Abstract
Pleistocene environments are among the most studied issues in paleoecology and human evolution research in eastern Africa. Many data have been recorded from archaeological sites located at low and medium elevations (≤ 1500 m), whereas few contexts are known at 2000 m and above. Here, we present a substantial isotopic study from Melka Kunture, a complex of prehistoric sites located at 2000—2200 m above sea level in the central Ethiopian highlands. We analyzed the stable carbon and oxygen isotopic composition of 308 faunal tooth enamel samples from sites dated between 2.02 and 0.6 Ma to investigate the animal diets and habitats. The carbon isotopic results indicate that the analyzed taxa had C4-dominated and mixed C3-C4 diets with no significant diachronic changes in feeding behavior with time. This is consistent with faunal and phytolith analyses, which suggested environments characterized by open grasslands (with both C3 and C4 grasses), patches of bushes and thickets, and aquatic vegetation. However, palynological data previously documented mountain forests, woodlands, and high-elevation grasslands. Additionally, the carbon isotopic comparison with other eastern African localities shows that differences in elevation did not influence animal feeding strategies and habitat partitioning, even though plant species vary according to altitudinal gradients. In contrast, the oxygen isotopic comparison suggests significant differences consistent with the altitude effect. Our approach allows us to detect diverse aspects of animal behavior, habitat, and vegetation that should be considered when reconstructing past environments.
Similar content being viewed by others
Introduction
Over the past decades, intensive paleoanthropological research has shown that the fossil record in eastern Africa preserves key information on the early stages of the evolutionary history of hominins1. Diverse hypotheses have been developed to explain why and how the evolution of mammals is linked to climatic and environmental changes throughout the Pleistocene2,3,4,5,6,7,8. These hypotheses are based on the current understanding of the global paleoclimate records, as well as on the development of the East Africa Rift System, where tectonic events have generated significant regional variations in climate, hydrological landscape, and distribution of vegetation9,10,11.
The paleoenvironmental archives are mostly from Pleistocene archaeological and paleontological sites in eastern Africa at low and medium elevations (≤ 1500 m a.s.l.) (e.g., Olduvai Gorge, Lake Turkana Basin, Busidima Formation)12,13,14,15,16,17,18,19, whereas few archaeological sites are known at higher elevations (≥ 2000 m a.s.l.), such as Melka Kunture (hereafter MK)20,21,22,23,24, Melka Wakena25,26,27,28, Gadeb29, Fanta30, and Mount Dendi31 in Ethiopia; and Kilombe32 in Kenya. Among the Ethiopian sites at high elevations, MK yields a long geo-archaeological sequence spanning from 2.02 Ma to the Holocene22,33,34,35,36,37, where the archaeological layers that recorded fauna and hominin remains, pollen, phytoliths, and ichnological evidence are coupled with accurate stratigraphic positioning.
The present study discusses a comprehensive stable isotopic composition of carbon and oxygen in 308 faunal tooth enamel samples from MK at sites dated between 2.02 and 0.6 Ma. We apply the isotopic data to provide information on the diet and habitat of the Pleistocene fauna and evaluate possible dietary variability over time. Furthermore, we integrate our interpretation with other proxies (e.g., faunal taxonomy, pollen, and phytolith data) to better characterize the environmental conditions of the area. Finally, we compare our isotopic results with other published data from eastern African sites at low and medium elevations to evaluate whether the differences in altitude and vegetation composition influenced animal feeding strategies and behavior. This is the first substantial stable isotopic study along the MK archaeological sequence.
Archeological background
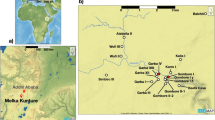
MK is a cluster of prehistoric sites located approximately 50 km southwest of Addis Ababa, on the western shoulder of the Main Ethiopian Rift, between 2000 and 2200 m a. s. l.20,21,22, extending over 100 km2 on the banks of the Upper Awash River (Fig. 1A, B). The stratigraphic sequence was controlled by the interplay between variable tectonic, fluvial, and volcanic activities placed in a low-energy floodplain38,39,40,41. The archaeological sequence begins with the Early Stone Age, with Oldowan lithic tools (2.02 Ma) and Early, middle, and final Acheulean artifacts (1.95–0.6 Ma)23,24,35,36,42,43. The Early Middle Stone Age is documented at ~ 200 ka, whereas the Late Stone Age, mainly found in surface dispersions, is now investigated in a stratified deposit dated to the Holocene22. Fossil remains of Homo erectus, of a hominin related to H. heidelbergensis, and of an archaic H. sapiens were discovered within clear stratigraphic contexts and are directly associated with lithic technocomplexes and faunal remains24,44,45,46,47. The faunal assemblage is dominated by Hippopotamus cf. amphibius, a large-sized hippo similar to the modern one. In contrast, the dwarf hippo (Hippopotamus cf. aethiopicus) is less represented. Alcelaphini is the most common bovid tribe recorded so far, whereas a few specimens refer to buffaloes (Bovini), kobs (Reduncini), and gazelles (Antilopini). The development of endemic mammal sub-species (e.g., Connochaetes gentry leptoceras and Damaliscus strepsiceras) points to a relative degree of isolation in the highlands due to the topographic relief. Equids are represented by the bones and teeth of Equus and Hipparion s.l., but the taxonomic identification was difficult. Suids are quite rare, with remains of Kolpochoerus, Metridiochoerus, and Phacochoerus sp. For giraffids, the record includes Giraffa sp. and the short-limbed Sivatherium maurusium. A few remains of elephants, rhinos, and crocodiles are recorded. The baboon Theropithecus cf. oswaldi is the only non-human primate attested. Carnivores are extremely rare, consisting of a few remains of Pseudocivetta ingens, Megantereon, Lupulella, and Crocuta sp. In addition, two rodent species have been identified as Oenomys kunturensis and Tachyoryctes konjitae48,49 (Tab. S1). Mammals, including hominins, are also attested by footprints in many Pleistocene horizons within the Gombore gully50,51,52.
(A) Location of MK in the Upper Awash Valley of Ethiopia; (B) map of the archaeological sites involved in this study (Apple Maps version 3.0 – 2811.22.9.28.12); (C) MK stratigraphic sequences: the sedimentary interpretation of Garba, Gombore, and Simbiro is from one of us (R.T.M.), whereas the succession from Karre is according to Chavaillon and Piperno20. Chronology is from Morgan et al.33 and Perini et al.35.
All over the MK sequence, the vegetation was of the Dry evergreen Afromontane Forest and Grassland Complex (DAF) type53, ranging from forest to grassland and bushland, with variations in the distribution of trees, grasses, and herbs over time (Tab. S2; Fig. S1). The DAF vegetation currently characterizes the higher mountain ranges of eastern Africa with a cool and rainy climate. The plant species greatly differs from the warmer and drier plant and tree species of the African savanna at lower elevations53,54. In Ethiopia, the DAF vegetation is found between 1800 and 3000 m a.s.l. In the MK area, where it would still develop, it has disappeared in modern times due to anthropic impact54,55.
The modern rainfall distribution is bimodal, influenced by the Indian Monsoon and the seasonal oscillation of the Inter-Tropical Convergence Zone (ITCZ). During a short rainy season (locally named Belg) from March to June and long summer rains (locally named Kiremt) from July to October, the wind direction changed from the northeast to southeast, which brought moisture from the Southern Indian Ocean. The dry season (locally named Bega) from November to February begins when the north-easterly trade winds from the Arabian Sea prevail56. According to the Ethiopian Meteorological Institute57, in the Upper Awash Valley at ~ 2000 m and above, 1000 mm of total rainfall was recorded in 2022, with mean maximum and minimum temperatures of 26 °C and 11 °C, respectively (Fig. S2).
Results
We report on δ13C and δ18O values of six mammalian families (Hippopotamidae, Bovidae, Equidae, Suidae, Hyaenidae, and Giraffidae) in order of their specimen abundance and combined with each archaeological locality, stratigraphic level, and chronology (Fig. 1C; Tab. S3). Overall, 308 isotopic results are reported with median and average values and ranges for each mammalian family. The sampling strategy reflects relative faunal abundance. Indeed, Hippopotamidae and Bovidae are the most abundant taxa in faunal assemblage and isotopic datasets.
Hippopotamidae
The median δ13C value of Hippopotamus cf. amphibius (n = 138) was—0.07 ‰, ranging from—6.6 ‰ to + 2.8 ‰. The δ18O values ranged from + 18.2 ‰ to + 30.9 ‰ with a median value of + 24.3 ‰ (Fig. 2A). The statistics indicate a non-normal distribution of δ13C values (Shapiro–Wilk Test: p = 0.0001), whereas δ18O values follow a normal distribution (Shapiro–Wilk Test: p = 0.9782), which are also confirmed by visual Kernel density and Normal Quantile Plot observations. Statistically significant differences in hippo δ13C (Wilcoxon/Kruskal–Wallis Rank-Sum Test: p = 0.0036) and δ18O values (Anova Test: p = 0.0064) were observed among the archaeological localities.
Box and whisker plots of δ13C and δ18O values of hippos (A), bovids (B), equids (C), suids (D), hyenas (E), and giraffids (F) from the MK archaeological sites (2.02 – 0.6 Ma). Isotopic data are plotted at the family level. Further details on Bovidae are provided in Fig. S3 (Supplementary Information). The vertical line in the boxes marks the median values; the box ends are the lower and upper quartiles; the lines define the range of data; a solid circle is equivalent to a value. Yellow and orange shades indicate mixed C3-C4 and C4-dominated diets, respectively16,58. Animal silhouettes are from Phylopic (https://www.phylopic.org).
Bovidae
The analyzed bovids (n = 105) comprise five tribes (Alcelaphini, Antilopini, Bovini, Hippotragini, and Reduncini) and others not identified below the family level.
Alcelaphini. Samples from alcelaphin bovids (n = 55) had a median δ13C value of + 2.9 ‰ with values ranging from—2.2 ‰ to + 5.2 ‰, whereas the median δ18O value was + 27.5 ‰ with values ranging from + 21 ‰ to + 33.1 ‰ (Fig. S3A).
Antilopini. Two samples of Antilopini had δ13C values of—9.7 ‰ and—2.8 ‰ (average =—6.2 ‰). The δ18O values were + 26.5 ‰ and + 30 ‰ (average = + 28.2 ‰) (Fig. S3B).
Bovini. Bovini samples (n = 5) had δ13C values ranging from—2.3 ‰ to + 3.8 ‰ (average = + 0.3 ‰). The δ18O values ranged from + 26 ‰ to + 31.1 ‰ (average = + 27.9 ‰) (Fig. S3C).
Hippotragini. Samples of Hippotragini (n = 3) had δ13C values ranging from—2 ‰ to + 2.1 ‰ (average =—0.3 ‰). The δ18O values ranged from + 24.4 ‰ to + 29.1 ‰ (average = + 27 ‰) (Fig. S3D).
Reduncini. Samples of Reduncini (n = 3) had δ13C values ranging from – 5.5 ‰ to + 1.4 ‰ (average =—1.1 ‰). The δ18O values ranged from + 24.3 ‰ to + 30.7 ‰ (average = + 27.1 ‰) (Fig. S3E).
Bovidae sensu lato. This group comprises bovid specimens (n = 37) not identified at the tribe and species levels. The median δ13C value was + 1.3 ‰ with values ranging from—4.6 ‰ to + 5 ‰, whereas the median δ18O value was + 25.3 ‰, ranging from + 20.2 ‰ to + 31.7 ‰ (Fig. S3F).
Overall, bovid δ13C values ranged from—9.7 ‰ to + 5.2 ‰ (median = + 1.9 ‰) (Fig. 2B). The δ13C values do not follow a normal distribution (Shapiro–Wilk Test: p = 0.0001), and the Wilcoxon/Kruskal–Wallis Rank-Sum Test (p = 0.0124) showed differences in mean δ13C values over time. Alcelaphini and Bovidae sensu lato showed higher δ13C values than those of Antilopini, Bovini, Hippotragini, and Reduncini. However, we noted that the small sample size of Antilopini, Bovini, Hippotragini, and Reduncini limited the effectiveness of statistical analysis. Normal Quantile Plot and Shapiro–Wilk Test (p = 0.8177) indicate a normal distribution of δ18O values, with statistically significant differences (Anova Test: p = 0.0001) among the archaeological sites and levels.
Equidae
This group comprises specimens mostly not assigned to mammalian tribes and species, whereas a few samples belong to Equus (n = 4) and Hipparion (n = 2) sp. The overall median δ13C value of equid samples (n = 45) was + 0.1 ‰, with values ranging from—7.6 ‰ to + 6.4 ‰. The median δ18O value was + 26.7 ‰, ranging from + 23.1 ‰ to + 31.3 ‰ (Fig. 2C). Both δ13C and δ18O values are normally distributed (Shapiro–Wilk Tests: p = 0.0798; p = 0.1170, respectively). Across the sites, statistically significant variability in δ13C and δ18O values was observed (Anova Tests: p = 0.0427; p = 0.0179, respectively).
Suidae
This family (n = 12) comprises a few samples of Metridochoerus (n = 4), Kolpochoerus (n = 2), and other suids (n = 6). Overall, average δ13C value was + 0.05 ‰, ranging from—2.3 ‰ to + 3.3 ‰, whereas median δ18O value was + 26.8 ‰, ranging from + 22.6 ‰ to + 29.6 ‰ (Fig. 2D).
Hyaenidae
Five samples of hyena had an average δ13C value of + 0.1 ‰ (range between—3.3 ‰ and + 2.5 ‰), whereas average δ18O value was + 25.5 ‰, ranging from + 24.3 ‰ to + 27 ‰ (Fig. 2E).
Giraffidae
Three samples of giraffids show an average δ13C value of + 2.2 ‰ (range between + 1.9 ‰ and + 2.7 ‰). The average δ18O value was + 27.4 ‰, ranging from + 24.5 ‰ to + 29 ‰ (Fig. 2F).
Due to the limited number of Suidae, Giraffidae, and Hyaenidae samples, a robust statistical evaluation was impossible. Therefore, our interpretation is only a preliminary one.
Discussion
The δ13C values suggest that the hippos had a diet dominated by C4 forage, although some individuals showed lower δ13C values, which reflects the opportunistic consumption of C3 grasses, fruits, aquatic vegetation, or a combination of these resources59,60,61,62,63,64,65. Indeed, modern hippos are known to spend most of their daily time close to a body of water, even though they regularly travel some kilometers to forage at night for preferred grass66. At MK, there is direct evidence of a 700-ka hippo trail produced by H. cf. amphibius, which is consistent with modern hippo behavior50,51. Bovid and equid δ13C values indicate that they had C4 diets, with some lower δ13C values interpreted as the outcome of a mixed C3-C4 diet. In contrast, suids and giraffids had full C4 diets, whereas hyena δ13C values reflect the various isotopic signatures of their prey67, which had both C4 and mixed C3-C4 diets (Fig. 2). The consumption of grazers and mixed feeders by hyenas is confirmed by carnivore tooth marks found on a hippo scapula and tibia at Gombore II-2, currently dated to 750 ka35,50,51. The δ18O values of all the taxa from different localities and stratigraphic levels point to variations in the oxygen isotopic composition of animal drinking water sources, food water, and possibly climatic and environmental changes over time. The hippos showed lower δ18O values than other taxa (e.g., bovid, equid, suid, and giraffid), which is consistent with the expected differences between semiaquatic and terrestrial habitats (Fig. 3), as already evidenced at MK64,68 and elsewhere69,70. In addition, hippos and hyenas have closely similar median δ18O values, which seems consistent with our δ13C values interpretation and previous taphonomic analysis50,51.
Box and whisker plots of δ18O values of hippos (semi-aquatic habitat) and bovids, equids, suids, and giraffids (terrestrial habitat). The horizontal line in the boxes marks the median values; the box ends are the lower and upper quartiles; the lines define the range of data; and the solid circle is for outliers.
The isotopic data agree with the analysis of faunal assemblage, suggesting environments characterized by open grasslands, patches of bushes and thickets, and aquatic vegetation. No evidence of pure browsers is attested by either faunal or isotopic analyses49,64. This is probably the result of taphonomic issues and the consequent sampling bias. In contrast, hippos are a significant component of the MK paleontological record because of their semi-aquatic habitat, which allows for a greater abundance within fluvial deposits. Caution is required when paleoenvironmental reconstruction is based on limited faunal remains. Pollen and phytolith analyses provide further environmental insights23,53. Within the DAF vegetation, which developed throughout the MK archaeological sequence, the proportion of mountain forests, woodlanS1ds, and grasslands was subjected to significant fluctuations53. The two periods when mountain grasslands were more extensive than forests are those evidenced at Garba IVD (1.95 Ma) and Garba IB (0.6 Ma) (Fig. S1), which are far from each other in time. Among various sites, there is ample evidence of phases with much more wooded and humid vegetation, with no trend toward increased aridity along the MK sequence. The changes should rather be seen as related to the global glacial and interglacial cycles53. The elevation also allowed for the development of diverse environments with C4 grass but also C3 types of grass, which at MK have been recorded by phytolith analysis23, and currently become dominant above 2000 m a. s. l.71. This is well-tracked by hippo, bovid, and equid dietary preferences in several archaeological localities with distinct chronologies, such as at Gombore IA (~ 1.6 Ma), Gombore Iγ (1.51 Ma), Simbiro III MS B (~ 1.2 Ma), Gombore II-1 (1.0 Ma), and Gombore II-2 (0.75 Ma). Changes in vegetation composition did not have a major impact on the feeding behavior of mammals, which selected the preferred plant species in the varied mosaic environments. Furthermore, we cannot rule out the possibility that C3 isotopic signals are associated with seasonal variations in the proportion of C3 and C4 grasses reflected in the animal diet. This was demonstrated elsewhere by Souron et al.63 using intra-tooth isotopic profiles.
In order to test whether the differences in elevation and vegetation influenced the animal feeding strategies and habitats, we compared δ13C and δ18O values (n = 1.064) of fauna tooth enamel from published data of eastern African sites at low and medium altitudes (≤ 1500 m a.s.l.), such as Olduvai Gorge13,14,15,16,19, Lake Turkana Basin17,65,72, and Busidima Formation12, with isotopic data from the higher elevation site of MK presented in this study. This isotopic dataset includes six faunal families (Hippopotamidae, Bovidae, Equidae, Suidae, Giraffidae, and Hyaenidae) dated between 2.1 and 0.6 Ma (Tab. S4). The comparison of δ13C values indicates a high degree of overlap in terms of dietary habits, with average diets dominated by C4 and mixed C3-C4 vegetation (Fig. 4). The only exception is represented by some bovids (Tragelaphini and Antilopini) and giraffids (Giraffa s.l., G. cf. stillei, G. cf. jumae) from the Lake Turkana Basin, Olduvai Gorge, and Busidima Formation, which only consumed C3 vegetation (Fig. S5). The median δ13C value of MK fauna is higher than those of other sites, which suggests a more C4-dominated open environment at MK than at the other archaeological sites for the sampled chronology. However, the hippos from MK and Olduvai Gorge show δ13C values close to each other, and the equids from MK and Lake Turkana have equal median δ13C values (Tab. S5, Fig. S5). In addition, an increase in δ13C values has been observed for C3 plants with altitude73,74. Therefore, the end-member δ13C value of C3 vegetation probably increased with altitude and could explain this slight increase of δ13C values of herbivorous taxa in MK compared to lower altitude sites due to this shift of δ13C values for C3 plants at higher altitude in taxa that consume some C3 plants.
Scatter plots of δ13C and δ18O values of hippos (A), bovids (B), equids (C), suids (D), hyenas (E), and giraffids (F) from Lake Turkana Basin, MK, Olduvai Gorge, and Busidima Formation. Animal silhouettes are from Phylopic (https://www.phylopic.org).
Based on carbon isotopic analysis, we argue that differences in elevation did not influence animal feeding strategies and habitat partitioning, even though plant species vary according to altitudinal gradients71. Furthermore, we emphasize that carbon isotopic data alone is insufficient to reconstruct the paleoenvironments directly. Indeed, at MK, δ13C values suggest the presence of extended grasslands in the landscape. However, there is evidence from pollen and phytolith analyses that the local mountain environment was characterized by a variable proportion of forests, woodlands, and grasslands (with both C3 and C4 grasses)23,53 through time.
Comparing δ18O values, we observe that taxa from MK had lower δ18O values than those from the other localities (Tab. S6, Fig. S6), consistent with the altitude effect 57,75,76,77. This is attributed to the progressive condensation of atmospheric vapor and rainout along the mountain slopes and its cool-off, with a consequent loss of H218O in the form of rain from cloud moisture. As a result, high-altitude precipitation shows lower δ18O values than low-altitude precipitation57. The altitude effect is reinforced by the difference in temperatures between low- and high-elevation localities76,77,78. Thus, when interpreting δ18O values from fossil tooth enamel as a paleoclimatic proxy, we should consider that many factors (continentality, source, altitude, temperature, seasonality, and relative humidity) may influence δ18O variability, making it difficult to establish a clear and direct link with past temperatures77.
Conclusion
Isotopic analyses of fauna tooth enamel from MK provide information on feeding behavior and their adaptation. Overall, between 2.02 and 0.6 Ma, the sampled taxa show a low variability in foraging strategies, which are all within the range of C4 and mixed C3-C4 diets. The DAF vegetation changed over the Lower and Middle Pleistocene times, with variable proportions of mountain plant species53, including both C3 and C4 grasses23,64, but this did not influence animal feeding habits. The analyzed mammals were able to select the preferred plants and successfully adapt to the mountain ecosystems. Comparisons with δ13C values from eastern African sites at low, medium (≤ 1500 m), and high elevations (≥ 2000 m) point to variability in the isotopic signal but not in the animal diets, which is constantly within the range of C4 and mixed C3-C4 diet. The plant species of the African lowlands are different from those at high-elevation53,71, but the animal habitat and behavior in selecting food were not influenced along the altitudinal gradient by the changing vegetation.
The δ18O values recorded significant differences, which were explained as resulting from the altitude effect. The climate of modern-day Ethiopia is influenced by variable altitudes, with mountains up to 4000 m and more, high plateaus between 1300 and 3000 m, and lowlands stretching to sea level. These differences are also reflected in diverse local climates56,76.
Between 2.02—0.6 Ma, the human presence at MK is directly recorded by paleoanthropological remains24,44,45,46, as well as by lithic artifacts23,36,42,43 and fossil footprints50,51,52. Together with the isotopic results, this allows speculating on human behavior and adaptation. The hominins were able to live and thrive in mountain mosaic ecosystems24,53,79, likely exploiting the diverse food resources provided by the DAF vegetation spectrum.
The detailed paleoecological reconstruction is the outcome of the implementation of multiple methodologies, such as stable isotopes, pollen, phytoliths, and faunal taxonomy. Based on our experience with an integrative and complementary approach, caution should be exercised when paleoenvironmental interpretations are determined instead by a single proxy or analyses that are limited by taphonomic bias.
Material and methods
Stable isotope analyses
We collected fauna tooth enamel samples (n = 308) from several archaeological localities within the MK complex of sites to analyze the stable carbon and oxygen isotopic compositions. The specimens include Artiodactyla (Hippopotamidae, Bovidae, Suidae, Giraffidae), Perissodactyla (Equidae), and Carnivora (Hyaenidae) in order to represent the full dietary diversity. The enamel samples were collected at the National Museum of Ethiopia (Addis Ababa) in agreement with the Ethiopian Heritage Authority (EHA). Enamel was sampled using a drilling device equipped with a diamond-tipped bit to obtain 12—15 mg of powder. Powdered samples were soaked in 2—3% NaOCl for 24 h at 20 °C to oxidize organic residues and rinsed thrice with Millipore water (Milli-Q H2O) to remove all NaOCl. The remaining samples were treated with 0.1 M acetic acid-calcium acetate buffer (pH = 4.66) for 24 h at 20 °C to remove exogenous carbonate. Samples were rinsed thrice with Milli-Q H2O and placed in an oven to dry at 40 °C for 72 h. Only 2.5—3 mg of structural carbonate was subjected to Isotopic Ratio Mass Spectrometry (IRMS) at the Biogeology Research Group of the University of Tübingen (Germany). Further details are provided in Supplementary A and B.
Statistical analysis
Statistical tests were performed using JMP 17 Pro (licensed by Sapienza University of Rome), with the significance level set at p = 0.05. The Shapiro–Wilk test was used to verify whether the data followed a normal distribution. When the hypothesis of the normal distribution was accepted, parametric tests such as the t-test and the analysis of variance (ANOVA) test were used to test whether the δ13C and δ18O values significantly differed over time for each archaeological site. For the same purpose, non-parametric tests such as the Mann–Whitney U-test (also known as Wilcoxon Rank Sum Test) or the Kruskal–Wallis test were used when the hypothesis of the normal distribution was rejected. The t-test and Mann–Whitney U-test were applied with two groups or levels, whereas ANOVA and Kruskal–Wallis tests were applied with three or more groups.
Data availability
The data generated and analyzed during the study are available in this published article and its Supplementary Information files.
References
Veldhuis, D., Kjærgaard, P. C. & Maslin, M. Human evolution: Theory and progress. In Encyclopedia of Global Archaeology (ed. Smith, C.) (Springer, New York, NY, 2014).
Potts, R. Evolution and climatic variability. Science 273, 922–923. https://doi.org/10.1126/science.273.5277.922 (1996).
Reed, K. E. Early hominid evolution and ecological change through the African Plio-Pleistocene. J. Hum. Evol. 32, 289–322. https://doi.org/10.1006/jhev.1996.0106 (1997).
deMenocal, P. African climate change and faunal evolution during the Pliocene-Pleistocene. Earth Planet. Sci. Lett. 220, 3–24. https://doi.org/10.1016/S0012-821X(04)00003-2 (2004).
Trauth, M. H. et al. High- and low-latitude controls and East African forcing of Plio-Pleistocene East African climate and early human evolution. J. Hum. Evol. 53, 475–486. https://doi.org/10.1016/j.jhevol.2006.12.009 (2007).
Maslin, M. A. & Trauth, M. H. Plio-Pleistocene East African pulsed climate variability and its influence on early human evolution. In The first human origins of the genus Homo (eds Grine, F. E. et al.) 151–158 (Springer Science, Berlin, 2009).
Potts, R. Hominin evolution in settings of strong environmental variability. Quat. Sci. Rev. 73, 1–13. https://doi.org/10.1016/j.quascirev.2013.04.003 (2013).
Faith, J. T. et al. Rethinking the ecological drivers of hominin evolution. Trends Ecol. Evo. 36(9), 797–807. https://doi.org/10.1016/j.tree.2021.04.011 (2021).
Bonnefille, R. Evidence for a cooler and drier climate in the Ethiopian uplands towards 2.5 Myr ago. Nature 303, 487–491 (1983).
Maslin, M. A. & Christensen, B. Tectonics, orbital forcing, global climate change, and human evolution in Africa. J. Hum. Evol. 53, 443–464. https://doi.org/10.1016/j.jhevol.2007.06.005 (2007).
Cuthbert, M. O. et al. Modelling the role of groundwater hydro-refugia in East African hominin evolution and dispersal. Nat. Commun. 8, 15696. https://doi.org/10.1038/ncomms15696 (2016).
Bedaso, Z., Wynn, J. G., Alemseged, Z. & Geraads, D. Paleoenvironmental reconstruction of the Asbole fauna (Busidima Formation, Afar, Ethiopia). Geobios 43, 165–177. https://doi.org/10.1016/j.geobios.2009.09.008 (2010).
van der Merwe, N. J. Isotopic ecology of fossil fauna from Olduvai Gorge at ca 1.8 Ma. compared with modern fauna. S. Afr. J. Sci. 109, 11–12 (2013).
Ascari, S. H., Njau, J. K., Sauer, P. E., Polly, P. D. & Peng, Y. Fossil herbivores and crocodiles as paleoclimatic indicators of environmental shifts from Bed I and Bed II times of the Olduvai Gorge, Tanzania. Palaeogeogr. Palaeoclimatol. Palaeoecol. 511(550–557), 2018. https://doi.org/10.1016/j.palaeo.2018.09.021 (2018).
Rivals, F. et al. Dietary traits of the ungulates from the HWK EE site at Olduvai Gorge (Tanzania): Diachronic changes and seasonality. J. Hum. Evol. 120, 203–214. https://doi.org/10.1016/j.jhevol.2017.08.011 (2018).
Uno, K. T. et al. Large mammal diets and paleoecology across the Oldowan-Acheulean transition at Olduvai Gorge, Tanzania from stable isotope and tooth wear analyses. J. Hum. Evol. 120, 76–91. https://doi.org/10.1016/j.jhevol.2018.01.002 (2018).
Negash, E. W. et al. Dietary trends in herbivores from Shungura Formation, southwestern Ethiopia. PNAS 117(36), 21921–21927. https://doi.org/10.1073/pnas.2006982117 (2020).
Semaw, S. et al. Co-occurrence of Acheulian and Oldowan artifacts with Homo erectus cranial fossils from Gona, Afar Ethiopia. Sci. Adv. https://doi.org/10.1126/sciadv.aaw4694 (2020).
Mercader, J. et al. Earliest Olduvai hominins exploited unstable environments ~2 million years ago. Nat. Commun. 12, 3. https://doi.org/10.1038/s41467-020-20176-2 (2021).
Chavaillon, J., Piperno, M. Studies on the early paleolithic site of melka kunture, ethiopia. Origines, Istituto Italiano di Preistoria e Protostoria (2004).
Mussi, M. et al. Une vue d’ensemble sur Melka Kunture, grand complexe de sites pléistocènes dans la vallée supérieure de l’Awash (Ethiopie). L’Anthropologie https://doi.org/10.1016/j.anthro.2022.102999 (2022).
Mussi, M., et al. Melka Kunture Alto Awash, Etiopía entre 2000000 y 5000 años. Salduie 23(1), 1–11, https://doi.org/10.26754/ojs_salduie/sald.202318553(2023)
Mussi, M., et al. A surge in obsidian exploitation more than 1.2 million years ago at Simbiro III (Melka Kunture, Upper Awash, Ethiopia). Nature Ecol. Evol. 7, 337–346, https://doi.org/10.1038/s41559-022-01970-1 (2023).
Mussi, M. et al. Early Homo erectus lived at high altitudes and produced both Oldowan and Acheulean tools. Science 382(6671), 713–718. https://doi.org/10.1126/science.add9115 (2023).
Hovers, E. et al. The expansion of the Acheulian to the Southeastern Ethiopian Highlands: Insights from the new early Pleistocene site-complex of Melka Wakena. Quat. Sci. Rev. 253, 106763. https://doi.org/10.1016/j.quascirev.2020.106763 (2021).
Gossa, T. & Hovers, E. Continuity and change in lithic techno-economy of the early Acheulian on the Ethiopian highland: A case study from locality MW2; the Melka Wakena site- complex. PLoS ONE 17(12), e0277029. https://doi.org/10.1371/journal.pone.0277029 (2022).
Martínez-Navarro, B. et al. The earliest Ethiopian wolf: implications for the species evolution and its future survival. Comm. Biol. 6, 530. https://doi.org/10.1038/s42003-023-04908-w (2023).
Gossa, T., et al. Melka Wakena, Ethiopia. In: Beyin, A., Wright, D.K., Wilkins, J., Olszewski, D.I. (Eds), Handbook of Pleistocene Archaeology of Africa. Springer, Cham. https://doi.org/10.1007/978-3-031-20290-2_27 (2023).
Clark, J. D. Transitions: Homo erectus and the acheulian: the Ethiopian sites of Gadeb and the middle Awash. J. Hum. Evol. 16, 809–826. https://doi.org/10.1016/0047-2484(87)90025-X (1987).
Lanzarone, P., Garrison, E., Bobe, R. & Getahun, A. Examining fluvial stratigraphic architecture using ground-penetrating radar at the Fanta stream fossil and archaeological site Central Ethiopia. Geoarchaeology 31, 577–591. https://doi.org/10.1002/gea.21584 (2016).
Vogelsang, R., et al. When Hominins Conquered Highlands - an Acheulean Site at 3000 m a.s.l. on Mount Dendi/Ethiopia. J. Paleolit. Archaeol. 1, 302–313, (2018).
Gowlett, J. A. J. et al. At the heart of the African Acheulean: the physical, social and cognitive landscapes of Kilombe. In Settlement, Society and Cognition in Human Evolution: Landscapes in Mind (eds Coward, F. et al.) 75–93 (Cambridge University Press, 2015).
Morgan, L. E. et al. A chronological framework for a long and persistent archaeological record: Melka Kunture Ethiopia. J. Hum. Evol. 62, 104–115. https://doi.org/10.1016/j.jhevol.2011.10.007 (2012).
Tamrat, E., Thouveny, N., Taieb, M. & Brugal, J. P. Magnetostratigraphic study of the Melka Kunture archaeological site (Ethiopia) and its chronological implications. Quat. Int. 343, 5–16. https://doi.org/10.1016/j.quaint.2013.11.030 (2014).
Perini, S., Muttoni, G., Monesi, E., Melis, R. T. & Mussi, M. Magnetochronology and age models of deposition of the Melka Kunture stratigraphic sequence (Upper Awash, Ethiopia) and age assessments of the main archaeological levels therein contained. Quat. Sci. Rev. 274, 107259. https://doi.org/10.1016/j.quascirev.2021.107259 (2021).
Sánchez-Dehesa Galán, S. et al. Age and formation processes of an Acheulean site with extensive accumulation of large cutting tools: Garba I (Melka Kunture, Upper Awash, Ethiopia). Archaeol. Anthropol. Sci. 14, 55. https://doi.org/10.1007/s12520-022-01521-6 (2022).
Muttoni, G., Perini, S., Melis, R. T. & Mussi, M. Chronology of the earliest peopling of the Ethiopian highlands at Melka Kunture pre-dating the 1.925 Ma base of the Olduvai subchron. Quat. Sci. Rev. 319, 108330. https://doi.org/10.1016/j.quascirev.2023.108330 (2023).
Kieffer, G., Raynal, J.-P., Bardin, G. Volcanic markers in coarse alluvium at Melka Kunture (Upper Awash, Ethiopia). In: Chavaillon, J., Piperno, M. (Eds), Studies on the Early Paleolithic site of Melka Kunture, Ethiopia. Origines, Istituto Italiano di Preistoria e Protostoria, 93–101 (2004).
Raynal, J.-P., Kieffer, G., Bardin, G. 2004. Garba IV and the Melka Kunture Formation. A preliminary lithostratigraphic approach. In: Chavaillon, J. and Piperno, M. (Eds), Studies on the Early Paleolithic site of Melka Kunture, Ethiopia. Origines, Istituto Italiano di Preistoria e Protostoria, 137–166 (2004).
Salvini, R., Riccucci, S. & Francioni, M. Topographic and geological mapping in the prehistoric area of Melka Kunture (Ethiopia). J. Maps 8, 169–175. https://doi.org/10.1080/17445647.2012.680779 (2012).
Pioli, L., Melis, R.T., Mussi, M. Volcaniclastic sedimentation in a closed, marginal rift basin: the case of the Melka Kunture area (upper Awash, Ethiopia). In: Di Capua, A., De Rosa, R., Kereszturi, G., Le Pera, E., Rosi, M., Watt, S.F.L. (Eds), Volcanic Processes in the Sedimentary Record: When Volcanoes Meet the Environment. Geological Society, London, Special Publications 520, https://doi.org/10.1144/SP520-2022-158 (2023).
Gallotti, R., Mussi, M. The Unknown Oldowan: ~1.7-Million-Year-Old Standardized Obsidian Small Tools from Garba IV, Melka Kunture, Ethiopia. PLOS ONE 10, e0145101, https://doi.org/10.1371/journal.pone.0145101 (2015).
Gallotti, R. & Mussi, M. Two Acheuleans, two humankinds: From 1.5 to 0.85 Ma at Melka Kunture (Upper Awash, Ethiopian highlands). JASs Reports 95, 1–46. https://doi.org/10.4436/JASS.95001 (2017).
Le Cabec, A. et al. Insights into the palaeobiology of an early Homo infant: multidisciplinary investigation of the GAR IVE hemi-mandible, Melka Kunture, Ethiopia. Sci. Rep. 11, 23087. https://doi.org/10.1038/s41598-021-02462-1 (2021).
Di Vincenzo, F. et al. The massive fossil humerus from the Oldowan horizon of Gombore I, Melka Kunture (Ethiopia,> 1.39 Ma). Quat. Sci. Rev. 122, 207–221. https://doi.org/10.1016/j.quascirev.2015.05.014 (2015).
Profico, A., Di Vincenzo, F., Gagliardi, L., Piperno, M., Manzi, G. Filling the gap. Human cranial remains from Gombore II (Melka Kunture, Ethiopia; ca 850 ka) and the origin of Homo heidelbergensis. J. Anthropol. Sci. 94, 41–63, https://doi.org/10.4436/JASS.94019 (2016).
Mussi, M., Altamura, F., Macchiarelli, R., Melis, R. T. & Spinapolice, E. E. Garba III (Melka Kunture, Ethiopia): a MSA site with archaic Homo sapiens remains revisited. Quat. Int. 343, 28–39. https://doi.org/10.1016/j.quaint.2013.08.028 (2014).
Geraads, D., Eisenmann, V., Petter, G. The large mammal fauna of the Oldowan sites of Melka Kunture. In: Chavaillon, J. and Piperno, M. (Eds), Studies on the Early Paleolithic site of Melka Kunture, Ethiopia. Origines, Istituto Italiano di Preistoria e Protostoria, 169–192 (2004).
Geraads, D., Gallotti, R., Raynal, J.-P., Bonnefille, R. & Mussi, M. Melka Kunture, Ethiopia: Early Pleistocene Faunas of the Ethiopian Highlands. In African Paleoecology and Human Evolution (eds Reynolds, S. C. & Bobe, R.) 256–268 (Cambridge University Press, 2022).
Altamura, F., Melis, R.T., Mussi, M. A Middle Pleistocene hippo tracksite at Gombore II-2 (Melka Kunture, Upper Awash, Ethiopia). Palaeogeogr., Palaeoclimatol., Palaeoecol. 470, 122–131, https://doi.org/10.1016/j.palaeo.2017.01.022 (2017).
Altamura, F. et al. Archaeology and ichnology at Gombore II-2, Melka Kunture, Ethiopia: everyday life of a mixed-age hominin group 700,000 years ago. Sci. Rep. 8, 2851. https://doi.org/10.1038/s41598-018-21158-7 (2018).
Altamura, F. et al. Ichnological and archaeological evidence from Gombore II OAM, Melka Kunture, Ethiopia: An integrated approach to reconstruct local environments and biological presences between 12–085 Ma. Quat. Sci Rev. 244, 106506. https://doi.org/10.1016/j.quascirev.2020.106506 (2020).
Bonnefille, R., Melis, R. T. & Mussi, M. Variability in the Mountain Environment at Melka Kunture Archaeological Site, Ethiopia, During the Early Pleistocene (~1.7 Ma) and the Mid-Pleistocene Transition (0.9–0.6 Ma). In The Emergence of the Acheulean in East Africa (eds Gallotti, R. & Mussi, M.) (Springer, Cham, 2018).
Friis, I., Demissew, S. & Van Breugel, P. Atlas of the potential vegetation of Ethiopia. Biologiske Skrifter 58, 1–307 (2010).
Mairal, M. et al. Geographic barriers and Pleistocene climate change shaped patterns of genetic variation in the Eastern Afromontane biodiversity hotspot. Sci. Rep. 7, 45749. https://doi.org/10.1038/srep45749 (2017).
Lemma, B. et al. Spatial and temporal 2H and 18O isotope variation of contemporary precipitation in the Bale Mountains Ethiopia. Isotopes Environ. Health Stud. 56(2), 1–14. https://doi.org/10.1080/10256016.2020.1717487 (2020).
Ethiopian Meteorological Institute – Data and climatology directorate (Federal Democratic Republic of Ethiopia, Ministry of water and energy). Annual climate bulletin for the year 2022. Available at: www.ethiomet.gov.et (2022).
Cerling, T. E. & Harris, J. M. Carbon isotope fractionation between diet and bioapatite in ungulate mammals and implications for ecological and paleoecological studies. Oecologia 120, 347–363. https://doi.org/10.1007/s004420050868 (1999).
Ansell, W. F. M. Feeding habits of Hippopotamus amphibius Linn. Puku 3, 171 (1965).
Mugangu, T. E. & Hunter, M. L. Jr. Aquatic foraging by Hippopotamus in Zaïre: Response to a food shortage?. Mammalia 56(3), 345–349. https://doi.org/10.1515/mamm.1992.56.3.345 (1992).
Boisserie, J. R. et al. Diets of modern and late Miocene hippopotamids: evidence from carbon isotope composition and microwear of tooth enamel. Palaeogeogr. Palaeoclimatol. Palaeoecol. 221, 153–174. https://doi.org/10.1016/j.palaeo.2005.02.010 (2005).
Cerling, T. E. et al. Stable isotope ecology of the common hippopotamus. J. Zool. https://doi.org/10.1111/j.1469-7998.2008.00450 (2008).
Souron, A., Balasse, M. & Boisserie, J. R. Intra-tooth isotopic profiles of canines from extant Hippopotamus amphibius and late Pliocene hippopotamids (Shungura Formation, Ethiopia): insights into the seasonality of diet and climate. Palaeogeogr. Palaeoclimatol. Palaeoecol. 342–343, 97–110. https://doi.org/10.1016/j.palaeo.2012.05.007 (2012).
Briatico, G., Bocherens, H., Bonnefille, R., Geraads, D., Mussi M. Isotopic insights into the Early Acheulean (1.95 Ma – 1.66 Ma) high-elevation paleoenvironments at Melka Kunture (Upper Awash Valley, Ethiopia). Archaeol. Anthropol. Sci., 15, 184, https://doi.org/10.1007/s12520-023-01879-1.
Harris, J. M., Cerling, T. E., Leakey, M. G. & Passey, B. H. Stable isotope ecology of fossil hippopotamids from the Lake Turkana Basin of East Africa. J. Zool. 275, 323–331. https://doi.org/10.1111/j.1469-7998.2008.00444.x (2008).
Eltringham, S. K. The Hippos (Academic Press, 1999).
Bocherens, H. & Drucker, D. Trophic level isotopic enrichment of carbon and nitrogen in bone collagen: Case studies from recent and ancient terrestrial ecosystems. Int. J. Osteoarchaeol. 13, 46–53. https://doi.org/10.1002/oa.662 (2013).
Bocherens, H., Koch, P. L., Mariotti, A., Geraads, D. & Jaeger, J.-J. Isotopic biogeochemistry (13C, 18O) of mammalian enamel from African Pleistocene hominid sites. Palaios 11, 306–318. https://doi.org/10.2307/3515241 (1996).
Clementz, M. T. & Koch, P. L. Differentiating aquatic mammal habitat and foraging ecology with stable isotopes in tooth enamel. Oecologia 129, 461–472. https://doi.org/10.1007/s004420100745 (2001).
Clementz, M. T., Holroyd, P. A. & Koch, P. L. Identifying aquatic habits of herbivorous mammals through stable isotope analysis. Palaios 23(9), 574–585. https://doi.org/10.2110/palo.2007.p07-054r (2008).
Tieszen, L. L., Senyimba, M. M., Imbamba, S. K. & Troughton, J. H. The distribution of C3 and C4 grasses and carbon isotope discrimination along an altitudinal and moisture gradient in Kenya. Oecologia 37, 337–350 (1979).
Braun, D. R. et al. Early hominin diet included diverse terrestrial and aquatic animals 195 Ma in East Turkana Kenya. PNAS 107(22), 10002–10007. https://doi.org/10.1073/pnas.1002181107 (2010).
Gerschlauer, F. et al. Stable carbon and nitrogen isotopic composition of leaves, litter, and soils of various ecosystems along an elevational and land-use gradient at Mount Kilimanjaro, Tanzania. Biogeosciences 16(2), 409–424. https://doi.org/10.5194/bg-16-409-2019 (2019).
Körner, C., Farquhar, G. D. & Wong, S. C. Carbon isotope discrimination by plants follows latitudinal and altitudinal trends. Oecologia 88, 30–40 (1991).
Dansgaard, W. Stable isotopes in precipitation. Tellus 16, 436–468 (1964).
Gonfiantini, R., Roche, M.-A., Olivry, J.-C., Fontes, J.-C. & Zuppi, G. M. The altitude effect on the isotopic composition of tropical rains. Chem. Geol. 181, 147–167. https://doi.org/10.1016/S0009-2541(01)00279-0 (2001).
Poage, M. A. & Chamberlain, C. P. Empirical relationships between elevation and the stable isotope composition of precipitation and surface waters: considerations for studies of paleoelevation change. Am. J. Sci. 301, 1–15. https://doi.org/10.2475/ajs.301.1.1 (2001).
Pederzani, S. & Britton, K. Oxygen isotopes in bioarchaeology: principles and applications, challenges and opportunities. . Earth-Sci. Rev. 188, 77–107. https://doi.org/10.1016/j.earscirev.2018.11.005 (2019).
Mussi, M., Altamura, F., Bonnefille, R., De Rita, D. & Melis, R. T. The environment of the Ethiopian highlands at the Mid Transition: Fauna, flora, and hominins in the 850–700 ka sequence at Gombore II (Melka Kunture). Quat. Sci. Rev. 149, 259–268. https://doi.org/10.1016/j.quascirev.2016.07.033 (2016).
Acknowledgements
We thank the Ethiopian Heritage Authority (EHA) and the National Museum of Ethiopia (Addis Ababa), which granted permission to sample the paleontological collection and export the samples. We are thankful to all the staff of the Museum for having facilitated the laboratory work in many ways. G.B. is personally grateful to Dr. Raymonde Bonnefille for her valuable comments on the palynology. We acknowledge support from the Open Access Publication Fund of the University of Tübingen (Germany). Finally, we thank the Editor, Prof. Randolph E. Donahue, and two anonymous reviewers for their constructive comments that greatly improved the article. This paper was revised when G.B. was a visiting researcher at the Department of Anthropology of the University of Connecticut (U.S.A.) in the Spring Semester 2024 as part of the Lady David postdoctoral program (2023-2024) at the Institute of Archaeology of the Hebrew University of Jerusalem (Israel).
Funding
Open Access funding enabled and organized by Projekt DEAL. This article is the outcome of the G.B.’s Ph.D. project supported by the Sapienza Università di Roma (Italy) (three-year scholarship), in co-tutorship with the Eberhard Karls University of Tübingen (Germany). The project was also funded by research grants provided by the Deutscher Akademischer Austauschdienst (DAAD) (Forschungsstipendien – Kurzstipendien 57552336), and the Sapienza Università di Roma (“Borse di mobilità all’estero”—Decreto N.16052020, Prot. N.0044296) awarded to G.B. The Hiwi contract provided by the Biogeology Research Group (University of Tübingen) and awarded to G.B. in November 2022 contributed to the preparation of additional samples. Stable isotope analyses were funded by the Biogeology Research Group, “Grandi Scavi di Ateneo”—grant SA11715C7C936C01 of Sapienza Università di Roma, and grant ARC-001666 of Ministero degli Affari Esteri e della Cooperazione Internazionale, awarded to M.M.
Author information
Authors and Affiliations
Contributions
G.B. designed the research and wrote the original manuscript with contributions from all authors (H.B., D.G., R.T.M., M.M.). G.B. collected and pretreated the samples and elaborated on the isotopic results. All authors discussed, revised, and approved the manuscript and agreed to its submission to the journal.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Briatico, G., Bocherens, H., Geraads, D. et al. The Pleistocene high-elevation environments between 2.02 and 0.6 Ma at Melka Kunture (Upper Awash Valley, Ethiopia) based upon stable isotope analysis. Sci Rep 14, 6619 (2024). https://doi.org/10.1038/s41598-024-56768-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-56768-x
- Springer Nature Limited