. The analysis measured the linear correlation between dry matter and cyanide composition to indicate their direction within the sample populations31. A strong positive linear relationship exists between CNP-leaf, CNP-root (r = 0.158), DM-leaf, and DM-root (r = 0.056). Also, CNP-roots had a negative correlation (p ≤ 0.001, r = − 0.194) with DM-root, while DM-Leaf and CNP-Leaf had a negative correlation (p ≤ 0.001, r = − 0.15), respectively. Furthermore, DM-root had a significant negative correlation (r = − 0.194) with CNP-root at p ≤ 0.001, whereas it had a positive significant (p ≤ 0.05, r = 0.082) correlation with CNP-leaf. The relationship between CNP-leaf and CNP roots had the highest positive correlation of 0.158, indicating that the root’s cyanide content could be drawn from the leaf.
On the other hand, the dry matter of the leaf shows a negative trend in the CNP of the roots. Nweke et al.10,11 also reported a strong negative relationship between CNP roots by establishing a positive correlation with moisture which could be linked to the solubility of hydrogen cyanide in water. The current study also confirms the negative correlation d DM-roots and CNP roots, which agrees with the previous study. A positive correlation between traits means they could be bred together in breeding programs, and negatively correlated traits would be challenging to combine32. The relationship between the CNP-leaf and CNP roots could be valuable in their “inter-prediction” in cassava breeding programmes.
Linear regression of CNP and DM in cassava root and leaf
Figure 3 and Table 5 describe the regression between the CNP and DM of the roots and leaves of cassava. The regression equations of DM and CNP were validated using an independent set of samples by predicting the DM of roots using the DM of the leaf and then the CNP of the roots from the CNP of the leaf, respectively. The coefficient of determination (R2) was 0.95 with a Root Mean Square Error of 7.83 for DM, while R2 of 0.54 was obtained for CNP with an RMSE of 13.86. Results showed that the prediction of DM of roots from the leaves gives more accuracy than CNP of roots from the CNP leaf. The regression equations for DM and CNP of the roots and leaves are as follows:
The regression models could be used as a rapid prediction tool for DM and CNP of the roots using their equivalent content in the leaves. However, Hidayat et al.7 reported a significant linear regression of CNP in the leaves and the roots of 99 genotypes from Indonesia, with a regression coefficient (R2) of 0.52. The regression coefficient in the study showed slightly improved performance compared to those previously reported. This could be due to the large datasets used and significant variations among the genotypes. However, we planned to apply the equations to predict the DM root and CNP root from the DM leaf and CNP leaf, respectively, using a new set of genotypes and improving the models’ accuracy and robustness.
Conclusions
Genetic Gain Assessment (GGA) trials which have attained the advanced breeding stage and are next to take for on-farm multilocation trials, have been purposively selected to establish the relationship between CNP-roots vs CNP-leaf and DM-roots vs DM-leaf. This study has shown that location and genotype had a highly significant effect on the CNP of roots and leaves and DM of roots and leaves. The maximum CNP in the roots and leaves were found in genotypes 99/0110 and MM97/0016, respectively, while the genotypes 93/0658 and 01/1412 had the maximum DM in roots and leaves, respectively. Minimum CNP and DM in the roots were observed in genotypes 92/1154 and 99/214, respectively. Linear regressions were established between cyanide and dry matter of roots and leaves, providing an indirect determination of cassava roots from the leaves, which could be relevant to monitor the build-up of cyanide in cassava as its ages. Generally, the findings from this study will significantly benefit the cassava breeding program to select low cyanide genotypes using either the cyanogenic potential or dry matter of the leaves to determine the roots at different maturity periods of the plant.
Data availability
The data associated with this study are presented in the supplementary tables, and upon request, the corresponding author will provide additional data.
References
Asante-Pok, A. Analysis of incentives and disincentives for cassava in Nigeria. Tecnhinical note series, MAFAP, FAO Rome. www.fao.org/mafap (2013).
Montagnac, J. A., Davis, C. R. & Tanumihardjo, S. A. Nutritional value of cassava for use as a staple food and recent advances for improvement. Comprehensive Rev. Food Sci. Food Saf. 8(3), 181–194. https://doi.org/10.1111/j.1541-4337.2009.00077.x (2009).
Ndubuisi, N. D. & Chidiebere, A. C. U. Cyanide in cassava: A review. Int. J. Genom. Data Min 118, 1. https://doi.org/10.29011/2577-0616.000118 (2018).
Makanjuola, O. M., Ogunmodede, A. S., Makanjuola, J. O. & Awonorin, S. O. Comparative study on quality attributes of gari obtained from some processing centres in South West, Nigeria. Adv. J. Food Sci. Technol. 4(3), 135–140 (2012).
CAC/RCP 73. Codex Alimentarius commission/code of practice for reducing hydrocyanic acid (HCN) in cassava and cassava products, http://www.fao.org/input/download/standards/13605/CXP_073e.pdf (2013).
Orjiekwe, C. L., Solola, A., Iyen, E. & Imade, S. Determination of cyanogenic glycosides in cassava products sold in Okada, Edo State, Nigeria. Afr. J. Food Sci. 7, 468–472 (2013).
Hidayat, A., Zuraida, N. & Hanarida, I. The cyanogenic potential of roots and leaves of ninety-nine cassava cultivars. Indones. J. Agric. Sci. 3(1), 25–32 (2002).
Omotioma, M. & Mbah, G. O. Kinetics of natural detoxification of hydrogen cyanide contained in retted cassava roots. Int. J. Exp. Res. 1, 9 (2013).
Oluwole, O., Onabolu, A., Link, H. & Roslin, H. Persistence of tropical ataxic neuropathy in a Nigerian community. J. Neurol. Neurosurg. Psych. 69, 96–101 (2000).
Nweke, F. I., Dunstan, S. C., Spencer, D. S. C. & Lyman, T. K. Cassava transformation 272 (University Press, Michigan State, USA, 2002).
Nweke, F., Spencer, D. & Lynam, J. The cassava transformation: Africa’s best-kept secret mich (St. Univ, 2002).
Jørgensen, K. et al. Cassava plants with a depleted cyanogenic glucoside content in leaves and tubers: Distribution of cyanogenic glucosides, their site of synthesis and transport, and blockage of the biosynthesis by RNA interference technology. Plant Physiol. 139(1), 363–374. https://doi.org/10.1104/pp.105.065904 (2005).
Dixon, A. G. O., Asiedu, R. & Bokanga, M. Breeding of cassava for low cyanogenic potential: Problems, progress, and prospects. Acta Hort. 375, 153–162. https://doi.org/10.17660/actahortic.1994.375.13 (1994).
Mburu, F. W., Swaleh, S. & Njue, W. Potential toxic levels of cyanide in cassava (Manihot esculenta Crantz) grown in Kenya. Afr. J. Food Sci. 6, 416–420 (2013).
Rao, P. V. & Hahn, S. K. An automated enzymatic assay for determining the cyanide content of cassava (Manihot esculenta Crantz) and cassava products. J. Sci. Food Agric. 35, 426–443 (1984).
Ekanayake, I.J., & Bokanga, M. A review on the production agronomy and cyanogenesis. In Proceedings of the Second International Scientific Meeting, Bogor, Indonesia, 22–26 August 1994. Centro International de Agricultura Tropical (CIAT), Cali, Colombia pp. 548–563 (1994).
Morales, I., & Villegas, L. Linamarase accumulation in cassava leaves (Manihot esculenta Crantz). p. 404–413. In Proceedings of the Second International Scientific Meeting, Bogor, Indonesia, 22–26 August 1994. Centro International de Agricultura Tropical (CIAT), Cali, Colombia (1994).
Alamu, E. O., Maziya-Dixon, B., Lawal, O. & Dixon, G. A. Assessment of chemical properties of yellow-fleshed cassava (Manihot esculenta) roots as affected by genotypes and growing environments. J. Agric. Sci. 43(2), 409–421. https://doi.org/10.17503/agrivita.v43i2.2804 (2021).
AOAC Method 925.09. Official methods of analysis, 16th and. In: Cuniff P (ed). AOAC International, Arlington, VA, USA, pp 32–41 (1995).
Essers, S. A. J. A., Bosveld, M., Van Grift, R. M. D. & Voragen, A. G. J. Studies on quantifying specific cyanogens in cassava products and introducing a new chromogen. J. Sci. Food Agric. 63(3), 287–296. https://doi.org/10.1002/jsfa.2740630305 (1993).
Addinsoft. XLSTAT statistical and data analysis solution. New York, USA. https://www.xlstat.com/en (2022).
Burns, A. E. et al. Variations in the chemical composition of cassava (Manihot esculenta Crantz) leaves and roots as affected by genotypic and environmental variation. J. Agric. Food Chem. 60, 4946–4956. https://doi.org/10.1021/jf2047288 (2012).
FAO/WHO. Codex standard for edible cassava flour. In Joint FAO/WHO Food Standards Programme, Codex Alimentarius Commission; Food and Agriculture Organisation and World Health Organisation of the United Nations: Rome, Italy,; Vol. Codex Standard 176−1989 (1995).
Kundy, A.C., Mkamilo G.S., & Misangu R.N. Understanding some of the nutritional characteristics of twelve cassava (Manihot esculenta Crantz) genotypes in southern (2015).
Oly-Alawuba, N. M. & Agbugbaeruleke, J. N. Effect of processing on the chemical composition and sensory evaluation of white cassava (TME 419) Manihot utilissima and improved yellow cassava (TMS 01/1368) Manihot palmate varieties. Int. J. Innov. Res. Sci. Eng. Technol. 6(11), 21566–21575 (2017).
Chaiareekitwat, S. et al. Protein composition, chlorophyll, carotenoids, and cyanide content of cassava leaves (Manihot esculenta Crantz) as influenced by cultivar, plant age, and leaf position. Food Chem. 372, 131173. https://doi.org/10.1016/j.foodchem.2021.131173 (2022).
Hue, K. T., Van, D. T. T., Ledin, I., Wredle, E. & Sporndly, E. Effect of harvesting frequency, variety and leaf maturity on nutrient composition, hydrogen cyanide content and cassava foliage yield. Asian-Aust. J. Anim. Sci. 25(12), 1691–1700 (2012).
Ndungu, J. F. et al. Influence of the environment on cassava quality traits in central rift valley of Kenya. Am. J. Plant Sci. 3(10), 1504–1512. https://doi.org/10.4236/ajps.2012.310181 (2012).
Nduwumuremyi, A., Melis, R., Shanahan, P., & Theodore, A. The interaction of genotype and environment affects important traits of cassava (Manihot esculenta Crantz). Crop J. 5(5), 373-386 (2017).
Everitt, B. S., Landau, S., Leese, M., & Stahl, D. Cluster analysis. John Wiley & Sons Ltd. https://doi.org/10.1002/9780470977811 (2011).
Kozak, M., Krzanowski, W., & Tartanus, M. Use of the correlation coefficient in agricultural sciences: Problems, pitfalls and how to deal with them. An. Acad. Bras. Ciênc. 84(4), 1147–1156. https://doi.org/10.1590/S0001-37652012000400029 (2012).
Adeboye, A. S., Bamgbose, A., Adebo, O. A., Okafor, D. C., & Azeez, T. B. Physicochemical, functional, and sensory properties of tapioca with almond seed (Terminalia catappa) flour blends. Afr. J. Food Sci. 13(8), 182–190. Retrieved from https://academicjournals.org/journal/AJFS/article-full-text-pdf/C99E2D461756 (2019).
Acknowledgements
The authors acknowledged the CGIAR Research Program on Roots, Tubers, Bananas (RTB), and Harvest Plus. We warmly thank the assistance of the Food and Nutrition Sciences Laboratory and Cassava Breeding Unit staff, IITA, Ibadan, Nigeria, particularly Drs. Parkes Elizabeth and Kulakow Peter.
Funding
This research and the APC were funded by the CGIAR Research Program on Roots, Tubers and Bananas (RTB) and the Bill and Melinda Gates Foundation (BMGF) through OPP1019962.
Author information
Authors and Affiliations
Contributions
E.O.A., G.A.D., B.M.D.: conceived, designed, and performed the experiments; E.O.A., M.A.: analyzed and interpreted the data; wrote the first draft of the paper. All authors read and revised the final draft before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alamu, E.O., Dixon, G.A., Adesokan, M. et al. Correlate the cyanogenic potential and dry matter content of cassava roots and leaves grown in different environments. Sci Rep 13, 15382 (2023). https://doi.org/10.1038/s41598-023-42425-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-42425-2
- Springer Nature Limited